Massive Pulmonary Embolism With Shock: Role of Thrombolysis Using Central Venous Access
ABSTRACT: Background. Massive pulmonary embolism (PE) complicated with shock has an extremely high mortality rate with medical treatment. Since access to emergency vascular surgery or endovascular specialists is not readily available in most centers, patients are frequently treated with thrombolytic agents delivered via a peripheral venous access. Patients with shock, however, have poor peripheral perfusion, and peripheral administration of thrombolytic agents may thus not reliably deliver the agent to the embolus, reducing treatment efficacy. Objective. This report discusses the role of thrombolysis administered via central venous access in PE with shock. Case report. This report describes the case of a 46-year-old man presenting with new-onset atrial fibrillation, right bundle branch block, and shock from a massive PE. In view of shock, thrombolytics were given via a subclavian central venous catheter. He improved dramatically within 1 hour, with prompt resolution of the shock and the dysrhythmia.
J INVASIVE CARDIOL 2012;24(12):E321-E324
Key words: thrombolysis, central venous access, pulmonary embolism
___________________________________________
Shock resulting from pulmonary embolism (PE) has a dismal prognosis even when the diagnosis is confirmed expeditiously.1 Accepted treatment options are thrombolysis using peripheral venous access, emergent surgical embolectomy, or percutaneous mechanical thrombectomy (with/without intrapulmonary arterial thrombolysis).2 The latter two options are more effective, but are limited by requirement of advanced surgical or endovascular facilities. Peripheral venous administration of thrombolytics, on the other hand, is available almost ubiquitously. However, in the setting of reduced cardiac output and venous return in patients with shock, its effectiveness is poor.3 In this report we describe a case of shock from massive PE that showed a striking response to thrombolysis using a subclavian central venous access. We discuss the rationale for using central thrombolysis via the internal jugular or subclavian vein in this setting and also review the pertinent literature.
Case Report. A 46-year-old, African-American man presented to the emergency department with three episodes of atypical chest discomfort lasting 15 minutes and “not feeling well” for 3 days. He reported mild dyspnea along with chest discomfort. He denied lightheadedness or syncope. Past medical history included neurosarcoidosis, hypertension, depression, obstructive sleep apnea, osteoarthritis, and bilateral avascular necrosis of the hip. He was on nocturnal continuous positive airway pressure therapy and was on valproic acid, calcium, furosemide, levetiracetam, levothyroxine, phenytoin, prednisone, vitamin D, infliximab, and travopost eye drops. He did not smoke or drink.
 At presentation, the patient was lethargic, tachycardic, and tachypneic. He had an irregular pulse at 89 beats/min (heart rate, 105 beats/min; pulse deficit, 16 beats/min), respiratory rate of 35/min, oxygen saturation of 95% on room air, and blood pressure of 81/35 mm Hg. He was afebrile and though lethargic was oriented and easily arousable. Due to moderate obesity and tachypnea, jugular venous distension could not be assessed reliably. Heart sounds were normal. Lungs were clear to auscultation. Abdomen was soft and his extremities were cool with weak pulses.
At presentation, the patient was lethargic, tachycardic, and tachypneic. He had an irregular pulse at 89 beats/min (heart rate, 105 beats/min; pulse deficit, 16 beats/min), respiratory rate of 35/min, oxygen saturation of 95% on room air, and blood pressure of 81/35 mm Hg. He was afebrile and though lethargic was oriented and easily arousable. Due to moderate obesity and tachypnea, jugular venous distension could not be assessed reliably. Heart sounds were normal. Lungs were clear to auscultation. Abdomen was soft and his extremities were cool with weak pulses.
 Laboratory examination revealed biochemical surrogates of shock in the form of lactic acidosis, acute renal insufficiency, and markedly elevated hepatic enzymes (Table 1). ABG showed pH 7.1, PCO2 of 47.6 mm Hg, and PO2 of 115.9 mm Hg. Urine drug screen was negative. Electrocardiogram (ECG) showed new-onset atrial fibrillation with new-onset right bundle branch block (RBBB) with a ventricular rate of 100 (Figure 1). Chest x-ray showed unremarkable lung fields and borderline cardiomegaly.
Laboratory examination revealed biochemical surrogates of shock in the form of lactic acidosis, acute renal insufficiency, and markedly elevated hepatic enzymes (Table 1). ABG showed pH 7.1, PCO2 of 47.6 mm Hg, and PO2 of 115.9 mm Hg. Urine drug screen was negative. Electrocardiogram (ECG) showed new-onset atrial fibrillation with new-onset right bundle branch block (RBBB) with a ventricular rate of 100 (Figure 1). Chest x-ray showed unremarkable lung fields and borderline cardiomegaly.
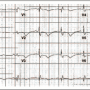 In view of shock in the setting of only minimally elevated troponin, no ST elevations and no pulmonary edema, acute myocardial infarction (MI) was considered unlikely. Based on new-onset atrial fibrillation, new-onset RBBB, and the patient’s sedentary state, pulmonary embolism was the immediate working diagnosis. Septic shock, cardiac tamponade, and iatrogenic Addison’s syndrome were also considered. Intravenous fluids, antibiotics, and intravenous steroids were administered and a stat bedside echo was ordered. Intravenous inotropic agents were very rapidly titrated to maximal doses of neosynephrine, dopamine, and phenylephrine. In spite of these measures, his systolic blood pressure remained in the 60s. A right subclavian central venous catheter and a radial arterial monitoring line were
In view of shock in the setting of only minimally elevated troponin, no ST elevations and no pulmonary edema, acute myocardial infarction (MI) was considered unlikely. Based on new-onset atrial fibrillation, new-onset RBBB, and the patient’s sedentary state, pulmonary embolism was the immediate working diagnosis. Septic shock, cardiac tamponade, and iatrogenic Addison’s syndrome were also considered. Intravenous fluids, antibiotics, and intravenous steroids were administered and a stat bedside echo was ordered. Intravenous inotropic agents were very rapidly titrated to maximal doses of neosynephrine, dopamine, and phenylephrine. In spite of these measures, his systolic blood pressure remained in the 60s. A right subclavian central venous catheter and a radial arterial monitoring line were 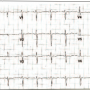 placed. He was intubated and developed cardiac arrest with pulseless electrical activity, but was resuscitated quickly with cardiopulmonary resuscitation (CPR). Stat echo showed moderate to severely dilated right ventricle (RV) with severely depressed right ventricular systolic function. There was moderate tricuspid regurgitation and pulmonary artery systolic pressure was estimated to be 60 mm Hg. The left ventricular systolic function was normal. In view of the high clinical likelihood of PE along with consistent echo findings, an immediate decision was made to administer intravenous thrombolytics. As the patient was in severe shock with poor perfusion, we decided to use central venous access rather than peripheral venous line.
placed. He was intubated and developed cardiac arrest with pulseless electrical activity, but was resuscitated quickly with cardiopulmonary resuscitation (CPR). Stat echo showed moderate to severely dilated right ventricle (RV) with severely depressed right ventricular systolic function. There was moderate tricuspid regurgitation and pulmonary artery systolic pressure was estimated to be 60 mm Hg. The left ventricular systolic function was normal. In view of the high clinical likelihood of PE along with consistent echo findings, an immediate decision was made to administer intravenous thrombolytics. As the patient was in severe shock with poor perfusion, we decided to use central venous access rather than peripheral venous line.
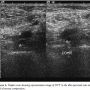 Fifty mg of alteplase was administered as a bolus through the right subclavian central venous line. In less than 60 minutes, the patient’s blood pressure improved remarkably and within 2 hours he was weaned off all vasopressors. The ECG showed normal sinus rhythm (atrial fibrillation resolved) and the RBBB became partial RBBB after 90 minutes (Figure 2). Partial RBBB resolved completely by the next day (Figure 3). The patient was extubated the following day. A Doppler ultrasound of lower extremities was performed, which showed thrombus in bilateral tibial and peroneal veins and right popliteal vein (Figure 4). He was continued on enoxaparin and then warfarin. Within the week, the patient recovered completely and was discharged with plan for long-term anticoagulation.
Fifty mg of alteplase was administered as a bolus through the right subclavian central venous line. In less than 60 minutes, the patient’s blood pressure improved remarkably and within 2 hours he was weaned off all vasopressors. The ECG showed normal sinus rhythm (atrial fibrillation resolved) and the RBBB became partial RBBB after 90 minutes (Figure 2). Partial RBBB resolved completely by the next day (Figure 3). The patient was extubated the following day. A Doppler ultrasound of lower extremities was performed, which showed thrombus in bilateral tibial and peroneal veins and right popliteal vein (Figure 4). He was continued on enoxaparin and then warfarin. Within the week, the patient recovered completely and was discharged with plan for long-term anticoagulation.
Discussion. This case demonstrates use of subclavian central venous access for administration of thrombolytic agents rather than the conventional peripheral intravenous route. We report this case to discuss this approach in patients with massive pulmonary embolism with shock where immediate surgical/endovascular intervention is not available.
PE is a life-threatening condition, causing 50,000-200,000 deaths annually.4 Five percent of acute PE patients present with hemodynamic instability and in these patients acute mortality is extremely high (up to 65%), especially in those requiring CPR.1 In patients presenting with shock, fibrinolysis is often the first line of treatment.5 This indication is based on a small, randomized controlled trial of 8 patients where all patients receiving thrombolysis survived while others died.6 Also, in a meta-analysis of 11 randomized controlled trials, a subgroup analysis of 5 trials enrolling patients with massive PE with hemodynamic instability demonstrated that thrombolysis reduced the risk of death or recurrent PE by 55%.7 Pooled data from that analysis also showed no benefit of thrombolysis in normotensive patients without evidence of RV dysfunction.7 Thus, patients with mild-to-moderate acute pulmonary embolism are treated with parenteral anticoagulation alone (unfractionated or low molecular weight heparin followed by warfarin).8 Pulmonary artery recanalization is performed medically by administration of a fibrinolytic agent or through surgical/ endovascular interventions.2 Fibrinolytic agents that have been approved by the Food and Drug Administration to treat PE are streptokinase, urokinase, and recombinant tissue plasminogen activator (r-tPA).9 Surgical options include surgical embolectomy and, more recently, minimally invasive procedures like catheter-directed thrombolysis, percutaneous embolectomy, and thrombus fragmentation.10,11 Surgical embolectomy has been shown in specialized centers to have good survival rates (of up to 89%), but that requires advanced centers and a dedicated interdisciplinary team effort, which is not available at most centers especially during an emergency.12 Catheter thrombectomy may be performed if fibrinolysis and surgical embolectomy are either contraindicated or not available. It, however, requires advanced endovascular expertise and is not without complications.11
Alteplase (rt-PA) is currently the recommended agent for thrombolysis.13 The recommended dosage is 100 mg of alteplase administered peripherally over 2 hours. A bolus regimen of alteplase where 0.6 mg/kg/15 min (maximum dose of 50 mg) is administered in 15 minutes has also been studied and compared with the above regimen.14,15 While there are no significant differences between the outcomes of these regimens, it is suggested that in cases of cardiac arrest from massive PE, bolus administration of alteplase is justified since the drug reaches the therapeutic level in the serum quickly.16
So far, only one study compared the use of peripheral administration of thrombolytic with central infusion. Verstraete et al in 1988 showed that intrapulmonary infusion of the thrombolytic agent did not offer any significant benefit over peripheral administration in PE.17 They administered r-tPA to 34 patients who were diagnosed with large acute PE. However, all patients in the study were hemodynamically stable, with a mean systolic blood pressure of 121 mm Hg and 131 mm Hg in each group, respectively, and shock was an exclusion criterion. Thus the results of that study do not apply to patients in shock such as the case reported here.
In cardiogenic shock from acute MI, thrombolysis is associated with a higher mortality and percutaneous coronary intervention is the preferred treatment of choice.18,19 The excess mortality is presumed to be secondary to poor perfusion of the tissues preventing the delivery of the thrombolytic agent infused peripherally to the site of thrombus occlusion in the coronaries.20 Similarly, in hemodynamically unstable patients with massive PE, peripheral perfusion is poor due to decreased cardiac output and peripheral thrombolysis may not be effective.3 Administration of a thrombolytic agent through the subclavian/internal jugular vein may thus facilitate enhanced delivery of the drug to the site of the emboli.
In the case presented, bolus of r-tPA in a patient with shock from PE was administered via the subclavian vein and here it led to almost immediate and dramatic recovery. Due to the anatomically advantageous location of the pulmonary artery, it can be easily accessed through the subclavian vein or the jugular vein in cases of emergency. Central venous infusion is easy, convenient, relatively inexpensive, and delivers the agent to the pulmonary artery quickly and at higher concentrations than peripheral infusion. One experimental canine model clearly describes the positive effect of thrombolytic agent delivered directly to the emboli as compared to peripheral infusion.21
Though thrombolytic therapy carries a risk of major bleeding that varies from 6%-13%,3,7,15,17,18 studies have confirmed that in cases of PE or acute MI with shock, the benefits of thrombolysis outweigh the risks.22,23 The 2003 British Thoracic Society guidelines also indicate that in cases of massive shock from PE, a bolus dose of 50 mg of alteplase should be administered as a life-saving measure.13
In a recent analysis of the ICOPER (International Co-operative Pulmonary Embolism Registry) data, fibrinolytics did not reduce the mortality or incidence of recurrent PE at 90 days in patients with massive PE.3 Similarly, another recent placebo controlled randomized study failed to show any benefit of thrombolysis when performed during resuscitation for out-of-hospital cardiac arrest.24 All the patients in these studies received only peripheral thrombolysis. The authors in the latter study opined that poor peripheral perfusion in shock may have been a contributing factor for failure of thrombolysis.
Randomized trials of central versus peripheral thrombolysis in patients with shock from PE are unlikely to be conducted. We make a case that central venous thrombolysis given as a bolus preferably through the subclavian or internal jugular vein should be considered in patients with shock from PE, especially in centers that do not have access to emergent advanced endovascular and surgical technology or expertise.
Study limitations. This case report discusses several reasons why central subclavian/internal jugular thrombolysis may be more efficacious than peripherally administered thrombolysis in patients with shock. However, this is a single case report and lack of good randomized trials makes any definite conclusions regarding efficacy differences between the two treatments difficult. There is an increased risk of bleeding with central venous access and use of thrombolytics. We would recommend that if thrombolysis is considered through a central line, then extreme caution be used including use of ultrasound guidance and use of small-gauge entry needles to minimize the risk from multiple sticks and that of inadvertent arterial puncture.
Conclusion. For patients in shock due to massive PE, administration of thrombolytic agents via a central venous route through the subclavian or the internal jugular vein may be more efficacious compared to administration using a peripheral venous route.
References
- Kasper W, Konstantinides S, Geibel A, et al. Management strategies and determinants of outcome in acute major pulmonary embolism: results of a multicenter registry. J Am Coll Cardiol. 1997;30(5):1165-1171.
- De Gregorio MA, Gimeno MJ, Mainar A, et al. Mechanical and enzymatic thrombolysis for massive pulmonary embolism. J Vasc Interv Radiol. 2002;13(2 Pt 1):163-169.
- Kucher N, Rossi E, De Rosa M, Goldhaber SZ. Massive pulmonary embolism. Circulation. 2006;113(4):577-582
- Giuntini C, Di Ricco G, Marini C, Melillo E, Palla A. Pulmonary embolism: epidemiology. Chest. 1995;107(Suppl):3S-9S.
- Büller HR, Agnelli G, Hull RD, Hyers TM, Prins MH, Raskob GE. Antithrombotic therapy for venous thromboembolic disease: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126(3 Suppl):401S-428S.
- Jerjes-Sanchez C, Ramírez-Rivera A, de Lourdes García M, et al. Streptokinase and heparin versus heparin alone in massive pulmonary embolism: a randomised controlled trial. J Thromb Thrombolysis. 1995;2(3):227-229.
- Wan S, Quinlan DJ, Agnelli G, Eikelboom JW. Thrombolysis compared with heparin for the initial treatment of pulmonary embolism: a meta-analysis of the randomized controlled trials. Circulation. 2004;110(6):744-749.
- Büller HR, Agnelli G, Hull RD, Hyers TM, Prins MH, Raskob GE. Antithrombotic therapy venous thromboembolic disease: the seventh ACCP conference on antithrombotic and thrombolytic therapy. Chest. 2004;126(3 Suppl):401S-428S
- Almoosa K. Is thrombolytic therapy effective for pulmonary embolism? Am Fam Physician. 2002;65(6):1097-1102.
- Tajima H, Murata S, Kumazaki T, et al. Recent advances in interventional radiology for acute massive pulmonary thromboembolism. J Nippon Med Sch. 2005;72(2):74-84.
- Uflacker R. Intervention therapy for pulmonary embolism. J Vasc Interv Radiol. 2001;12(2):147-164.
- Aklog L, Williams CS, Byrne JG, Goldhaber SZ. Acute pulmonary embolectomy: a contemporary approach. Circulation. 2002;105(12):1416-1419.
- British Thoracic Society Standards of Care Committee Pulmonary Embolism Guideline Development Group. British Thoracic Society guidelines for the management of suspected acute pulmonary embolism. Thorax. 2003;58(6):470-484.
- Sors H, Pacouret G, Azarian R, Meyer G, Charbonnier B, Simonneau G. Hemodynamic effects of bolus vs. 2-h infusion of alteplase in acute massive pulmonary embolism: a randomized controlled multicenter trial. Chest. 1994;106(3):712-717.
- Goldhaber SZ, Agnelli G, Levine MN; the Bolus Alteplase Pulmonary Embolism Group. Reduced dose bolus alteplase vs. conventional alteplase infusion for pulmonary embolism thrombolysis. An international multicenter randomized trial. Chest. 1994;106(3):718-724.
- Capstick T, Henry MT. Efficacy of thrombolytic agents in the treatment of pulmonary embolism. Eur Respir J. 2005;26(5):864-874.
- Verstraete M, Miller GA, Bounameaux H, et al. Intravenous and intrapulmonary recombinant tissue-type plasminogen activator in the treatment of acute massive pulmonary embolism. Circulation. 1988;77(2):353-360.
- Chou TM, Amidon TM, Ports TA, Wolfe CL. Cardiogenic shock: thrombolysis or angioplasty. J Intensive Care Med. 1996;11(1):37-48.
- Bates ER, Topol EJ. Limitations of thrombolytic therapy for acute myocardial infarction complicated by congestive heart failure and cardiogenic shock. J Am Coll Cardiol. 1991;18(4):1077-1084.
- Levine GN, Hochman JS. Thrombolysis in acute myocardial infarction complicated by cardiogenic shock. J Thromb Thrombolysis. 1995;2(1):11-20.
- Tapson VF, Gurbel PA, Witty LA, Pieper KS, Stack RS. Pharmacomechanical thrombolysis of experimental pulmonary emboli. Rapid low-dose intraembolic therapy. Chest. 1994;106(5):1558-1562.
- Spohr F, Böttiger BW. Thrombolytics in CPR, current advantages in cardiopulmonary resuscitation. Minerva Anestesiol. 2005;71(6):291-296.
- Snyder-Ramos SA, Motsch J, Martin E, Bottiger BW. Thrombolytic therapy during cardiopulmonary resuscitation. Minerva Anestesiol. 2002;68(4):186-191.
- Böttiger BW, Arntz HR, Chamberlain DA, et al; TROICA trial investigators; European Resuscitation Council Study Group. Thrombolysis during resuscitation for out-of-hospital cardiac arrest. N Engl J Med. 2008;359(25):2651-2662.
__________________________________________
From the Division of Cardiovascular Diseases, Cardiovascular Research Institute, University of Kansas Medical Center and Hospital, Kansas City, Kansas.
The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. The authors report no conflicts of interest regarding the content herein.
Manuscript submitted January 26, 2012, provisional acceptance given February 28, 2012, final version accepted July 6, 2012.
Address for correspondence: Jayasree Pillarisetti, MD, 3901 Rainbow Blvd, Kansas City, KS 66209. Email: jayasreep24@gmail.com
















