Predictive Value of Renal Resistive Index in Percutaneous Renal Interventions for Atherosclerotic Renal Artery Stenosis
Abstract: Background. The clinical benefit of percutaneous interventional therapies for atherosclerotic renal artery stenosis (ARAS) is still obscure. Randomized trials conducted on general patient populations provided unsatisfactory results in justifying the interventional treatment. In this study, the predictive value of renal resistive index (RRI) was retrospectively analyzed in identifying the patients who may benefit from renal angioplasty and stenting. Methods. The records of patients who underwent percutaneous intervention for ARAS were analyzed between 2006 and 2010; we compared the clinical outcomes with preprocedural RRI values. Seventy-three patients were included in the analysis. RRI is calculated as follows: RRI = 1 – (Vmin/Vmax). Patients with RRI ≤0.75 were grouped as low RRI (group I) and compared with high RRI patients (RRI >0.75, group II).The comparison was in follow-up systolic and diastolic blood pressures, blood creatinine levels, estimated glomerular filtration rate (eGFR) and need for anti-hypertensive therapy. Data were collected by a customized online database created using Research Electronic Data Capture (REDCap) application. Results. The mean follow-up was 12.4 (9-14) months and 11.1 (5-14) months for groups I and II, respectively (P=NS). After follow-up, group I patients significantly improved compared to baseline in their blood creatinine levels (2.0 ± 1.2 mg/dL vs 1.5 ± 0.60 mg/dL; P<.05), eGFR (45.2 ± 26.2 mL/min vs 51.6 ± 23.8 mL/min; P<.05), systolic blood pressure (143.6 ± 31.0 mm Hg vs 129.6 ± 18.4 mm Hg; P<.05), diastolic blood pressure (73.6 ± 13.4 mm Hg vs 69.5 ± 9.5 mm Hg; P<.05), and need for anti-hypertensive drugs (2.2 ± 0.9 vs 2.0 ± 0.9; P<.05). However, in group II, follow-up blood creatinine levels (1.8 ± 0.7 mg/dL vs 2.1 ± 1.0 mg/dL; P<.05) increased and eGFR (39.99 ± 22.53 mL/min vs 36.3 ± 23.2 mL/min; P<.05) decreased, indicating continuing clinical deterioration despite the intervention. Conclusion. RRI ≤0.75 may predict better clinical outcomes after renal angioplasty and stenting. Preprocedural RRI can be considered a useful parameter in defining patients who may benefit from interventional procedures.
J INVASIVE CARDIOL 2012;24(10):504-509
Key words: renal resistive index, angioplasty, stenting, creatinine, glomerular filtration rate, hypertension
_____________________________________________
Atherosclerotic renal artery stenosis (ARAS) is an increasingly recognized cause of chronic kidney disease with a death rate of about 16% per year, largely from cardiovascular disease.1,2 Stenosis of the renal artery is associated with hypertension and chronic kidney disease. With an aging population, the prevalence of renal artery stenosis is expected to increase. Options for treatment include anatomical correction of the stenosis, either surgically or percutaneously, and conservative therapy with management of high blood pressure and other atherosclerotic risk factors with medications. In the last decade, percutaneous interventional treatment has emerged as the first choice therapy, due to high technical success and relatively low complication rates.3 However, despite successful angiographic results, the clinical benefit of percutaneous therapy is still obscure. Small-scale randomized trials indicated little clinical improvement in blood pressure control and renal function in patients treated with percutaneous procedures compared to medical treatment, at the expense of increased procedural complications.4-6 A recently published study by the ASTRAL (Angioplasty and Stenting for Renal Artery Lesions) investigators also showed no evidence of a worthwhile clinical benefit in the initial years after revascularization in patients with ARAS.7 However, all these studies reflect real-life clinical practice without any specific patient selection criteria for the percutaneous procedures. Patient selection may have a major influence on the clinical outcomes.
In this study, the aim was to retrospectively analyze the Cleveland Clinic experience using renal resistive index (RRI) — a Doppler ultrasonography derived index — as a patient selection criteria and its effects on the clinical outcomes.
Methods
Definitions. Hypertension was defined as systolic blood pressure (SBP) ≥140 mm Hg or diastolic blood pressure (DBP) ≥90 mm Hg or the patient being on antihypertensive medications. Renal failure was diagnosed when serum creatinine was ≥1.5 mg/dL. A reduction of the trans-stenotic gradient to <15 mm Hg or residual lesion <50% was considered as technical success. Periprocedural complications (complications occurring within 24 hours after the procedure) including pulmonary edema, myocardial infarction, renal artery embolism, dissection, cholesterol embolism, kidney infarct, and access-site complications (bleeding, groin hematoma) were considered as procedure-related complications.
Patients. This is a non-randomized analysis of consecutive patients who had renal artery intervention for ARAS at our clinic. A retrospective analysis of records from patients who underwent percutaneous intervention for ARAS between 2006 and 2010 was performed. Patients with fibromuscular dysplasia, transplanted kidneys, age <18 years at first renovascular intervention, aortic dissection with renal artery involvement, or previous renovascular surgery were excluded. Eighty-seven patients underwent renal angioplasty and stenting for ARAS during this time period at the Cleveland Clinic Department of Cardiovascular Medicine.
Sones Catheterization Laboratories. The decision to proceed with renal intervention was mainly based on clinical findings. Patients with recurrent congestive heart failure (CHF), recurrent sudden onset unexplained pulmonary edema, malignant, accelerated or resistant hypertension, elevated creatinine (creatinine ≥1.5 mg/dL), or decreased estimated glomerular filtration rate (eGFR) (based on Cockcroft-Gault 5 formula)8 were considered as symptomatic patients eligible for percutaneous intervention. Renal artery stenosis in the absence of clinical symptoms but in the presence of: (a) greater than or equal to 50%-70% diameter stenosis by visual estimation with a peak translesional gradient (measured with a diagnostic catheter or pressure wire) of greater than or equal to 20 mm Hg or a mean gradient greater than or equal to 10 mm Hg; (b) any stenosis greater than or equal to 70% diameter stenosis (either angiographically or by intravascular ultrasound measurement) was classified as hemodynamically significant asymptomatic (incidental) renal artery stenosis and considered an indication for interventional treatment. Of those 87 patients who had interventional treatment for ARAS, 6 were lost to follow-up and 8 did not have preprocedural RRI measurement and were excluded from the study. Seventy-three patients who had complete preprocedural renal duplex ultrasound work-up and subsequent follow-up data were included in the study. For comparisons, patients were divided into two groups. Group I included patients with low RRI (≤0.75; n = 44) and group II included patients with high RRI (>0.75; n = 44).
Data collection. Electronic patient charts were reviewed using Epic Mypractice (Epic Systems Corporation) software interface. Data were collected by a customized online database created using Research Electronic Data Capture (REDCap) application. REDCap is a secure, web-based application for building and managing online databases and provides a novel methodology and software solution for rapid development and deployment of electronic data capture tools to support clinical research.9 All data about baseline patient characteristics, procedure, complications, and follow-up were entered into the REDCap database specifically designed for that study. Web-based access to the database allowed multi-user data entry in a short period of time. The follow-up data are based on clinical parameters 1 year after the procedure. Complication data refer to procedural complications and complications during the hospital stay.
Renal duplex ultrasound. The duplex scans were performed with an ultrasound machine (Logiq E9, GE Healthcare) with the use of either a multi-frequency curved-array transducer (2-4 MHz) or a multi-frequency sector transducer (2-3 MHz) with a 2.5 MHz pulsed Doppler frequency and a focal zone at the depth of the renal arteries. An interlobar artery located in the upper, middle, and lower third of the kidney was measured using color Doppler imaging for RRI analysis. The peak systolic velocity (Vmax, in centimeters per second) and the end-diastolic velocity (Vmin, in centimeters per second) were measured in order to calculate the dimensionless RRI values using the formula: RRI = 1 – (Vmin/Vmax). The average of three measurements was entered into the database as the RRI value.
Percutaneous renal angioplasty and stenting. The intervention was performed according to a technique previously described.10,11 All patients received stents and distal protection device was not used in any of the cases. If angiographic lesion diameter was questionable for justifying an intervention, additional studies using intravascular ultrasound or pressure wire were employed. Occluded renal arteries and non-functioning kidneys were not treated. Patients with creatinine ≥1.5 mg/mL were hydrated overnight with normal saline and received N-acetyl cysteine 600 mg twice a day orally 24 hours preoperatively and for 48 hours postoperatively.
Statistical analysis. Analyses were performed using a statistical software (Statistica version 8.0, StatSoft Inc). Unless stated otherwise, all data were expressed as means ± standard deviation. Independent sample t-test was used for evaluating between the groups variation and dependent sample t-test was used for assessing the correlation of preprocedural data with clinical outcome. P-value less than .05 was considered statistically significant. All P-values were derived from two-tailed tests. One-way ANOVA was used to analyze single categorical variables.
Product-moment and partial correlations analysis was used to test the variables for linear relationship. Factorial ANOVA was used to analyze the interactive effects of multiple categorical variables. Chi-square test was used to analyze the relation between categorical variables.
Results
 Data from 73 eligible patients were included in the analyses. RRI was ≤0.75 in 44 patients and these constituted group I, while 23 patients had RRI >0.75 and constituted group II. Other than the prevalence of diabetes and blood glucose levels, there were no differences in the baseline demographic and clinical features of the patients (Table 1). The proportion of patients with diabetes in group I and II was 27% (n = 12) and 62% (n = 18), respectively, and the 35% difference in the prevalence of diabetes between the groups was statistically significant (95% CI, 11.6%-53.6%; P<.05). The blood glucose levels in groups I and II were 109.43 ± 42.5 mg/dL and 133.76 ± 42.4 mg/dL, respectively (95% CI, 4.3-43.7 mg/dL; P<.05). One-way ANOVA testing revealed diabetics had significantly higher RRI values (0.751 ± 0.06; n = 30) compared to non-diabetics (0.7 ± 0.09; n = 43) in the overall patient cohort (P<.05).
Data from 73 eligible patients were included in the analyses. RRI was ≤0.75 in 44 patients and these constituted group I, while 23 patients had RRI >0.75 and constituted group II. Other than the prevalence of diabetes and blood glucose levels, there were no differences in the baseline demographic and clinical features of the patients (Table 1). The proportion of patients with diabetes in group I and II was 27% (n = 12) and 62% (n = 18), respectively, and the 35% difference in the prevalence of diabetes between the groups was statistically significant (95% CI, 11.6%-53.6%; P<.05). The blood glucose levels in groups I and II were 109.43 ± 42.5 mg/dL and 133.76 ± 42.4 mg/dL, respectively (95% CI, 4.3-43.7 mg/dL; P<.05). One-way ANOVA testing revealed diabetics had significantly higher RRI values (0.751 ± 0.06; n = 30) compared to non-diabetics (0.7 ± 0.09; n = 43) in the overall patient cohort (P<.05).
 Indications for percutaneous intervention are listed in Table 2. In either of the groups, the main indication for intervention was clinical symptoms and signs including recurrent congestive heart failure (CHF), recurrent sudden onset unexplained pulmonary edema, malignant, accelerated or resistant hypertension, and elevated creatinine levels (creatinine ≥1.5 mg/dL). In group I, 25% (n = 11) of the patients received the procedure solely based on angiographic and/or invasive hemodynamic measurements without any accompanying clinical symptoms. In group II, the proportion of asymptomatic patients received the procedure was 6.9%. The Chi-square test revealed that 18.1% difference between the groups is statistically significant (x2 = 3.91; df = 1; P<.05).
Indications for percutaneous intervention are listed in Table 2. In either of the groups, the main indication for intervention was clinical symptoms and signs including recurrent congestive heart failure (CHF), recurrent sudden onset unexplained pulmonary edema, malignant, accelerated or resistant hypertension, and elevated creatinine levels (creatinine ≥1.5 mg/dL). In group I, 25% (n = 11) of the patients received the procedure solely based on angiographic and/or invasive hemodynamic measurements without any accompanying clinical symptoms. In group II, the proportion of asymptomatic patients received the procedure was 6.9%. The Chi-square test revealed that 18.1% difference between the groups is statistically significant (x2 = 3.91; df = 1; P<.05).
The technical success was 100% in both groups. Only 2 patients (1 in each group, P=NS) suffered from minor access-site complications.
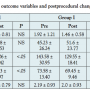 The mean follow-up was 12.4 months (range, 9-14 months) and 11.1 months (range, 5-14 months) for groups I and II, respectively (P=NS). Changes in the outcome measures are listed in Table 3. In the general patient population, only systolic and diastolic blood pressures were found to be improved significantly after the procedure. However, there was no significant change in blood creatinine levels and eGFR at the end of the follow-up period. Subgroup analysis revealed significant clinical improvement in terms of blood creatinine levels (Figure 1A), eGFR (Figure 1C), systolic and diastolic blood pressures and number of anti-hypertensive drugs required to keep the blood pressure under control, in group I. However, in group II, blood creatinine levels increased (Figure 1B) and eGFR decreased (Figure 1D) significantly despite the intervention, indicating an ongoing clinical deterioration. There was no evidence of benefit in systolic and diastolic blood pressures and the number of anti-hypertensive drugs required in group II.
The mean follow-up was 12.4 months (range, 9-14 months) and 11.1 months (range, 5-14 months) for groups I and II, respectively (P=NS). Changes in the outcome measures are listed in Table 3. In the general patient population, only systolic and diastolic blood pressures were found to be improved significantly after the procedure. However, there was no significant change in blood creatinine levels and eGFR at the end of the follow-up period. Subgroup analysis revealed significant clinical improvement in terms of blood creatinine levels (Figure 1A), eGFR (Figure 1C), systolic and diastolic blood pressures and number of anti-hypertensive drugs required to keep the blood pressure under control, in group I. However, in group II, blood creatinine levels increased (Figure 1B) and eGFR decreased (Figure 1D) significantly despite the intervention, indicating an ongoing clinical deterioration. There was no evidence of benefit in systolic and diastolic blood pressures and the number of anti-hypertensive drugs required in group II.
 Since RRI values do not have a linear relationship with the outcome variables, they are treated as categorical variables (low RRI [group I] vs high RRI [group II]). Chi-square test revealed that patients with diabetes tend to have higher RRI values (x2 = 8.7; df = 1; P<.05). Any interaction between diabetes and RRI on follow-up renal functions was analyzed with factorial ANOVA. The analysis revealed no significant interaction between RRI category and the presence of diabetes on the outcome in renal functions (creatinine and eGFR) (F(2.58) = 2.4285; P>.05). RRI category was still a significant predictor of renal functions after adjusting the means against the presence of diabetes (F(2.58) = 5.1281; P<.05).
Since RRI values do not have a linear relationship with the outcome variables, they are treated as categorical variables (low RRI [group I] vs high RRI [group II]). Chi-square test revealed that patients with diabetes tend to have higher RRI values (x2 = 8.7; df = 1; P<.05). Any interaction between diabetes and RRI on follow-up renal functions was analyzed with factorial ANOVA. The analysis revealed no significant interaction between RRI category and the presence of diabetes on the outcome in renal functions (creatinine and eGFR) (F(2.58) = 2.4285; P>.05). RRI category was still a significant predictor of renal functions after adjusting the means against the presence of diabetes (F(2.58) = 5.1281; P<.05).
Discussion
This study indicates that lower preprocedural RRI values (≤0.75) predict better clinical outcomes after percutaneous treatment for ARAS compared to higher preprocedural RRI values. Patients with lower preprocedural RRI values (≤0.75) showed marked improvement in renal function after the interventional treatment. In contrast, at follow-up, patients with higher preprocedural RRI values (>0.75) showed clinical deterioration in renal function despite the procedure (Figure 1). High RRI values were associated with presence of diabetes. However, multivariate analysis revealed the predictive effect of RRI is independent from diabetes. Small randomized trials have shown no significant benefits of percutaneous interventional treatment, either with or without stent implantation, over medical treatment in patients with ARAS.4-6,12 The recently published ASTRAL trial (Angioplasty and Stenting for Renal Artery Lesions) randomized 806 patients with ARAS to undergo revascularization in addition to receiving medical therapy or to receive medical therapy alone.7 The outcome measures of this study was serum creatinine levels, blood pressure, time to first renal event, time to first cardiovascular event, and mortality. After 5-year follow-up, the investigators did not find any evidence of a worthwhile clinical benefit from revascularization compared to medical therapy. Given the invasive nature of the interventional procedures, the results of the randomized trials hardly justifies percutaneous treatment. However, all those randomized studies reflect real-life clinical practice in which no criteria is employed to preselect the patients who may benefit more from the interventional therapy. There is an obvious need for predictive factors to identify patients who are likely to respond an interventional approach.
The earliest and most pronounced pathologic features in renal ischemia consist of tubulo-interstitial changes that may mediate the progression of renal disease towards irreversible damage.13,14 In addition to tubulo-interstitial injury, patients with ARAS exhibit intra-renal microvascular disease leading to microvascular remodeling (increase in wall/lumen ratio) and rarefaction (reduced number or combined length of peritubular capillaries within vascular beds) in the early phase of ischemic renal disease. Decreased renal parenchymal perfusion leads to glomerulosclerosis, tubulo-interstitial fibrosis, and/or vascular sclerosis.15 Glomerulosclerosis is a relatively late event in ARAS and often linked to long duration, pre-existing injury, and exacerbating co-morbid conditions, like dyslipidemia and atherosclerosis. In ARAS renal decompensation occurred late in the course of the disease, and the diagnosis was generally delayed till the disease became clinically manifest. One possible reason for a poor response to interventional treatment may be structural alterations in the renal microvasculature and parenchyma, induced by long standing disease. Decreased renal parenchymal perfusion leads to ischemic nephropathy, which causes glomerular atrophy, tubulo-interstitial lesions and fibrosis.16 Besides the ischemia induced by the stenotic lesion, associated conditions like prolonged hypertension and diabetes may cause irreversible renal injury before the intervention which may limit the benefits of a revascularization procedure.17,18
RRI is a simple parameter that can be derived from a renal ultrasound Doppler spectrum. The index describes the percentage reduction of end-diastolic blood flow (Vmin) in the investigated vessel (interlobar artery in that particular study) in relation to maximal systolic blood flow (Vmax): resistive index = (Vmax – Vmin)/Vmax. It is considered as a marker of downstream vascular impedance and therefore has been suggested as a measure of renal arterial stiffness.19 The index was found to be correlated with glomerular sclerosis, tubulo-interstitial changes, and arteriolosclerosis in renal parenchymal disease.20
Radermacher et al tested RRI as a predictor of outcomes in a study conducted among 131 patients who underwent corrective surgery or angioplasty.21 Authors suggested, RRI value of less than 0.8 is a reliable marker to identify patients in whom angioplasty or surgery is likely to improve renal function, blood pressure, or kidney survival. However, only 42 patients received renal stenting in that study. In a recently published retrospective analysis of 592 renal interventions, parenchymal volume preservation after the renal interventions was assessed as an outcome measure after renal interventions.22 In this series parenchymal volume loss occurred in one-third of patients and higher RRI values are (0.75 ± 0.9 vs 0.73 ± 0.10; P=.0001) found to be correlated with renal parenchymal loss even after a successful procedure.
In contrast to larger retrospective series which analyzed data spread over two decades,22,23 this study included patients starting from 2006 to 2010 and all patients received stents in addition to angioplasty, which is more parallel to the current intervention practice. This study also encompass a relatively shorter time span compared to other retrospective series and the standard medical management of patients with ARAS did not change significantly throughout the study, which may otherwise present as a confounding factor.
Excluding diabetes and mean blood glucose levels, baseline clinical characteristics of our patients were similar between the patient groups. Prevalence of diabetes and mean blood glucose level was significantly higher in group II (P<.05). This is in compliance with the series reported by Radermacher et al.21 In that study patients with high RRI (≥0.8) had significantly higher prevalence of diabetes. However unlike our study their study groups were not age matched. Both diabetes and age have significant effects on renal functions which may affect the results.24,25 A factorial ANOVA analysis was performed to test for any interaction between diabetes and RRI on follow-up renal functions (creatinine and eGFR). The analysis revealed no significant interaction between RRI category and presence of diabetes on the outcome of renal function (F(2.58) = 2.4285; P>.05). RRI category was still a significant predictor of renal function after adjusting the means against the presence of diabetes (F(2.58)= 5.1281; P<.05).
Another important finding in this study was the difference in the indications of the procedure between the groups. The chi-square test revealed that significantly more (18.1%) asymptomatic patients were treated with balloon angioplasty and stenting in the low RRI group (P<.05). The rationale of the intervention in the asymptomatic patients was (a) greater than or equal to 50% to 70% diameter stenosis by visual estimation with a peak translesional gradient (measured with a diagnostic catheter or pressure wire) of greater than or equal to 20 mm Hg or a mean gradient greater than or equal to 10 mm Hg, (b) any stenosis greater than or equal to 70% diameter stenosis (either angiographically or by intravascular ultrasound measurement). These patients received the interventional treatment solely based on the hemodynamic significance of the lesions. The chi-square analysis showed that symptom status of the patients and RRI values are associated. While asymptomatic patients tend to have a lower RRI values, symptomatic patients generally had higher RRI values (x2=3.91; df=1; P<.05). This finding suggests that the renal function of those patients was still intact to compensate for the effects of stenosis which represents the earlier stages of the disease.
The pathogenesis of chronic kidney disease in ARAS is not fully understood. A hemodynamically significant lesion can undoubtedly lead to a decrease in renal perfusion, however atherosclerotic nephropathy seems more than an ischemic disease caused by a vascular stenosis. ARAS is frequently associated with other systemic diseases and vascular pathologies. Renal parenchymal damage may occur long before the stenosis become hemodynamically significant. Evidence suggest that pre-existing chronic parenchymal damage plays an important role in the pathogenesis of renal dysfunction in ARAS.1,26 Factors other than the severity of the vascular lesions are likely to contribute to the disease progression.27 In this study, chi-square analysis revealed that patients with diabetes tend to have higher RRI values (x2 = 8.7; df=1; P<.05). This finding is consistent with the previous studies.17 Hamano et al studied RRI values of 150 patients with type 2 diabetes and without clinically overt renal artery stenosis and proposed RRI may indicate the presence of any type of underlying renal damage in type II diabetes.28 In this study, group II patients had higher RRI values indicative of advanced stage renal parenchymal damage. Besides, diabetes is a very important co-morbidity, causing renal parenchymal damage which is more prevalent in group II. Overall group II consisted those with advanced nephropathy who are less likely to benefit from revascularization. Presence and extent of nephropathy is indicative of established damage, therefore revascularization is less likely to benefit. Patients with ischemia but minimal or no nephropathy would benefit most from revascularization, while those with advanced nephropathy would be less likely to benefit.29 Clinical evaluation for nephropathy may include urinalysis, blood tests for renal function, and renal duplex ultrasound assessment. In our study, better outcomes achieved in low RRI patients may be derived from the lack of associated nephropathies, particularly diabetic nephropathy.
Conclusion
Current evidence-based data do not justify interventional approaches as a first-line treatment option for ARAS. However, ARAS is the clinical end-point of multiple etiologies and pathobiological pathways. Associated renal parenchymal injury adversely affects the clinical outcomes of an interventional procedure. This study suggests early intervention and lack of associated parenchymal injury may provide better outcomes and interventional treatment should be reserved to that patient subgroup. Patients having an associated nephropathic condition like diabetes and suffering from longstanding renal parenchymal injury are less responsive to revascularization. Medical treatment may provide better results in those patients. RRI can be used as a reliable indicator for predicting better short term outcomes and can be used to identify patients who are likely to benefit from an interventional treatment. Future randomized studies with long term follow-up should use such renal parenchymal injury indices to evaluate the efficacy of interventions in ARAS patients.
Study limitations. This is a retrospective study without a control group. The data is derived from a tertiary referral center catheterization laboratory. Most of the patients were evaluated by their primary physicians and referred to our hospital because they were considered eligible for the intervention. Therefore there is a strong selection bias towards successful interventional treatment. As in all diseases the most important outcome is survival which has not been evaluated in this study because of the short follow-up period. However, the medical treatment of conditions associated with ARAS has changed considerably during the last decades and stenting of the stenosis after the angioplasty became an integral part of the procedure in the last years. Including older patient data to achieve a larger group may have caused significant heterogeneities in those aspects. Long term randomized trials are required to evaluate the clinical implications of the findings.
References
- Cheung CM, Wright JR, Shurrab AE, et al. Epidemiology of renal dysfunction and patient outcome in atherosclerotic renal artery occlusion. J Am Soc Nephrol. 2002;13(1):149-157.
- Kalra PA, Guo H, Kausz AT, et al. Atherosclerotic renovascular disease in United States patients aged 67 years or older: risk factors, revascularization, and prognosis. Kidney Int. 2005;68(1):293-301.
- Weibull H, Bergqvist D, Bergentz SE, Jonsson K, Hulthen L, Manhem P. Percutaneous transluminal renal angioplasty versus surgical reconstruction of atherosclerotic renal artery stenosis: a prospective randomized study. J Vasc Surg. 1993;18(5):841-850; discussion, pp. 50-52.
- Plouin PF, Chatellier G, Darne B, Raynaud A. Blood pressure outcome of angioplasty in atherosclerotic renal artery stenosis: a randomized trial. Essai Multicentrique Medicaments vs Angioplastie (EMMA) Study Group. Hypertension. 1998;31(3):823-829.
- van Jaarsveld BC, Krijnen P, Pieterman H, et al. The effect of balloon angioplasty on hypertension in atherosclerotic renal-artery stenosis. Dutch Renal Artery Stenosis Intervention Cooperative Study Group. N Engl J Med. 2000;342(14):1007-1014.
- Webster J, Marshall F, Abdalla M, et al. Randomised comparison of percutaneous angioplasty vs continued medical therapy for hypertensive patients with atheromatous renal artery stenosis. Scottish and Newcastle Renal Artery Stenosis Collaborative Group. J Hum Hypertens. 1998;12(5):329-335.
- Wheatley K, Ives N, Gray R, et al. Revascularization versus medical therapy for renal-artery stenosis. N Engl J Med. 2009;361(20):1953-1962.
- Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron. 1976;16(1):31-41.
- Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap) — a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377-381.
- Geyskes GG. Treatment of renovascular hypertension with percutaneous transluminal renal angioplasty. Am J Kidney Dis. 1988;12(4):253-265.
- Rees CR, Palmaz JC, Becker GJ, et al. Palmaz stent in atherosclerotic stenoses involving the ostia of the renal arteries: preliminary report of a multicenter study. Radiology. 1991;181(2):507-514.
- Ives NJ, Wheatley K, Stowe RL, et al. Continuing uncertainty about the value of percutaneous revascularization in atherosclerotic renovascular disease: a meta-analysis of randomized trials. Nephrol Dial Transplant. 2003;18(2):298-304.
- Greco BA, Breyer JA. Atherosclerotic ischemic renal disease. Am J Kidney Dis. 1997;29(2):167-187.
- Shanley PF. The pathology of chronic renal ischemia. Semin Nephrol. 1996;16(1):21-32.
- Meyrier A. Renal vascular lesions in the elderly: nephrosclerosis or atheromatous renal disease? Nephrol Dial Transplant. 1996;11(Suppl 9):45-52.
- Dean RH, Tribble RW, Hansen KJ, O’Neil E, Craven TE, Redding JF 2nd. Evolution of renal insufficiency in ischemic nephropathy. Ann Surg. 1991;213(5):446-455; discussion pp. 55-56.
- Marzano MA, Pompili M, Rapaccini GL, et al. Early renal involvement in diabetes mellitus: comparison of renal Doppler US and radioisotope evaluation of glomerular hyperfiltration. Radiology. 1998;209(3):813-817.
- Ubara Y, Hara S, Katori H, Yamada A, Morii H. Renovascular hypertension may cause nephrotic range proteinuria and focal glomerulosclerosis in contralateral kidney. Clin Nephrol. 1997;48(4):220-223.
- Mostbeck GH, Kain R, Mallek R, et al. Duplex Doppler sonography in renal parenchymal disease. Histopathologic correlation. J Ultrasound Med. 1991;10(4):189-194.
- Ikee R, Kobayashi S, Hemmi N, et al. Correlation between the resistive index by Doppler ultrasound and kidney function and histology. Am J Kidney Dis. 2005;46(4):603-609.
- Radermacher J, Chavan A, Bleck J, et al. Use of Doppler ultrasonography to predict the outcome of therapy for renal-artery stenosis. N Engl J Med. 2001;344(6):410-417.
- Davies MG, Saad WE, Bismuth J, Naoum JJ, Peden EK, Lumsden AB. Renal parenchymal preservation after percutaneous renal angioplasty and stenting. J Vasc Surg. 2010;51(5):1222-1229; discussion, p. 9.
- Bonelli FS, McKusick MA, Textor SC, et al. Renal artery angioplasty: technical results and clinical outcome in 320 patients. Mayo Clin Proc. 1995;70(11):1041-1052.
- Rowe JW, Andres R, Tobin JD, Norris AH, Shock NW. The effect of age on creatinine clearance in men: a cross-sectional and longitudinal study. J Gerontol. 1976;31(2):155-163.
- Afsar B, Elsurer R, Sezer S, Ozdemir FN. Insulin resistance is associated with increased renal resistive index independent of other factors in newly diagnosed type 2 diabetes mellitus and hypertensive patients. Metabolism. 2010;59(2):279-284.
- Wright JR, Duggal A, Thomas R, Reeve R, Roberts IS, Kalra PA. Clinicopathological correlation in biopsy-proven atherosclerotic nephropathy: implications for renal functional outcome in atherosclerotic renovascular disease. Nephrol Dial Transplant. 2001;16(4):765-770.
- Suresh M, Laboi P, Mamtora H, Kalra PA. Relationship of renal dysfunction to proximal arterial disease severity in atherosclerotic renovascular disease. Nephrol Dial Transplant. 2000;15(5):631-636.
- Hamano K, Nitta A, Ohtake T, Kobayashi S. Associations of renal vascular resistance with albuminuria and other macroangiopathy in type 2 diabetic patients. Diabetes Care. 2008;31(9):1853-1857.
- Safian RD, Madder RD. Refining the approach to renal artery revascularization. JACC Cardiovasc Interv. 2009;2(3):161-174.
_____________________________________________
*Joint first authors.
From the Department of Cardiovascular Medicine, Cleveland Clinic, Cleveland, Ohio.
Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. The authors report no conflicts of interest regarding the content herein.
Manuscript submitted January 26, 2012, provisional acceptance given February 20, 2012, final version accepted April 17, 2012.
Address for correspondence: Samir R. Kapadia, MD, FACC, Professor of Medicine, Director, Sones Cardiac Catheterization Lab, Director, Interventional Cardiology Fellowship, Cleveland Clinic. Email: kapadis@ccf.org
















