Elective Mechanical Circulatory Support in the Percutaneous Treatment of Patients With Combined Complex Coronary Artery Disease and Severe Aortic Valve Stenosis
Abstract: Objectives. One of the procedural concerns during percutaneous treatment of patients with concomitant coronary artery disease (CAD) and aortic valve stenosis (AS) is the risk of hemodynamic instability. In the present study, we assessed the safety and effectiveness of elective hemodynamic support with the Impella 2.5 system (Abiomed, Inc) in patients undergoing combined high-risk percutaneous coronary intervention (PCI) and balloon aortic valvuloplasty (BAV), as a bridge to transcatheter aortic valve replacement. Methods. Ten patients suffering from acute coronary syndrome (ACS) and severe AS were included. In all patients, we first performed PCI with the hemodynamic support of the Impella 2.5 system, and then BAV. Results. The Impella catheter was successfully positioned in the left ventricle in all instances. All cases were successful, without any intraprocedural complications. All patients were discharged without any major clinical events. Conclusions. These preliminary results suggest that elective use of the Impella 2.5 system is safe and effective when performing PCI and BAV in high-risk ACS patients suffering from concomitant severe CAD and AS.
Key words: aortic valve stenosis, high-risk PCI, transcatheter aortic valve replacement
The concomitant presence of significant coronary artery disease (CAD) and severe aortic valve stenosis (AS) has been more frequently recognized.1,2 Treatment of these patients is challenging. Indeed, in the subset at high surgical risk, the percutaneous approach is indicated. In patients with stable CAD, percutaneous aortic valve treatment may be carried out with or without percutaneous coronary intervention (PCI),3 whereas patients suffering from acute coronary syndrome (ACS) and concomitant AS require the combined percutaneous treatment.1 One of the procedural concerns during percutaneous treatment is the risk of hemodynamic instability, which can occur during both the coronary artery procedure and the aortic valve treatment.4,5
Elective use of percutaneous left ventricular assist devices may help to improve the safety and tolerability of such complex procedures. In the last few years, studies have shown that the Impella system (Abiomed, Inc) gives superior hemodynamic support in comparison with intra-aortic balloon pump (IABP).6 Although concerns exist on the potential detrimental effect due to the reduced effective valve orifice area, there are some reports suggesting that the Impella system may be safe and useful in patients with severe AS.7,8 Herein, we present our experience with elective hemodynamic support using the Impella system in high-risk ACS patients undergoing combined PCI and balloon aortic valvuloplasty (BAV) as a bridge to transcatheter aortic valve replacement (TAVR).
Methods
Patient population. This is a prospective, non-randomized study assessing the safety and efficacy of elective hemodynamic support by the Impella system in high-risk ACS patients undergoing PCI and BAV. Severe AS was defined as an aortic-valve area of ≤0.8 cm2 or an aortic valve index of ≤0.5 cm2/m2 and either mean aortic valve gradient ≥40 mm Hg or peak aortic jet velocity ≥4.0 m/s. From January 2016 to July 2018, all consecutive ACS patients with severe CAD and symptomatic AS who were deemed inoperable or at high risk were considered eligible for the present study, according to the heart team consensus.9 High surgical risk was defined as a Society of Thoracic Surgeons (STS) score ≥10 and/or European System for Cardiac Operative Risk Evaluation (EuroScore II) of ≥6. All patients included in the present study also had at least one of the following characteristics: (1) hemodynamic instability; and (2) complex CAD.
Hemodynamic monitoring. Elective hemodynamic support was carried out with the Impella 2.5 device. After preclosing with 2 Proglide sutures (Abbott Vascular), the right or left common femoral artery was cannulated with a 14 Fr sheath (Cook Medical). Transfemoral access was obtained percutaneously, under fluoroscopic guidance. Therefore, a 6 Fr Amplatz left 1 (AL-1) diagnostic catheter, loaded with a 0.035˝, straight, soft-end wire was advanced across the aortic valve. Once the valve was crossed, two 0.018˝ wires were advanced through the AL-1 diagnostic catheter into the apex of the left ventricle. One 0.018˝ guidewire was used to position the Impella 2.5 into the left ventricle; the second wire was left in the left ventricle in order to avoid crossing the stenotic aortic valve a second time to perform BAV.
Percutaneous coronary intervention. Radial access was used for PCIs except in cases of access failure. All patients received intravenous unfractionated heparin (70 IU/kg) before starting the procedure in order to achieve an activated clotting time >250 seconds; additional heparin was administered if deemed necessary. All patients were on dual-antiplatelet therapy. In patients who had not taken thienopyridine for at least 5 days before the procedure, a loading dose (clopidogrel 600 mg, ticagrelor 180 mg, or prasugrel 30 mg) was administered at least 2 hours before PCI. The technique and strategy for PCI were left to the operator’s preference. Periprocedural myocardial infarction was defined according to the third universal definition of myocardial infarction.10 Major bleeding was defined according to the Bleeding Academic Research Consortium criteria.11
BAV procedure. All procedures were carried out after PCI. After a successful PCI, the Impella system was retrieved and the second 0.018˝ guidewire previously left in the left ventricle was exchanged with a 0.035˝ extra-stiff guidewire (Boston Scientific). The default balloon diameters were 18 mm and 20 mm (VACS-II; Osypka), and were selected according to aortic annulus diameter size by transesophageal echocardiography. Balloon inflation was carried out during rapid ventricular pacing (180-200 beats/min), and was started as soon as pulse abrogation was observed. Pacing was continued until the balloon was fully deflated. The strategy was to perform an inflation and then to assess hemodynamic result after each inflation. Additional inflations (with the same balloon or with a larger balloon) were carried out in case of unsatisfactory hemodynamic effect (<50% reduction in the mean aortic valve gradient). The procedure was terminated if one of the following criteria was met: at least 50% reduction in the mean aortic valve gradient; three dilations with complete pulse abrogation independently from hemodynamic results; balloon rupture; or severe aortic valve regurgitation. Access-site closure was obtained with a percutaneous approach using Perclose Proglide, or surgery if necessary.
Statistical analysis. Continuous variables are given as mean ± 1 standard deviation or median and interquartile range (IQR), when appropriate. The Student’s t-test and non-parametric Mann-Whitney test were used to determine differences between mean values for normally and non-normally distributed variables, respectively. Categorical variables are reported as percentages and were analyzed by the Fisher’s exact test, as appropriate.
Results
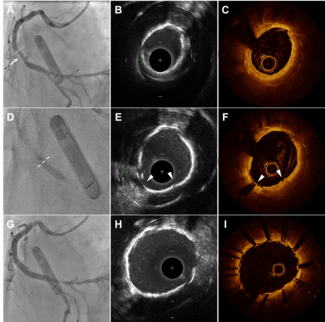
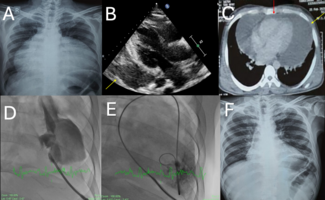
Patient characteristics. Ten patients were included in the present study. The principal clinical, angiographic, and procedural characteristics are reported in Table 1. PCI was carried out through the radial approach in 8 patients. PCI was performed first (before BAV) in all patients. In all instances, hemodynamic support was elective before starting PCI. Rotational atherectomy was performed in 7 patients. Details of the PCI procedures are reported in Table 1. PCI was successful in all patients. Hypotension during PCI occurred in 6 patients (60%); slow-flow or no-reflow occurred in 5 of these 6 patients. Hypotension was managed by vasopressors. Following PCI, and after withdrawing the Impella catheter, BAV was performed. This procedure was successful in all cases (Table 1). Emergent TAVR was not required in any case. Two cases are represented in Figures 1 and 2.
Clinical follow-up. Periprocedural myocardial infarction occurred in 2 patients (5%). No major bleeding or in-hospital death occurred. Patients were discharged home 5 ± 3 days after the procedure.
Discussion
A large proportion of patients (40%-75%) with severe AS requiring AVR also have concomitant severe CAD.1,2 Treatment of high-risk patients with concomitant ACS and severe AS is challenging. Indeed, surgical AVR (SAVR) and concomitant coronary artery bypass grafting have been the standard management strategy for these patients,10 and pending the results of randomized studies addressing this issue, there is consensus that patients suffering from ACS and concomitant severe AS who are at high surgical risk require the combined percutaneous treatment.1,12 Procedural issues during percutaneous treatment of these patients are: (1) the risk of hemodynamic instability; and (2) the role of BAV.
Hemodynamic instability. Hemodynamic instability may occur both during the coronary artery procedure and during the aortic valve treatment as a consequence of the significant unrevascularized CAD, especially during rapid ventricular pacing and balloon inflation.4,5 For example, no-reflow in the presence of critical AS can start a vicious cycle, leading to rapid hemodynamic collapse. In the cases described herein, we observed that the Impella system provided excellent hemodynamic support in the absence of clinically significant periprocedural complications. This left ventricular support device has proven greater safety and efficacy in high-risk PCI patients compared with the IABP.6 Indeed, the Impella system improves coronary flow, unloads the left ventricle, and protects myocardium from ischemic damage during PCI.6
Our experience is among the first in the literature to report on the Impella system for hemodynamic support in high-risk patients undergoing combined PCI and BAV.13,14 Due to the lack of data on this topic, it is unknown whether it is better to perform BAV before or after PCI. We decided to perform PCI with the Impella support first, and then BAV. This option has the following advantages: (1) it avoids the period of hypotension during BAV that may worsen coronary flow in patients with concomitant severe and clinically unstable CAD; and (2) it prevents increase of left ventricular wall stress and myocardial oxygen demand (occurring during left ventricular systole against the occlusive balloon), which may be fatal in these high-risk patients.14 Recent data coming from the Global cVAD registry confirmed that the elective use of the Impella system to support BAV was associated with significantly better 1-year survival when compared with emergency placement of the device.8 Other studies have also shown the Impella system to support BAV.15,16 Disadvantages include: (1) the need for a 14 Fr sheath to allow the advancement of the Impella 2.5 device plus an additional 0.018˝ guidewire to be left in the left ventricle in order to avoid recrossing the stenotic aortic valve to perform BAV; and (2) troubles in advancing the Impella catheter through the stenotic aortic valve. Indeed, patients with AS have been excluded from studies involving the Impella catheter due to theoretical concern that its size could have reduced effective valve orifice area and/or induced significant valve regurgitation. Actually, the device that crosses the aortic valve measures 12 Fr (0.4 cm); an aortic area of 0.58 cm2 corresponds to 0.86 cm. Therefore, the Impella system is quite safe in aortic valves with an area >0.4 cm2.16 We were able to position the Impella catheter in the left ventricle in all cases. However, Spiro et al reported that in some patients, the balloon-assist technique or partial BAV were required to facilitate positioning of the Impella catheter through the stenotic aortic valve.7
The strategy of performing BAV first has the following advantages: (1) it decreases the left ventricular afterload and thereby allows a global decrease in systolic wall stress and subendocardial ischemia before attempting dilation of the coronary stenosis;14,17 and (2) it facilitates positioning of the Impella catheter in the left ventricle through the stenotic aortic valve. Disadvantages include the risk of urgent/emergent TAVR in cases of severe aortic insufficiency following BAV.18 Although PCI may be performed after TAVR, we acknowledge that this approach is of some concern due to access issues, especially when dealing with complex coronary lesions.19-21 The valve struts could interfere with cannulation of coronaries, and catheter manipulation could potentially even dislodge the valve, although this is unlikely.
Balloon aortic valvuloplasty. TAVR is recommended for patients with prohibitive or high surgical risk or with an absolute contraindication to SAVR (class I, level of evidence A).9 Moreover, percutaneous BAV may be considered as a bridge to SAVR or TAVR for symptomatic patients with severe AS (class IIb, level of evidence C).9 We believe that in the clinical scenario represented by patients with ACS and severe AS, the role for BAV as a bridge to TAVR should be reconsidered.17,22 Indeed, although concomitant TAVR and PCI has been reported,23-25 we should take into account the following issues. First, it has been shown that patients who have CAD are 10 times more likely to die within 30 days of TAVR compared to those with no CAD.1,2,26 Patients with severe AS and left ventricular ejection fraction ≤30% and those with STS-PROM scores ≥10% were found to be at the highest risk of 30-day mortality after PCI.1,2 Second, the Multicenter Canadian TAVR study4 showed that need for hemodynamic support during TAVR (with intra-aortic balloon counter-pulsation or extracorporeal circulation) was an independent predictor of early as well as late mortality after TAVR. Third, the risk for acute kidney injury may increase with concomitant TAVR procedure, especially in patients with baseline renal dysfunction.
Study limitations. The small sample size and the single-center design represent the major limitations of the present study.
Conclusion
The present experience suggests that elective use of the Impella 2.5 system is safe and effective when performing PCI and BAV in high-risk patients suffering from ACS and severe AS.
References
- Goel SS, Agarwal S, Tuzcu EM, et al. Percutaneous coronary intervention in patients with severe aortic stenosis: implications for transcatheter aortic valve replacement. Circulation. 2012;125:1005-1013.
- Paradis JM, Fried J, Nazif T, et al. Aortic stenosis and coronary artery disease: what do we know? What don’t we know? A comprehensive review of the literature with proposed treatment algorithms. Eur Heart J. 2014;35:2069-2082.
- Wenaweser P, Pilgrim T, Guerios E, et al. Impact of coronary artery disease and percutaneous coronary intervention on outcomes in patients with severe aortic stenosis undergoing transcatheter aortic valve implantation. EuroIntervention. 2011;7:541-548.
- Rodes-Cabau J, Webb JG, Cheung A, et al. Transcatheter aortic valve implantation for the treatment of severe symptomatic aortic stenosis in patients at very high or prohibitive surgical risk: acute and late outcomes of the multicenter Canadian experience. J Am Coll Cardiol. 2010;55:1080-1090.
- Singh Rao R, Shapiro RL, Lasala JM. No reflow leading to catastrophic hemodynamic collapse in a patient with severe aortic stenosis and its management. Catheter Cardiovasc Interv. 2016;87:983-988.
- O’Neill WW, Kleiman NS, Moses J, et al. A prospective, randomized clinical trial of hemodynamic support with Impella 2.5 versus intra-aortic balloon pump in patients undergoing high-risk percutaneous coronary intervention: the PROTECT II study. Circulation. 2012;126:1717-1727.
- Spiro J, Venugopal V, Raja Y, Ludman PF, Townend JN, Doshi SN. Feasibility and efficacy of the 2.5 L and 3.8 L Impella percutaneous left ventricular support device during high-risk, percutaneous coronary intervention in patients with severe aortic stenosis. Catheter Cardiovasc Interv. 2015;85:981-989.
- Singh V, Yadav PK, Eng MH, et al. Outcomes of hemodynamic support with Impella in very high-risk patients undergoing balloon aortic valvuloplasty: results from the Global cVAD registry. Int J Cardiol. 2017;240:120-125.
- Nishimura RA, Otto CM, Bonow RO, et al. 2017 AHA/ACC focused update of the 2014 AHA/ACC guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2017;135:e1159-e1195.
- Thygesen K, Alpert JS, Jaffe AS, et al. Third universal definition of myocardial infarction. Circulation. 2012;126:2020-2035. Epub 2012 Aug 24.
- Mehran R, Rao SV, Bhatt DL, et al. Standardized bleeding definitions for cardiovascular clinical trials: a consensus report from the Bleeding Academic Research Consortium. Circulation. 2011;123:2736-2747.
- Khawaja MZ, Wang D, Pocock S, Redwood SR, Thomas MR. The percutaneous coronary intervention prior to transcatheter aortic valve implantation (ACTIVATION) trial: study protocol for a randomized controlled trial. Trials. 2014;15:300.
- Badawi RA, Grise MA, Thornton SN. Impella 2.5 assisted balloon aortic valvuloplasty and percutaneous coronary intervention as a bridge to heart transplantation. J Invasive Cardiol. 2012;24:229-230.
- Martinez CA, Singh V, Londono JC, et al. Percutaneous retrograde left ventricular assist support for interventions in patients with aortic stenosis and left ventricular dysfunction. Catheter Cardiovasc Interv. 2012;80:1201-1209.
- Londono JC, Martinez CA, Singh V, O’Neill WW. Hemodynamic support with Impella 2.5 during balloon aortic valvuloplasty in a high-risk patient. J Interv Cardiol. 2011;24:193-197.
- Ludeman DJ, Schwartz BG, Burstein S. Impella-assisted balloon aortic valvuloplasty. J Invasive Cardiol. 2012;24:E19-E20.
- McKay RG, Safian RD, Berman AD, et al. Combined percutaneous aortic valvuloplasty and transluminal coronary angioplasty in adult patients with calcific aortic stenosis and coronary artery disease. Circulation. 1987;76:1298-1306.
- Alkhouli M, Zack CJ, Sarraf M, et al. Morbidity and mortality associated with balloon aortic valvuloplasty: a national perspective. Circ Cardiovasc Interv. 2017;10:e005730.
- Geist V, Sherif MA, Khattab AA. Successful percutaneous coronary intervention after implantation of a CoreValve percutaneous aortic valve. Catheter Cardiovasc Interv. 2009;73:61-67.
- Zajarias A, Eltchaninoff H, Cribier A. Successful coronary intervention after percutaneous aortic valve replacement. Catheter Cardiovasc Interv. 2007;69:522-524.
- Chakravarty T, Sharma R, Abramowitz Y, et al. Outcomes in patients with transcatheter aortic valve replacement and left main stenting: the TAVR-LM registry. J Am Coll Cardiol. 2016;67:951-960.
- Pedersen WR, Klaassen PJ, Pedersen CW, et al. Comparison of outcomes in high-risk patients >70 years of age with aortic valvuloplasty and percutaneous coronary intervention versus aortic valvuloplasty alone. Am J Cardiol. 2008;101:1309-1314.
- Salhab KF, Al Kindi AH, Lane JH, et al. Concomitant percutaneous coronary intervention and transcatheter aortic valve replacement: safe and feasible replacement alternative approaches in high-risk patients with severe aortic stenosis and coronary artery disease. J Cardiac Surg. 2013;28:481-483.
- Conradi L, Seiffert M, Franzen O, et al. First experience with transcatheter aortic valve implantation and concomitant percutaneous coronary intervention. Clin Res Cardiol. 2011;100:311-316.
- Ahn JM, Lee KH, Yoo SY, et al. Prognosis of variant angina manifesting as aborted sudden cardiac death. J Am Coll Cardiol. 2016;68:137-145.
- Dewey TM, Brown DL, Herbert MA, et al. Effect of concomitant coronary artery disease on procedural and late outcomes of transcatheter aortic valve implantation. Ann Thorac Surg. 2010;89:758-767.
From the 1Interventional Cardiology Unit; 2Vascular Surgery Unit; and 3Cardiac Surgery Unit, Clinica Mediterranea, Naples, Italy.
Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. The authors report no conflicts of interest regarding the content herein.
The authors report that patient consent was provided for publication of the images used herein.
Manuscript submitted November 11, 2018, accepted November 20, 2018.
Address for correspondence: Carlo Briguori MD, PhD, FACC, FSCAI, Interventional Cardiology, Clinica Mediterranea, Via Orazio, 2, I-80121, Naples, Italy. Email: carlobriguori@clinicamediterranea.it