Association of Atretic Left Internal Mammary Graft with Internal Mammary to Pulmonary Vasculature Fistula: A Case Series
A fistula between the left internal mammary artery (LIMA) and the pulmonary vasculature (PV) is a rare occurrence. Coronary artery bypass grafting (CABG) with a LIMA conduit is responsible for most iatrogenic cases; however, only a handful of cases have been reported in the literature. The cases reported in literature are usually the larger fistulae that are quite prominent on angiography and may be clinically significant due to hemodynamic shunting of blood and thus most are in the setting of a patent LIMA. Although mostly asymptomatic and discovered incidentally, these patients may occasionally present with angina or with a continuous murmur detected during physical examination.1–4
We report six consecutive cases of small LIMA to PV fistulae that were recognized incidentally on coronary angiography in a 1-year period. All were associated with an atretic LIMA graft. The association of atretic LIMA to pulmonary vasculature fistula has not been previously reported. We describe these cases and discuss possible mechanisms for this association.
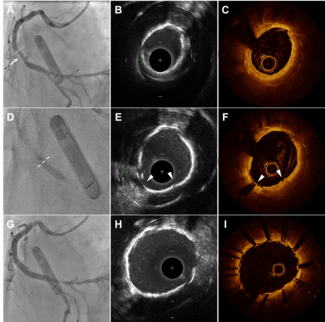
Case 1. A 54-year-old male with a CABG in 2001 with a LIMA to left anterior descending (LAD) and saphenous vein graft (SVG) to a second diagonal presents 6 years later with chest discomfort. His examination was unremarkable and a myocardial perfusion  scan did not show perfusion abnormalities. Cardiac catheterization was performed to evaluate his chest pain further. The left main artery had 50% ostial stenosis, LAD had 100% chronic total occlusion in the mid portion. Circumflex and right coronary artery did not have significant stenoses. SVG to second diagonal was patent and the LIMA to distal LAD was atretic with presence of a small fistula from distal LIMA to the distal PV via small vascular channels (Figure 1). Further work up identified Barrett’s esophagus and gastro-esophageal reflux as the likely etiology for the chest discomfort.
scan did not show perfusion abnormalities. Cardiac catheterization was performed to evaluate his chest pain further. The left main artery had 50% ostial stenosis, LAD had 100% chronic total occlusion in the mid portion. Circumflex and right coronary artery did not have significant stenoses. SVG to second diagonal was patent and the LIMA to distal LAD was atretic with presence of a small fistula from distal LIMA to the distal PV via small vascular channels (Figure 1). Further work up identified Barrett’s esophagus and gastro-esophageal reflux as the likely etiology for the chest discomfort.
Case 2. A 77-year-old male with a history of CABG in 1991 with a LIMA to the mid LAD presented with new onset exertional chest pain. Physical exam was unremarkable.  Cardiac catheterization revealed single vessel coronary disease with diffuse moderate to severe disease of the distal and apical LAD. LIMA graft to mid LAD was found to be atretic. There was a small fistula from the distal LIMA to the distal pulmonary vasculature (Figure 2). A 64-slice CT angiogram was performed to better define the fistulous connection of the atretic LIMA graft to a distal segment of the lingular branch of the left pulmonary artery (Figure 7).
Cardiac catheterization revealed single vessel coronary disease with diffuse moderate to severe disease of the distal and apical LAD. LIMA graft to mid LAD was found to be atretic. There was a small fistula from the distal LIMA to the distal pulmonary vasculature (Figure 2). A 64-slice CT angiogram was performed to better define the fistulous connection of the atretic LIMA graft to a distal segment of the lingular branch of the left pulmonary artery (Figure 7).
Case 3. A 62-year-old male with a history of CABG (LIMA to LAD, right radial graft to the RCA, and left radial graft to a ramus) in 2000 with presented with exertional angina. An exercise echocardiographic stress test showed ischemic ST depression. Cardiac catheterization revealed a 90% stenosis of the proximal LAD, 90% stenosis of the mid-distal circumflex, 70% stenosis of the proximal ramus, patent radial graft to the RCA and patent radial graft to Ramus. The LIMA graft to LAD was atretic with a small fistula that connected the mid LAD to the distal stump of the LIMA to the distal PV (Figure 3). The fistula opacified with antegrade injections to the native LAD. Ejection fraction was preserved at 50%.
Case 4. A 64-year-old male with a history of coronary artery disease and a CABG in 2007 (LIMA to LAD, RIMA to RCA, SVG to diagonal and SVG to the obtuse  marginal) presented with chest discomfort 6 months after the CABG. A myocardial perfusion scan revealed inferior ischemia which prompted cardiac catheterization. The catheterization revealed diffuse, but non-critical disease in the LAD. The LIMA graft was subtotally occluded distally with formation of a small LIMA to distal PV fistula (Figure 4). The saphenous vein graft to obtuse marginal was occluded at the aortic anastamosis. The saphenous vein graft to the diagonal branch was patent with mild disease. The RIMA to distal RCA was subtotally occluded with “neovascularization” around the distal portion of the RIMA though no definite fistula to the PV was seen. The patient underwent successful stenting of the proximal right coronary artery and mid left circumflex vessels. He had preserved left ventricular function.
marginal) presented with chest discomfort 6 months after the CABG. A myocardial perfusion scan revealed inferior ischemia which prompted cardiac catheterization. The catheterization revealed diffuse, but non-critical disease in the LAD. The LIMA graft was subtotally occluded distally with formation of a small LIMA to distal PV fistula (Figure 4). The saphenous vein graft to obtuse marginal was occluded at the aortic anastamosis. The saphenous vein graft to the diagonal branch was patent with mild disease. The RIMA to distal RCA was subtotally occluded with “neovascularization” around the distal portion of the RIMA though no definite fistula to the PV was seen. The patient underwent successful stenting of the proximal right coronary artery and mid left circumflex vessels. He had preserved left ventricular function.
Case 5. A 50-year-old woman who had undergone a single-vessel CABG with LIMA to mid LAD presented with recurrent angina. Cardiac catheterization showed an atretic LIMA graft with a small fistula from distal LIMA to distal pulmonary vasculature (Figure 5). She was treated with a stent to the ostial LAD. Circumflex and RCA were without significant stenosis.
Case 6. A 51-year-old woman with a CABG in 2007 (LIMA to LAD, radial graft to RCA) presented with chest discomfort that was reminiscent of her prior angina. Cardiac catheterization showed only moderate (40%) stenosis in proximal LAD and RCA. It appeared that the lesions were significantly better than prior to the CABG and it was thought that there might have been spasm in the pre CABG angiograms. Radial graft was occluded. LIMA graft was patent but was quite atretic in its distal segment though there was some flow into the LAD from the graft (Figure 6). There was a small fistula noted from the mid LIMA to the distal PV.
Discussion. LIMA to PV fistulae are categorized as either congenital or acquired. Acquired LIMA to PV fistulae have been attributed a multitude of etiologies some of which are pulmonary infections, neoplasms, trauma, and chest surgery.1
Occurrence of these fistulae after CABG is rare and the few reports available in literature have generally been case reports. These patients have generally had relatively large fistulae that have been almost always been in the presence of a patent LIMA graft.2–7
The literature typically describes patients with LIMA to PV fistulae who present with early recurrent angina1,8 or a continuous murmur.4 The murmur has been described as a systolic murmur in the pulmonary area and to the left sternal edge with an accompanying short early diastolic mumur.4,9 The hypothesis is that angina symptoms are a result of a coronary steal from the LAD to pulmonary artery.1 According to a review by Heper,10 16 of 21 reported cases in the literature of LIMA-PV fistulae occurring following CABG surgery presented with angina.
It has been proposed that treatment of fistulae is appropriate if there is development of angina, ischemia from coronary steal, evidence of an enlarging shunt with CHF or pulmonary hypertension, aneurysmal expansion, mycotic aneurysm formation or hemoptysis.11 Management of these larger fistulae that cause symptoms or ischemia has included surgical division of the fistulous communication,9,12 percutaneous coil-spring closure in the case of the non-grafted LIMA to pulmonary parenchymal fistula,13 deployment of a drug-eluting (cypher) stent to obliterate the fistula,14 poltetrafluroethylene (PTFE) coated balloon-expandable stent,11 and use of a bovine pericardium covered stent (BPCS).11
Current literature is generally silent regarding the possible pathogenesis of these fistulae. There has been speculation that the fistulae are acquired in LIMA grafts in which side branches have not been clipped but instead electrocoagulated15,16 allowing communication between these branches and opposing lung parenchyma with subsequent fistula formation.15 Meticulously micro-clipping all IMA branches as well as covering the IMA with a flap of pericardium has been recommended to prevent IMA-to PA fistula formation.1,15,16
The cases described in this article, however, differ from these previously described cases, as these fistulae are associated with an atretic LIMA graft. These are also quite small and likely asymptomatic. The fistulae that we report show minute collateral channels connecting the LIMA graft and the PV and not a single robust vascular channel as was seen in most of the prior reported LIMA to PV fistula in the setting of a patent LIMA. CT angiography can also define the detailed anatomy of these fistulae, as in Case 2. Here it showed communication with a distal of the lingular branch of the left pulmonary artery. However, CTA is generally not indicated for diagnosis of these fistulae.
These fistulae are quite small and are often missed or ignored, as the communication between with the distal PV is often not appreciated. In 5 of the 6 cases we report here, the initial catheterization report did not mention the fistula at all or reported some neovascularization/collateral channels at the distal end of the atretic LIMA.
We offer the following possible mechanisms for this association of LIMA atresia with PV fistulae:
1. An atretic LIMA may indicate technical difficulty during surgery (or less optimal surgical technique), leading to more peri-operative inflammation and adhesions around the LIMA graft and adjacent pleura resulting in neo-revascularization and connections between the LIMA and PV. 2. Small vascular connections between LIMA and PV develop more commonly after CABG but are not appreciated on angiography if the graft is patent, as the blood preferentially directs itself to the widely patent LIMA to LAD anastamosis. However, if the graft is atretic or ineffective due to a severe stenosis at its anastamosis with the LAD then these connections become angiographically manifest as the blood flow in the LIMA is preferentially directed to the fistula. 3. While association does not denote causality, it may be that the fistula contributes to LIMA graft failure as it may direct blood flow away from the LAD. This theory is less likely as these fistulas are very small and unlikely to compete effectively with a patent LIMA to LAD anastamosis.
Study limitations. This study is a preliminary study that describes six consecutively identified cases of PV fistula in the setting of an atretic LIMA graft. In order to further confirm this association ,and look at possible causative etiology a larger systematic review is needed where the prevalence of the fistulae is studied in patients with atretic and patent LIMA grafts so as to identify an increased prevalence in the atretic LIMA group and study any associated variables.
In conclusion, this study is the first to identify a possible association of atretic LIMA graft with the presence of PV fistulae and discusses possible mechanisms for this association. This study provides preliminary data for a larger, more systematic review of this association and to look at possible causation.
References
1. Kimmelsteil CD, Udelson J, Salem D, et al. Recurrent angina due to a left internal mammary artery-to-pulmonary artery fistula. Am Heart J 1993;125:234–236. 2. Madu EC, Hanamanthu SK, Kim C, et al. Recurrent ischemia resulting from left internal mammary artery to pulmonary artery fistula. Angiography 2001;52:185–188. 3. Birnbaum, Y, Wurzel M, Nili M, et al. An unusual cause of recurrent angina two years after coronary artery bypass grafting: Fistula between internal mammary artery graft to pulmonary vasculature. Cathet Cardiovasc Diagn 1992;27:13–132. 4. Guray U, Guray Y, Ozbakir C, et al. Fistulous connection between internal mammary graft and pulmonary vasculature after coronary artery bypass grafting: A rare of cause of continuous murmur. Internat J Cardiol 2004;96:489–492. 5. Liu Y, Noveck H, Moreyra AE, et al. Plexus between internal mammary graft and pulmonary vasculature. Texas Heart Inst J 2000;27:395–397. 6. Kieso, H, Trivedi D, Qazi M, et al. Acquired internal mammary artery to pulmonary artery fistula following bypass surgery. J Invasive Cardiol 2000;12:280–282. 7. Imawaki S, Arioka I, Nakai M, et al. Development of a fistula between an internal mammary artery graft and pulmonary vasculature following coronary artery bypass grafting: Report of a case. Surgery Today 1995;25:461–464. 8. Ferreira AC, Marchena E, Liester M, et al. Internal mammary to pulmonary artery fistula presenting as early recurrent angina after coronary bypass. Arq Bras Cardiol 2002;79:181–182. 9. Musleh G, Jalal A, Deiraniya AK. Post-coronary artery bypass grafting left internal mammary artery to pulmonary artery fistula: A 6 year follow-up following successful surgical division. Eur J Cardiothorac Surg 2001;20:1258–1260. 10. Heper G, Barcin C, Iyisoy A, et al. Treatment of an iatrogenic left internal mammary artery to pulmonary artery fistula with a bovine pericardium covered stent. Cardiovasc Intervent Radiol 2006;29:879–882. 11. Abbott JD, Brennan JJ, Remetz MS. Treatment of a left internal mammary artery to pulmonary artery fistula with polytetrafluoroethylene covered stents. Cardiovasc Intervent Radiol 2004;27:74–76. 12. Johnson JA, Schmaltz R, Landreneau RJ, et al. Internal mammary artery graft to pulmonary vasculature fistula: A cause of recurrent angina. Ann Thorac Surg 1990;50:297–298. 13. Blanche C, Eigler N, Bairey CN. Internal mammary artery to lung parenchyma fistula after aortocoronary bypass grafting. Ann Thorac Surg 1991;52:141–142. 14. Glover, BM, Khan MM. The novel use of a covered stent in the management of a left internal mammary artery to pulmonary vasculature fistula. J Invasive Cardiol 2004;16:663–664. 15. Groh WJ, Hovaguimian H, Morton MJ. Bilateral internal mammary-to-pulmonary artery fistulas after a coronary operation. Ann Thorac Surg 1994;57:1642–1643. 16. Wood MK. Internal mammary artery to lung parenchyma fistula. Ann Thorac Surg 1992;54:603.