Carotid Artery In-Stent Restenosis in a Patient with Contralateral Total Occlusion, Resolved with Drug-Eluting Stenting
Case Presentation. A 70-year-old male was admitted to our clinic with a diagnosis of severe hypertension, right carotid artery stenosis, recent right sylvian artery ischemic stroke, insulin-dependent diabetes mellitus and bilateral peripheral artery occlusive disease stage II Fontaine. Doppler ultrasound of the carotid arteries showed: (1) 90% stenosis at the origin of the right internal carotid artery due to a hyperechogenic plaque; (2) suspicion of left internal carotid artery total occlusion; (3) significant stenosis of both subclavian arteries; and (4) total occlusion of the right vertebral artery. Neurological examination revealed mild left hemiparesis and a computed tomography (CT) scan showed a scarred right sylvian stroke. Due to these findings, cerebral angiography was performed, which confirmed total occlusion of the left internal carotid artery and severe stenosis of the right internal carotid artery and both subclavian arteries. The patient was considered to be at high risk for carotid endarterectomy and consequently, after premedication with aspirin and clopidogrel, we performed stenting of the right carotid artery (via the right common femoral artery). We used a 10 x 24 mm self-expandable carotid Wallstent (Boston Scientific Ireland Ltd., Galway, Ireland), and an EPI Filterwire (Boston Scientific Corp., Natick, Massachusetts) as distal protection. After stent placement, we performed postdilatation with a 5 mm diameter Savvy balloon (Cordis Corp., Miami, Florida), with good angiographic results and a residual stenosis of 15%. The patient was discharged 4 days after the intervention, and had an event-free recovery for 14 days, when he was admitted with grand mal seizures in the emergency room of the county hospital. The neurological examination, Doppler study and the computed tomography (CT) scan did not reveal any new ischemic causes for the seizures, which subsided under anti-epileptic therapy (no magnetic resonance imaging [MRI] examination was available at that hospital).
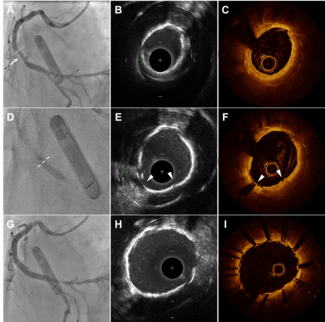
Doppler ultrasound of the carotid arteries at 6-month follow up showed a severe in-stent lesion with systolic and diastolic velocities corresponding to a stenosis of more than 70%, with the patient being asymptomatic at this time. Angiography confirmed in-stent carotid artery restenosis. Under EPI filter protection, we attempted dilatation with a 5 mm diameter Savvy balloon, but the procedure was relatively difficult due to repeated slipping of the balloon, occlusion of the filter and syncope. The postprocedure angiogram showed only a modest increase in luminal diameter, with 40% residual stenosis. The patient was asymptomatic at the 3-month control examination, despite the significant stenosis observed on Doppler ultrasound. Five months later, the patient experienced a possible minor transient ischemic attack corresponding to the right carotid artery (per the county medical report). The CT scan and MRI performed at our clinic found no other vascular event, but Doppler ultrasound revealed in-stent restenosis (ISR) (Figure 1), which was confirmed by angiography (Figure 2).
Due to these findings, under EPI filter protection, a Yukon 3.5 mm diameter drug-eluting stent (DES) (Translumina GmbH, Germany) with a 23 mm stent was implanted and expanded at minimal pressure, when the balloon exploded with acute thrombosis of the right internal carotid artery (Figure 3). During the procedure, soon after balloon explosion, the patient experienced sudden loss of consciousness and apnea and had to be intubated with mechanical ventilation. Concomitantly, 15 mg of tenecteplase was administered intracarotidian, followed by expansion of the stent with an ACS Rx Solaris 4 x 20 mm balloon (Guidant Corp., Indianapolis, Indiana) at a pressure of 18 atm, achieving spectacular re-establishment of carotid flow (Figure 4). The patient was completely recovered 12 hours later and was discharged after 3 days without any neurological deficit. He was seen 1 year later for follow up, when Doppler ultrasound of the carotid arteries revealed no restenosis and good stent expansion (Figure 5), confirmed by angiography (Figure 6).
Six months following the previously-mentioned study, the patient was admitted for intermittent claudication, fatigability and severe hypertension. A Doppler study of the carotid stent showed normal velocities, and his neurological exam was similar to the previous one. Because of the patient’s severe hypertension and mild renal failure, we performed a Doppler study of the renal arteries, which showed bilateral severe stenosis. Angiography confirmed bilateral ostial stenosis of the renal arteries, thus at that time, we performed bilateral renal stenting using two 5 x 17 mm renal Bridge Stents (Medtronic AVE, Düsseldorf, Germany) deployed at 12 atm. After the procedure, the patient experienced improvement in his clinical status, decreased fatigability and no changes in his creatine level (his creatine level remain as high as 1.7 mg %).
After the procedure, the patient received aspirin 250 mg and clopidogrel 75 mg indefinitely. We recommended this long-term drug regimen because of the DES thrombosis in the single arterial vessel of the brain. We also prescribed long-term medical therapy due to the potential for catastrophic late thrombosis similar to the left main coronary stenting and also due to the lack of knowledge about DES in the carotid arteries. Furthermore, we had to continue with this therapy because of the bilateral renal stenting performed in this patient 6 months after the previously-mentioned 1-year follow up.
HOW WOULD YOU TREAT THIS PATIENT?
Craig Walker, MD
Cardiovascular Institute of the South Vascular Laboratory
Houma, Louisiana
E-mail: drcrwalker@aol.com
The case reported here involves a patient who underwent right carotid artery stenting (CAS) utilizing a Wallstent with a post-dilation balloon size of 5 mm. The patient had contralateral carotid artery occlusion right subclavian and right vertebral occlusion, as well as a high-grade left subclavian artery stenosis. His index procedure was complicated by a grand mal seizure. The patient then developed symptomatic ISR that was treated by simple balloon dilatation associated with balloon slippage, some dissection and significant embolic debris that was captured by the distal protection filter. There appeared to be a suboptimal angioplasty result. The patient developed repeat ISR which was treated with a 3.5 mm DES. This was complicated by balloon rupture and vessel thrombosis that was successfully treated with thrombolytics and subsequent dilatation with a 4 mm balloon. Subsequent angiography disclosed no evidence of subsequent restenosis.
Fortunately, symptomatic carotid ISR is relatively uncommon. Although balloon-expandable stents have been used to treat CAS, the fear of stent crushing has led most physicians to utilize self-expanding stents. Two classes of self-expanding stents have been utilized: (1) nitinol tubular stents, and (2) stainless steel woven stents. In this case, a stainless steel woven stent was utilized. This stent must foreshorten longitudinally to grow axially. It has been my experience that balloon slippage and suboptimal angioplasty results are more common with this type of self-expanding stent restenosis. Because of this, I typically utilize cutting balloons or long balloons to achieve better angioplasty results. At the time of the second restenosis, a 3.5 mm DES was placed. Although the patient ultimately had a good angiographic result with no recurrent restenosis, the patient did require immediate resuscitation after balloon rupture. Obviously, the vessel was initially deemed to be at least 5 mm, which represented significant undersizing. There has been no trial yet demonstrating the safety or efficacy of DES in the carotid circulation, and one must still be concerned about stent crushing, as this was a balloon-expandable stent. As there were no localizing signs or symptoms in the second bout of ISR in this patient, I think several alternative strategies could have been considered. These strategies could include stenting of the left subclavian artery to improve vertebral blood flow, placement of a second self-expanding stent within the prior stent, or placement of an adequately sized (5 mm) non-DES balloon-expandable stent (as the largest DES are 3.5 mm).
Although this patient had an excellent final outcome, he had a stormy course getting there. He will need to be watched for possible stent crushing. Great caution must be utilized in the treatment of patients with symptomatic carotid stenosis.
Aravinda Nanjundappa, MD
East Carolina University Brody School of Medicine
Greenville, North Carolina
E-mail: dappamd@yahoo.com
This is an illustrative case of carotid artery ISR. The patient has an extensive history of vascular disease especially involving the ostium of the great vessels of the aortic arch. This should trigger a suspicion for a systemic cause of vascular disease such as Takayasu’s arteritis,1 giant-cell arteritis and other vasculitis pathologies. The age and sex of the patient are not suggestive of vasculitis, however, no atherosclerotic risk factors were mentioned. Figure 2 in the case demonstrates a self-expanding stent covering only the internal carotid artery and extends distally beyond the lesion site. The majority of carotid lesions occur at the bifurcation due to the vortex of flow that causes turbulence and nidus for atherosclerosis. Long segments of the vascular endothelium covered by stents potentiate ISR.
The Doppler study performed at 6 months showed ISR of up to 70%. The criteria used to determine the 70% ISR was not mentioned in this case. We consider ISR to be significant if the peak systolic velocity (PSV) is > 450 cm/sec and the ratio of the internal carotid artery to the common carotid artery PSV is > 4.02. If the patient is asymptomatic, medical treatment and serial ultrasound surveillance at closer intervals of 3 months should suffice. Preventive measures to reduce ISR include the use of cilostazol3 intense lipid therapy and valsartan.4 If the carotid artery stent demonstrates progressive elevation of velocity by duplex examination, confirmation of ISR should be performed with carotid angiography. If ISR is confirmed to be > 80%, we perform angioplasty with a long balloon to prevent the “watermelon seeding” effect. The occasional use of a Cutting Balloon can also be helpful. ISR in the peripheral vasculature differs from coronary ISR in several ways. Smoking5 plays a pivotal role in peripheral vessel ISR, thus cessation of tobacco can be helpful. The authors of the case should have used intravascular ultrasound (IVUS) to determine the cause of ISR such as an uncovered dissection flap, an underdeployed stent and malapposition of the stent struts. The nature of the restenotic thrombus versus neointimal hyperplasia might have been identified on the IVUS.
Coronary ISR presents as acute coronary syndrome in up to 50% of cases.6 This patient had a TIA and a Doppler study showed ISR, indicating that ISR is not a benign process. The material found in ISR of the peripheral vasculature has shown to be thrombus in at least 20% of cases, as demonstrated by Dr. Isner and colleagues.7 Sandwich treatment of ISR with a stent inside a stent will only yield more metal layers. The use of DES to treat ISR has limited experience in the coronary arteries. Despite the acute high success rate of the DES stent sandwich technique, late stent thrombosis is a real phenomenon that can have devastating sequelae.8 In this case, explosion of the balloon during postdilatation can result in leakage of trapped air in the balloon and air embolism. Air embolism to the cerebral circulation could explain the patient’s sudden loss of consciousness and apnea. The patient’s activated clotting time (ACT) during carotid artery stenting is not mentioned. An ACT of at least 350 seconds is crucial for carotid stenting. Dual-platelet therapy with aspirin and clopidogrel is mentioned only during the first stent placement in this patient. It would be of great interest to know if the patient was continued on dual-platelet therapy, as an important cause for late stent thrombosis in the coronary arteries is discontinuation of clopidogrel.
The use of a DES balloon-expandable stent in the carotid artery for ISR is difficult to justify, particularly in a solitary carotid artery with contralateral occlusion. Once ISR of the carotid artery was seen, an attempt should have been made to consult a vascular surgeon. Carotid endarterectomy (CECA) with stent extraction utilizing a carotid shunt would have been ideal. Bare-metal stents have a significant chance of stent compression due to twisting and kinking forces that act on the stent. DES have created enough controversy due to late stent thrombosis in the coronary arteries. Such a stent thrombosis in a solitary carotid artery can lead to a fatal stroke.
To summarize, CAS is a unique procedure that requires a thoughtful process. CAS should be restricted to nonoctogenerians who are at high risk for CECA. All patients must be preloaded with aspirin and clopidogrel prior to stenting, and during the procedure, weight-based unfractionated heparin should be given to maintain the ACT at > 350 seconds in order to minimize complications. Judicious use of self-expanding stents to cover the entire plaque is helpful. If asymptomatic ISR occurs, medical treatment with surveillance duplex ultrasound is a reasonable strategy. Symptomatic or severe stenosis should be treated with balloon angioplasty only. Surgical consultation is beneficial both for collegial working relations and superior patient care.
*See References below
Robert S. Dieter, MD, RVT
Loyola University
Interventional Cardiology Department Vascular and
Endovascular Medicine
Maywood, Illinois
E-mail: roberterin@aol.com
This is a very interesting case of a 70-year-old male with severe extracranial carotid artery stenosis. Due to high-risk features, including contralateral occlusion, he underwent endovascular treatment of a high-grade, symptomatic RICA stenosis using the Wallstent. Based upon one of the angiograms, it appears that the Wallstent was extended fairly distal to the bifurcation — which for most lesions is unnecessary. It is unclear why, but the patient remained in the hospital an additional 4 days after the carotid stenting procedure. It is noted that he developed grand mal seizures 2 weeks after the intervention. Although noninvasive testing was performed on the stent, the presence of fresh thrombus (hypoechoic) cannot be reliably excluded by duplex examination. Invasive angiography would be justified in such a circumstance — particularly given the patient’s lack of cerebral-vascular reserve (contralateral occlusion and vertebral artery stenosis/bilateral subclavian stenosis). Although not specifically mentioned (nor prospectively studied in a randomized fashion), the patient should have been on dual-antiplatelet therapy.
The patient then returned 6 months later with moderate asymptomatic restenosis (70%) by angiography. Some would advocate continued surveillance rather than intervening on an asymptomatic patient with moderate restenosis. In this case, attempts were made at balloon angioplasty, but due to balloon slippage, the procedure was abandoned, leaving both a reinjured endothelium and a 40% residual stenosis. Cutting balloons have been used to avoid balloon slippage for ISR in the carotid artery. Reportedly during the procedure, the patient had occlusion of the filter — it would be interesting to know what the debris collected was since this lesion was likely from intimal hyperplasia and smooth muscle cells. Some would advocate relining the lesion with a second stent (self-expanding) and repeat angioplasty if angioplasty alone is not successful.
At slightly less than 1-year follow up from the original procedure, the patient returned with a TIA — though no imaging evidence of overt cerebral infarction. Angiography demonstrated a smooth, tapering 60–70% stenosis without ulceration or obvious thrombus. It would be interesting to know if any other systemic evaluation for his TIA was performed, particularly in such a patient with diffuse atherosclerotic vascular disease. Recurrent events from ISR in the carotid is uncommon.
Presumably, because this was ISR, the lesion was treated with a DES. Surprisingly, the stent balloon ruptured, leading to thrombosis of the carotid artery. This was treated with local thrombolysis and expansion of the stent with a 4.0 mm diameter balloon. Remarkably, the patient did not suffer any permanent neurological sequelae. However, several concerns should be raised. First, the use of a balloon-expandable stent in the carotid artery has been all but abandoned due to the potential for stent compression. This has been attributed to poor hoop strength, extrinsic compression, movements of the neck exerting stretching, twisting and kinking forces on the carotid artery. Furthermore, the internal carotid artery is generally 4–7 mm diameter. IVUS studies have shown that final stent diameter in the internal carotid artery is a known predictor of restenosis after carotid stenting; thus, postdilatation to 4 mm is likely on the smaller size for a carotid that has demonstrated a propensity to have significant intimal hyperplasia. The consequences of a DES in the carotid circulation are unknown, as is the risk of stent thrombosis. Elution kinetics for DES have been developed for coronary application, thus their vascular biology in the extracranial carotid segment is entirely unknown. DES in the lower extremities have not shared the same results as in the coronary arteries.
In summary, this is a very interesting case of a patient with complex anatomy that the authors were able to successfully treat. Their ability to recover from acute thrombosis in the internal carotid artery highlights the spectrum of complications of carotid stenting and the necessity to be able to think and act quickly. Balloon-expandable stents are not yet approved for mid cervical carotid applications, but their use was successful in this case.
References (Dr. Nanjudappa)
- Hom C. Takayasu arteritis. e medicine March 27, 2006.
- Chi YW, White CJ, Woods TC, Goldman CK. Ultrasound velocity criteria for carotid in-stent restenosis. Catheter Cardiovasc Interv 2007;69:349–354.
- Douglas JS Jr, Holmes DR Jr, Kereiakes DJ, et al. Cilostazol for ISR Trial (CREST) Investigators. Coronary stent ISR in patients treated with cilostazol. Circulation 2005;112:2826–2832.
- Peters S, Gotting B, Trummel M, et al. Valsartan for prevention of ISR after stenting of type B2/C lesions: The VAL-PREST trial. J Invasive Cardiol 2001;13:93–97.
- Vignali C, Bargellini I, Lazzereschi M, et al. Predictive factors of in-stent ISR in renal artery stenting: A retrospective analysis. Cardiovasc Intervent Radiol 2005;28:296–302.
- Chen MS, John JM, Chew DP, et al. Bare metal stent ISR is not a benign clinical entity. Am Heart J 2006;151:1260–1264.
- Kearney M, Pieczek A, Haley L, et al. Histopathology of in-stent ISR in patients with peripheral artery disease. Circulation 1997;95:1998–2002.
- Harper RW. Drug-eluting coronary stents — A note of caution. Med J Aust 2007;186:253–255.