Percutaneous Closure of a Post-traumatic Muscular Ventricular Septal Defect Using the Amplatzer Duct Occluder
February 2005
Post-traumatic ventricular septal defect (VSD) after penetrating cardiac trauma is infrequent, with an estimated incidence of 1–4.5% of cases.1,3 The decision to close a traumatic VSD is similar to that of a non-traumatic VSD and is based on the degree of shunting, the patient’s symptoms, and cardiac hemodynamics. We report a case of post-traumatic muscular VSD in an 18-year-old after sustaining a penetrating stab wound to the chest. The patient developed symptoms of shortness of breath and exercise intolerance due to a moderately sized VSD with significant left-to-right shunting. Successful percutaneous closure was achieved with a 10–8 mm Amplatzer duct occluder (ADO).
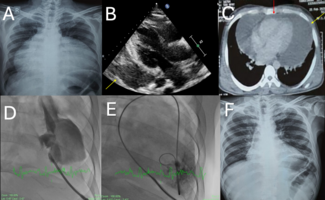
Case Report. An 18-year-old male sustained a stab wound to the chest one year prior to admission. At that time, he underwent emergent cardiac surgery with suture closure of a right ventricular (RV) laceration. His recovery was uncomplicated. He remained relatively asymptomatic until one year post-operatively when he experienced shortness of breath, fatigue, and dyspnea on exertion with competitive high school athletics. Physical examination revealed an afebrile adolescent male with normal vital signs. Cardiac auscultation revealed a loud, holosystolic murmur, best heard at the left sternal border. Transthoracic echocardiography (TTE) demonstrated the presence of a restrictive muscular ventricular septal defect (VSD) (Figure 1 A & B). It was felt that this VSD likely occurred at the time of the stab wound but remained undiagnosed at the time of initial surgery. Over time, a significant shunt developed and the patient developed symptoms of heart failure. The left ventricle was mildly reduced in function with a shortening fraction of 27% and was mildly enlarged with an end-diastolic dimension of 58 mm. The non-invasively estimated Qp:Qs ratio by TTE was 1.9:1.0. The right ventricle was mildly dilated with a mid-cavity minor axis of 4.6 cm and had decreased function. Pulmonary pressures were estimated to be mildly elevated.
Transesophageal echocardiography (TEE) confirmed the VSD location in the infero-posterior segment of the basal interventricular septum. Real-time three-dimensional transthoracic imaging revealed the serpiginous nature of the VSD track (Figure 2). The patient was referred for an attempt at percutaneous device closure of the VSD.
Procedure. Under general endotracheal anesthesia with continuous TEE monitoring, right and left heart catheterization was performed via a 5 Fr sheath in the right femoral artery and a 6 Fr sheath in the right femoral vein. An 8 Fr sheath was placed in the right internal jugular vein. The hemodynamic study revealed that the pulmonary artery pressure was mildly elevated (42% of systemic) with main pulmonary artery pressures of 34/16 mmHg and mean of 20 mmHg
An angiogram of the left ventricle (LV), performed in the hepatoclavicular projection (35º LAO, 35º cranial) revealed the presence of a moderate sized muscular VSD in the infero-posterior portion of the ventricular septum, measuring 5.3–7 mm with left-to-right shunt (Figure 3 A). The VSD was crossed from the LV side using a 5 Fr Judkins right coronary catheter. Then a 0.035 inch Terumo Glidewire™ (Terumo Medical Corp., Somerset, New Jersey) was advanced via the catheter and manipulated to the main pulmonary artery. The Judkins catheter was advanced over this wire to the main pulmonary artery. The Glidewire was removed and a 0.035 inch exchange length guidewire was advanced through this catheter to the main pulmonary artery. A 6 Fr gooseneck snare (Microvena Corp., White Bear Lake, Minnesota) was advanced via the right internal jugular vein to the main pulmonary artery.
The guidewire was snared in the main pulmonary artery and exteriorized out the right internal jugular vein forming an arteriovenous wire loop. Over this wire, an 8 Fr Mullins type delivery sheath (Cook Inc., Bloomington, Indiana) was advanced from the right internal jugular vein to the right ventricle, VSD, LV and up into the ascending aorta. The dilator was removed and the wire was exchanged for the Glidewire which was snared and exteriorized out the femoral artery. This was done to prevent kinking of the sheath at the septum once the dilator was removed (Figure 3B). Next, a 10–8 mm ADO (AGA Medical Corp., Golden Valley, Minnesota) was loaded and advanced beside the Glidewire from the internal jugular vein until it reached the tip of the sheath. The sheath was withdrawn into the cavity of the LV. The retention disk of the device was then deployed in the LV. At that time, it was safe to remove the Glidewire. The whole assembly (device, cable, and delivery sheath) was withdrawn until the disk was against the ventricular septum (Figure 3C). A repeat LV angiogram confirmed good device position (Figure 3D). Next, the tubular part of the device was deployed inside the septum with continuous traction until the narrowest part of the device was in the RV side of the septum.
Repeat LV angiogram and TEE confirmed good device position (Figure 3E). The device was released using the pin vise. Repeat LV angiogram demonstrated good device position and complete closure of the VSD (Figure 3F). Final TEE demonstrated good device position with no residual shunt and no AV valve regurgitation. Catheters were removed and hemostasis was achieved by direct compression. The patient received one gram cephazolin at the beginning of the procedure and two doses eight hours apart. The following day, repeat TTE demonstrated good device position with no residual shunt, improved LV function, and a LV end-diastolic dimension of 52 mm. The patient was discharged home on 81 mg aspirin per day for six months and was instructed to receive bacterial endocarditis precautions when needed.
Discussion
Ventricular septal defects can occur as congenital malformations or can be acquired. Acquired VSDs are very rare and most often develop as a mechanical complication of a myocardial infarction, or much less frequently, secondary to trauma. Post-traumatic VSDs have been reported to occur after both blunt and penetrating chest trauma. Information on the presentation and treatment options and the extent of supporting literature are variable depending on the type of VSD.
We hereby describe a case of post-traumatic VSD as a result of a penetrating stab wound which likely occurred at the initial time of injury, but remained unrecognized at the time of initial surgery. During the past year, the left-to-right shunt progressed, likely causing symptoms of shortness of breath. The use of real-time, three-dimensional echocardiography established the diagnosis of an infero-postero basal VSD and revealed its serpiginous course (Figure 3B). Hemodynamic assessment confirmed a significant shunt fraction and the patient was referred for device closure with an Amplatzer Duct Occluder after review of the information derived from the three-dimensional study.
The mechanisms of development of a VSD after blunt trauma include deceleration injury causing myocardial infarction from an intimal coronary artery tear (resulting in spasm, thrombosis, or dissection), or compression of the heart between the sternum and the spine, causing cardiac contusion.1,2 The blunt force of the impact itself, high intra-thoracic pressure, or the subsequent myocardial contusion or infarct may cause rupture of the septum at its thinnest, apical segment. VSDs secondary to contusion or infarction typically occur after a time delay during which constant shear and tension on the weakened infarct or peri-infarct zones via contraction of adjacent healthy myocardium occur. On the other side of the spectrum, a post-traumatic VSD occurring secondary to a stab or puncture wound, as described in this patient, often causes direct, immediate damage, typically has a serpiginous course, and often occurs in the muscular portion of the interventricular septum. Finally, congenital VSDs differ greatly in that the incidence is much higher than post-infarct or post-traumatic VSDs, and that the peri-membranous septum is the site of shunt in 75–80% of congenital cases.
The decision regarding the need for closure depends on the size of the defect, the degree of left-to-right shunting, symptoms of heart failure, and the presence of pulmonary hypertension. Limited experience suggests that small traumatic VSDs may be managed conservatively because the defect may spontaneously close over time.4 However, moderate-to-large VSDs, with evidence of pulmonary-to-systemic shunt ratio > 1.5:1.0, or the presence of symptoms, should be considered for closure.3
In the past, open surgical closure was commonly performed with considerable morbidity. With the development of two new devices, the Amplatzer muscular VSD occluder and the Amplatzer membranous occluder, the potential for successful percutaneous closure of VSDs exists with significantly less morbidity and very low mortality.13 The devices differ in the configuration of the retention disks and the interposed waist, and therefore the anatomic location of use. The muscular occluder has a thick waist (7–10 mm), with both LV and RV retention disks making it ideal to traverse the thick interventricular septum. The membranous occluder, however, has a narrow waist (1.5 mm) with offset RV and LV disks to accommodate the normal apical displacement of the tricuspid valve. The septal or ASD occluder has a 3–4 mm waist, and the LA and RA disks are much larger. Finally, the ductal occluder used in this reported case has no RV retention disk and is comprised only of an LV disk attached to an 8 mm waist which occluded the VSD. The lack of RV retention disk avoids any potential impingement on the TV apparatus. There is a growing body of literature on the use of these different percutaneous closure devices for the treatment of VSDs.
Use of the Amplatzer membranous VSD occluder has been shown to be a safe and effective alternative to surgical closure of peri-membranous congenital VSDs, with a design that avoids impingement of the device on the aortic and tricuspid valves.7,10–11 Three-dimensional echocardiography can help assess the likelihood of successful closure, evaluating for the presence of at least a 2 mm rim of tissue between the aortic valve and the defect. The Amplatzer muscular VSD occluder device has been well described as an excellent alternative to surgical closure of muscular VSDs from both congenital and ischemic etiologies.5–7,9
Reports of transcatheter occlusion of post-traumatic VSDs, on the other hand, have been rare. Three cases of successful closure of post-traumatic muscular VSDs due to blunt trauma have been reported. Two of the patients had undergone prior open surgical repair and upon finding a residual shunt, underwent percutaneous closure with an Amplatzer ASD septal occluder. The third, an adolescent, developed a post-traumatic VSD after sustaining a blunt trauma with crush injuries to the chest during an all-terrain vehicle accident. A post-infarct muscular VSD occluder was used to successfully close the ruptured apical ventricular septum.1,3,12
The case described here is the first, to our knowledge, to have a post-traumatic VSD due to penetrating trauma successfully closed with an Amplatzer duct occluder, and with the use of three-dimensional echocardiographic guidance.8 Three-dimensional imaging aided in localizing the VSD in close proximity to the tricuspid valve, allowing for adequate selection of the ductal occluder to avoid impingement of the tricuspid apparatus. The ADO is an ideal device to close muscular VSDs in the postero-inferior basal portion of the heart due to the lack of a RV retention disk.
At the last follow-up six weeks post-procedure, the patient reported no symptoms of shortness of breath, lower extremity edema, or exercise intolerance. Limited transthoracic echocardiography revealed no residual shunt.
1. Pesenti-Rossi D, Godart F, Dubar A, Rey C. Transcatheter closure of traumatic ventricular septal defect: An alternative to surgery. Chest 2003:123;2144–2145.
2. Genoni M, Jenni R, Turina M. Traumatic ventricular septal defect. Heart 1997;78:316–318.
3. Bauriedel G, Redel DA, Schmitz C, et al. Transcatheter closure of a posttraumatic ventricular septal defect with an amplatzer occluder device. Cathet Cardiovasc Intervent 2001;53:508–512.
4. Mattila S, Laustela E, Tala P. Penetrating and perforating thoracic injuries. Scand JThorac Cardiovasc Surg 1981;15:105–110.
5. Thanopoulos BD, Tsaousis GS, Konstandopoulou GN, Zarayelyan AG. Transcatheter closure of muscular ventricular septal defects with the amplatzer ventricular septal defect occluder: Initial clinical applications in children. J Am Coll Cardiol 1999;33:1395–1399.
6. Hijazi ZM, Hakim F, Al-Fadley F, et al. Transcatheter closure of single muscular ventricular septal defects using the amplatzer muscular ventricular septal defect occluder: Initial results and technical considerations. Cathet Cardiovasc Intervent 2000;49:167–172.
7. Thanopoulos BD, Konstandopoulou GN, Tsaousis GS, et al. Catheter closure of congenital/acquired muscular VSDs and perimembranous VSDs using the Amplatzer devices. J Intervent Cardiol 2003;16:399–407.
8. Masura J, Walsh KP, Thanopoulos B, et al. Catheter closure of a moderate-to-large sized patent ductus arteriosus using the new amplatzer duct occluder: Immediate and short-term results. J Am Coll Cardiol 1998;31:878–882.
9. Tofeig M, Patel RG, Walsh KP. Transcatheter closure of a mid-muscular ventricular septal defect with an Amplatzer VSD occluder device. Heart 1999;81:438–440.
10. Hijazi Z, Hakim F, Haweleh A, et al. Catheter closure of perimembranous ventricular septal defects using the new Amplatzer membranous VSD occluder: Initial clinical experience. Cathet Cardiovasc Intervent 2002;56:508–515.
11. Bass JL, Kalra GS, Masura J, et al. Initial human experience with the Amplatzer Perimembranous ventricular septal occluder device. Cathet Cardiovasc Intervent 2003;58:238–245.
12. Cowley CG, Shaddy RE. Transcatheter treatment of a large traumatic ventricular septal defect. Catheterization and Cardiovascular Interventions 2004;61:144–146.
13. Hijazi ZM. Device closure of ventricular septal defects. Cathet Cardiovasc Intervent 2003;60:107–114.