Amplatzer Device Closure of a Tortuous Gerbode
(Left Ventricleto-Right Atrium) Defect Complicated by Transient Hemolysis in an Octogenarian
Author Affiliations: From the *Department of Pediatrics, University of Nevada School of Medicine and the Childrens Heart Center, Las Vegas, Nevada, and the §Department of Medicine, University of California San Diego, San Diego, California. The authors report no conflicts of interest regarding the content herein. Address for correspondence: Abraham Rothman, MD, 3006 S. Maryland Parkway, #690, Las Vegas, NV 89109. E-mail: rothman@childrensheartcenter.com
_______________________________________________ ABSTRACT: An 86-year-old male presented with progressive shortness of breath and a murmur. Six years earlier, he had undergone mitral valve surgery. An echocardiogram revealed a significant left ventricular-to-right atrial connection (Gerbode defect). The defect was closed percutaneously using an Amplatzer Septal Occluder device. After the procedure, the patient developed transient hemolysis and renal dysfunction. A year later, there was no residual flow on echocardiography and the patient reported marked improvement in exercise tolerance.
_______________________________________________
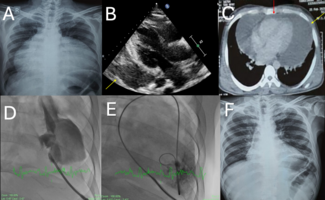
J INVASIVE CARDIOL 2008;20:E273–E276 Left ventricular-to-right atrial shunts, also called Gerbode defects, are very rare cardiac lesions. They can be congenital or acquired after surgery,1–4 infection,5–7 trauma8 or myocardial infarction.9,10 Gerbode et al described the nature and repair of these defects in 1958.11 If hemodynamically significant, these defects can cause a volume load of both right- and left-sided cardiac chambers and symptoms. They may also cause endocarditis and arrhythmias.10 In this report, we describe a significant Gerbode defect detected 6 years after mitral valve surgery in an octogenarian. The defect was closed percutaneously using an Amplatzer septal occluder device. Case Report. An 86-year-old man presented with progressive shortness of breath, peripheral edema, decreased exercise tolerance and a murmur. He had a history of rheumatic mitral regurgitation and 6 years earlier had undergone mitral valve repair followed 2 days later by mitral valve replacement using a 27 mm Medtronic Hancock II prosthesis (Medtronic, Inc., Minneapolis, Minnesota). The postoperative course was long and complicated with frequent fevers and placement of temporary tracheostomy and gastrostomy tubes. At age 84, he had a stent placed in the left anterior descending coronary artery. An echocardiogram revealed a left ventricular-to-right atrial fistula, which appeared to be hemodynamically significant. There was good left ventricular function, mild tricuspid regurgitation and moderate pulmonary hypertension by increased tricuspid regurgitation jet velocity. Cardiac catheterization was performed under conscious sedation. A 5 Fr sheath was placed in a femoral artery and 8 Fr sheaths were placed in both femoral veins. One venous sheath was used for intracardiac echocardiography (ICE) (AccuNav, Siemens Medical, Malvern, Pennsylvania). The following pressures were obtained: right atrium mean 12 mmHg, with a v-wave of 18 mmHg, pulmonary artery 60/15, mean 31 mmHg, and left ventricle 100/15 mmHg. The pulmonary-to-systemic flow ratio was 1.7. By ICE, the defect was noted to be tortuous, measuring approximately 15 mm on the left ventricular side and 10–11 mm at its minimum diameter (Figure 1). The left ventriculogram showed a funnel-shaped origin of the fistula on the left ventricular side measuring about 22 mm (Figure 2). A cut-off pigtail catheter was advanced through the femoral arterial sheath to the left ventricular origin of the defect. An exchange-length Wholey wire (Mallinckrodt Inc., Hazelwood, Missouri) was advanced to the right atrium where it was snared and exteriorized through the right femoral venous sheath. A 20 mm sizing balloon (NMT Medical Inc., Boston, Massachusetts) was advanced over the guidewire through the venous sheath and inflated gently in the defect. The balloon waist measured 8 mm (Figure 3). An 8 Fr long delivery sheath was placed in the right femoral vein and advanced over the veno-arterial wire to the left ventricle. The guidewire was removed. A 10 mm Amplatzer septal occluder device (AGA Medical Corp., Plymouth, Minnesota) was delivered with the distal disc in the left ventricle and the proximal disc in the right atrium. Good position was confirmed by echocardiography and angiography. The device was released. Final left ventriculography and echocardiographic imaging showed mild residual left-to-right flow through the device (Figures 4 and 5). Total contrast use was 138 cc of iopamidol (Isovue 370, Bracco Diagnostics, Inc., Princeton, New Jersey). Within a few hours, the patient developed hemolysis. He required packed red blood cell transfusions approximately every other day. The serum creatine increased from a baseline of 1.5 mg/dl to a maximum of 3.4 mg/dl, and the total bilirubin reached a peak of 7.6 mg/dl. The hemolysis and renal dysfunction resolved spontaneously 12 days after the procedure. An echocardiogram 17 days after the procedure showed trivial residual leak through the device. Echocardiograms 4 and 12 months after the procedure showed no residual flow from the left ventricle to the right atrium and the patient reported marked improvement in exercise tolerance. Discussion. Perry, Burchell and Edwards described anatomic variations of left ventricular-to-right atrial communications in 1949.12 Gerbode et al elegantly reported the clinical and intraoperative findings and surgical repair in 5 patients with these defects.11 In 4 of the 5 patients, the communication included a defect in the septal leaflet of the tricuspid valve. Thickening of the tricuspid valve leaflets suggested to the authors that infection or shear stress were the possible causes. Riemenschneider and Moss classified left ventricular-to-right atrial communications as supravalvular or infravalvular, depending on whether the defect was above or below the tricuspid valve.13 Our patient had a history of rheumatic mitral regurgitation and underwent unsuccessful mitral valve repair followed by a prosthetic valve implantation 2 days later. He had a long and complicated postoperative course with frequent fevers. It is not clear if the Gerbode defect was iatrogenic from the surgeries, or a result of infections in the convalescent period. Six years after the mitral valve surgery, the defect was detected on echocardiography. Repeat surgery to fix the Gerbode defect was considered but refused by the patient and his family. An attempt at transcatheter closure was proposed. At the time the procedure was being contemplated, there were scant reports of percutaneous closure of these types of acquired defects.14 Since then, Lorber et al described closure of a Gerbode defect acquired following mitral valve replacement using an Amplatzer duct occluder device,15 and Trehan et al reported closure of a Gerbode defect acquired following surgical closure of a ventricular septal defect using an Amplatzer muscular ventricular septal defect occluder device.16 In our patient, the fistula was long, tortuous and difficult to define. Balloon sizing was performed to aid with device size selection. A 10 mm Amplatzer septal occluder was chosen because the device waist slightly oversized the minimum sizing balloon diameter, the left-sided device disc was close to the diameter of the funnel-shaped left ventricular origin of the defect and the device had enough material on both sides to allow firm anchoring. An Amplatzer duct occluder device was considered, but the absence of a retention disc on the right atrial side was felt to be a disadvantage. At the time of the procedure, the Amplatzer muscular VSD occluder and the Gore Helex device had not received FDA approval and were not available for our use. The design of the Helex device could have minimized the propensity for residual shunting and hemolysis. A CardioSeal device (NMT Medical) would have probably fit with the two umbrellas partially collapsed inside the irregular defect, however, with the potential disadvantage of limited retrievability if malpositioned. Coils were considered, but were felt to be too unstable and not bulky enough for this type of high-pressure and flow lesion. A veno-arterial wire method was employed, similar to that used to close muscular and membranous ventricular septal defects.17 The defect was crossed easily with a very floppy Wholey wire. Snaring and exteriorization of the wire was also uncomplicated. The veno-arterial wire made it easy to advance the long delivery sheath to the left ventricle. Deployment of the device was aided by intracardiac ultrasound imaging, which demonstrated that the device was not interfering with the prosthetic mitral valve, the tricuspid valve or the coronary sinus ostium. The onset and resolution of hemolysis and hemoglobinurina coincided with the onset and resolution of the renal dysfunction. The etiology of the renal failure, therefore, was most likely due to effects of hemolysis on the kidneys. However, a contribution of the contrast material to this patient’s baseline renal dysfunction (estimated glomerular filtration rate 47 ml/min/m2) is also possible. Preprocedural hydration to prevent contrast-induced nephropathy was considered but not performed due to concerns of exacerbating the volume load on the preexisting left-to-right shunt lesion. Hemolysis has been described in patients after percutaneous closure of ventricular septal defects and patent ductus arteriosus.18–20 The porous nature of the Amplatzer and similar devices make this possible. No clear predictive factors for hemolysis have been identified. The large pressure gradient between left ventricle and right atrium and the stretched configuration of the device after deployment may have contributed to the development of hemolysis in our patient. These devices become impermeable initially by thrombus formation followed by progressive endothelialization. Patients are usually given antiplatelet agents for 6 months after device implantation. Our patient’s rhythm alternated between sinus and atrial flutter/fibrillation. He therefore had two conditions requiring opposing therapies: hemolysis and intermittent atrial fibrillation. The treatment for the former was to avoid antithrombosis agents so that the device could “seal”, while treatment for atrial fibrillation involved the use of antithrombotic agents and avoidance of embolic events. Low-dose subcutaneous low-molecular weight heparin was not started until 8 days after the procedure when hemolysis began abating. We presume that enough device thrombosis and endothelial cell formation eventually changed the flow mechanics and resolved the hemolysis. Four months later, the defect was completely closed on echocardiography. In conclusion, percutaneous device closure offers an alternative to surgical closure in patients with Gerbode defects. Albeit self-limiting, hemolysis with renal dysfunction may occur with permeable devices in high-pressure defects. We speculate that hemolysis was a rare, isolated event in our patient; however, more experience in similar circumstances will be revealing. Design of less permeable occlusion devices in the future may obviate this complication.
1. Silverman NA, Sethi GK, Scott SM. Acquired left ventricular-right atrial fistula following aortic valve replacement. Ann Thorac Surg 1980:30:482–486.
2. Seabra-Gomes R, Ross DN, Gonzalez-Lavin L. Iatrogenic left ventricular--right atrial fistula following mitral valve replacement. Thorax 1973;28:235–241.
3. Marsten JL, Hildner FJ. Left ventricular-right atrial communication following valve replacement. J Thorac Cardiovasc Surg 1969;58:588–591.
4. Kudo T, Ryo S, Shimakura T, et al. [LV-RA shunt developing after repair of VSD)]. Kyobu Geka 1974;27:93–98.
5. Battin M., Fong LV, Monro JL. Gerbode ventricular septal defect following endocarditis. Eur J Cardiothorac Surg 1991;5:613–614.
6. Ollitrault J, Pitel C, de Place C, et al. [Left ventricle-right atrium communications acquired in bacterial endocarditis]. Arch Mal Coeur Vaiss 1985;78:1833–1839.
7. Velebit V, Schonenberg A, Ciaroni S, et al. “Acquired” left ventricular-to-right atrial shunt (Gerbode defect) after bacterial endocarditis. Tex Heart Inst J 1995;22:100–102.
8. Olsovsky MR, Topaz o, DiSciascio G, Vetrovek GW. Acute traumatic ventricular septal rupture. Am Heart J 1996;131:1039–1041.
9. Newman JN Jr, Rozanski L, Kreulen T. Acquired left ventricular to right atrial intracardiac shunt after myocardial infarction: A case report and review of the literature. J Am Soc Echocardiogr 1996;9:716–720.
10. Raja Y, Jenkins N, Chauhan A, Millner RW. Acquired post-infarct Gerbode defect complicated by infective endocarditis with giant right atrial vegetation. Int J Cardiol 2006;113:E79–E80.
11. Gerbode F, Hultgren H, Melrose D, Osborn J. Syndrome of left ventricular-right atrial shunt; Successful surgical repair of defect in five cases, with observation of bradycardia on closure. Ann Surg 1958;148:433–446.
12. Perry El, Burchell HB, Edwards JE. Congenital communication between the left ventricle and the right atrium; Co-existing ventricular septal defect and double tricuspid orifice. Proc Staff Meet Mayo Clin 1949;24:198.
13. Riemenschneider TA, Moss AJ. Left ventricular-right atrial communication. Am J Cardiol 1967:19:710–718.
14. Cabalka AK, Hagler DJ, Mookadam F, et al. Percutaneous closure of left ventricular-to-right atrial fistula after prosthetic mitral valve rereplacement using the Amplatzer duct occluder. Catheter Cardiovasc Interv 2005;64:522–527.
15. Lorber A, Nair P, Gruberg L. Transcatheter closure of acquired Gerbode defect following mitral valve replacement using the Amplatzer duct occluder. J Invasive Cardiol 2006;18:E264–E266.
16. Trehan V, Ramakrishnan S, Goyal NK. Successful device closure of an acquired Gerbode defect. Catheter Cardiovasc Interv 2006:68:942–945.
17. Knauth AL, Lock JE, Perry SB, et al. Transcatheter device closure of congenital and postoperative residual ventricular septal defects. Circulation 2004;110:501–507.
18. Spence MS, Thompson JD, Weber N, Qureshi SA. Transient renal failure due to hemolysis following transcatheter closure of a muscular VSD using an Amplatzer muscular VSD occluder. Catheter Cardiovasc Interv 2006;67:663–667.
19. Anil SR, Sivakumar K, Philip AK, et al. Clinical course and management strategies for hemolysis after transcatheter closure of patent arterial ducts. Catheter Cardiovasc Interv 2003;59:538–543.
20. Chessa M, Carminati M, Cao QL, et al. Transcatheter closure of congenital and acquired muscular ventricular septal defects using the Amplatzer device. J Invasive Cardiol 2002;14:322–327.