Ureteroarterial Fistula: A Rare Cause of Hematuria Managed in the Cardiac Cath Lab
_______________________________________________
J INVASIVE CARDIOL 2008;20:E158-E160
Ureteroarterial fistulae (UAFs) are rare but potentially fatal entities that require prompt diagnosis and intervention. Moschowitz and colleagues were the first to describe this entity in 1908. There has been an increase in frequency of this entity due to various factors including the incorporation of radiation and chronic ureteral stent exchange in the management of gynecologic and urologic cancers, combined with increased reporting of this complication.1,2 This type of fistula has been reported to be managed by urologists, vascular surgeons and interventional radiologists. However, this is the first reported case of a UAF managed by interventional cardiologists, and this is also the oldest woman reported to have developed this rare entity after radiation therapy. Transcatheter coil embolization of the left internal iliac artery successfully excluded the left UAF, resulting in sustained resolution of the hematuria.
Case Report. An 87-year-old Caucasian female presented to our institution in April of 2007 with a 3-month history of intermittent gross hematuria and a hemoglobin of 8.9 g/dL. In 1997 she was diagnosed with adenocarcinoma of the uterus, which required her to undergo a hysterectomy and radiation therapy.
Subsequently, she developed a left ureteral stricture related to the prior radiation therapy, which was managed with chronic ureteral stent exchanges every 3 months for the past 3 years. On admission, a computed tomographic (CT) scan of the abdomen and pelvis revealed a left hydronephrosis and hydroureter, as well as blood in the left kidney, left ureter and urinary bladder. A DTPA (diethylenethiaminepentaacetic acid) nuclear study revealed that the left kidney was contributing 52 percent of the GFR (glomerular filtration rate).
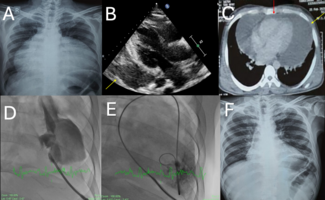
Urologists at our institution performed a cystoscopy, retrograde pyelogram and ureteroscopy, which revealed multiple areas of left ureteral stricture and left hydronephrosis. There was no evidence of active bleeding or an obvious mass, however, the ureteral mucosa near the left internal iliac artery revealed bounding, pulsatile changes (see Figure 1 for normal anatomy of ureter and iliac vessels). During ureteroscopy, a 7 mm x 24 mm double-J ureteral stent was replaced. The patient was asymptomatic for 24 hours post procedure, but thereafter had a recurrence of the gross hematuria. Subsequently, the patient was referred to interventional cardiology, where selective iliac arteriography from a left femoral artery approach revealed a small outpouching from the left internal iliac artery trunk as it traversed the ureter (Figure 2). Next, a Glidewire (Terumo Medical, Tokyo, Japan) was inserted into the left internal iliac artery, then retrogradely into the left ureter toward the renal pelvis, and the location of the ureteroarterial fistula was confirmed (Figure 3). This intentionally provocative maneuver (Glidewire insertion into the ureter) induced only minimal bleeding, therefore, contrast extravasation into the ureter was not visualized. However, the urinary bladder catheter had evidence of minimal hematuria after this maneuver. Next, utilizing the right femoral artery approach, 5 Tornado® coils (Cook Medical, Bloomington, Indiana) were deployed in the proximal left internal iliac artery. The final arteriogram revealed successful embolization and complete occlusion of the proximal left internal iliac artery with exclusion of the fistula (Figure 4). The patient was discharged 3 days after the intervention with resolution of the hematuria. Upon admission, she had received a total of 2 units of packed red blood cells, and upon discharge, her hemoglobin was stable at 10.8 g/dL. After 7 months, our patient has reported no recurrences of hematuria.
Discussion. Before 1980 there was a 69% mortality rate associated with UAFs, and thereafter, the mortality rate has decreased to 14–23% due to the utilization of new diagnostic and therapeutic modalities.3,4 In a review by Batter and colleagues of 37 patients with UAFs, 5 predisposing factors for the development of UAFs were identified: (1) previous genitourinary or pelvic surgery (68%); (2) chronic ureteral stent placement (65%); (3) radiation therapy (46%); (4) previous vascular surgery (19%); and (5) underlying vascular pathology (19%).2–6 Carcinomas involving the cervix, bladder, endometrium, rectum and vulva can be associated with this type of fistula, especially when surgery, irradiation or chronic ureteral stent exchange are involved.1,2 Other conditions or procedures associated with this rare entity include abdominal aortic aneurysms (AAAs), ureteral lithiasis, appendectomy, pelvic abscesses, trauma, diverting urinary conduits, aneurysms of pelvic vessels and pyelonephritis during pregnancy.2,3,5
The pathophysiology of UAF formation begins with damage to one or both components of the fistula: the ureter and/or the artery. The constant pulsation of an adjacent artery transmitted to an already radiation-induced damaged ureter may produce pressure necrosis at the site where the ureter crosses the iliac artery, resulting in the formation of a fistula between the two structures due to activation of the inflammatory cascade.4
The most common presentation of a ureteroarterial fistula is intermittent or continuous gross hematuria.7 Intermittent hematuria occurs in cases where the fistula may be occluded by the ureteral stent acting like a valve, therefore hematuria occurs whenever there is a favorable pressure gradient between the artery and the ureter.8 Patients occasionally present with flank and/or groin pain that is lateralized to the side of the fistula and sometimes, though rarely, present with hemorrhagic shock.4,7
Moreover, establishing the diagnosis is potentially challenging because the fistula can be occluded intermittently by a blood clot, thereby reducing the chance of observing the fistula on a diagnostic study.9 Diagnostic modalities that have been utilized to demonstrate this type of fistula include retrograde ureterography, aortography, nonselective iliac arteriography and provocative iliac arteriography. Nonselective arteriography alone may be insufficient for diagnosis, thus incorporating intentional provocative maneuvers during arteriography may aid in localizing the fistula. Keller and colleagues found that removal of a ureteral stent during arteriography coupled with provoking bleeding by moving a deflated angioplasty balloon to and from within the fistula aids in the diagnosis.10 At our institution, we promptly localized the fistula by intentionally inducing minimal hematuria after Glidewire insertion into the ureter during arteriography. Additionally, appropriate precautions were taken to support the patient and intervene in the event of sudden massive bleeding.
Standard arteriography has demonstrated a sensitivity of 23–41% in detecting ureteroarterial fistulae, while provocative angiography increased the sensitivity to 100% in a single series. A negative provocative angiogram does not necessarily exclude the diagnosis of a fistula, thus the physician must rely on a high index of suspicion.4
Various modalities for the management of UAFs have been utilized by urologists, vascular surgeons and interventional radiologists. This is the first reported management of this rare entity by interventional cardiologists. Urologists have utilized ureteral coiling with subsequent percutaneous nephrostomy, ureteral ligation and ipsilateral nephrectomy.11 Interventional radiologists have incorporated endovascular coiling, embolization and/or stent grafting during arteriography.2,7 Vascular surgeons treat this condition by primary repair of the iliac artery with a patch or graft, extra-anatomic arterial bypass after surgical ligation or embolization, depending on which vessel is obliterated.1
The common iliac artery has been reported to be involved more often than the internal or external iliac arteries.4,6 When embolization of the common or external iliac arteries is required, arterial bypass grafting is utilized to restore perfusion to the lower extremity.1 Covered stents or endografts may be an option as well. However, with internal iliac artery occlusion, arterial bypass grafting is not required, except when it provides extensive collateral blood flow to the colon, reproductive organs and the spinal cord in the presence of extensive atherosclerosis.
Currently, there are no published diagnostic or therapeutic algorithms for UAF management that are based on large prospective studies. However, Krambeck and colleagues have proposed a treatment algorithm for this type of fistula based on their large-scale, single-institution case series.4 With the advent of new diagnostic and therapeutic modalities, the rates of unnecessary exploratory lapartomy, ipsilateral nephrectomy, renal artery embolization and ureteral ligation for the management of UAFs have been tremendously reduced.
Ureteroarterial fistulae are rare yet potentially lifethreatening entities that usually present with continuous or intermittent gross hematuria. Chronic indwelling ureteral stents, irradiation and vascular abnormalities can predispose patients to fistula formation. Awareness of this condition along with a high index of suspicion will aid the clinician in prompt diagnosis and treatment. Provocative measures during arteriography increase the sensitivity in demonstrating the location of the fistula. A multidisciplinary approach among interventional cardiologists or radiologists, vascular surgeons and urologists will help determine the best case-specific treatment modality.
References
1. Quillin SP, Darcy MD, Picus D. Angiographic evaluation and therapy of ureteroarterial fistulas. Am J Roentgenol 1994;162:873–878.
2. Feuer DS, Ciocca RG, Nackman GB, et al. Endovascular management of ureteroarterial fistula. J Vasc Surg 1999;30:1146–1149.
3. Vandersteen DR, Saxon RR, Fuchs E, et al. Diagnosis and management of ureteroiliac artery fistula: Value of provocative arteriography followed by common iliac artery embolization and extraanatomic arterial bypass grafting. J Urology 1997;158:754–758.
4. Krambeck AE, DiMarco DS, Gettman MT, Segura JW. Ureteroiliac artery fistula: Diagnosis and treatment algorithm. Urology 2005;66:990–994.
5. Batter SJ, McGovern FJ, Cambria RP. Ureteroarterial fistula: Case report and review of the literature. Urology 1996;48:481–489.
6. Baldwin ZK, Sokoloff MM, Gerber GS, Schwartz LB. Ureteroarterial fistula with ruptured anastomotic pseudoaneurysm: Successful management with vascular exclusion, extra-anatomic bypass and nephrectomy. Vasc Endovasc Surg 2005;39:203–206.
7. Meester DJ, van Muiswinkel KW, Ameye F, et al. Endovascular treatment of a ureteroiliac fistula associated with ureteral double J-stenting and an aorticbifemoral stent graft for an inflammatory abdominal aortic aneurysm. Ann Vasc Surg 2006;20:408–410.
8. Bilbao JI, Cosin O, Bastarrika G, et al. Treatment of ureteroarterial fistulae with covered vascular endoprostheses and ureteral occlusion. Cardiovasc Interv Radiol 2005;28:159–163.
9. Fu JH, Liang HL, Yu CC, et al. Percutaneous endovascular repair of a ureteroarterial fistula with a stent graft. J Chin Med Assoc 2006;69:387–390.
10. Keller FS, Barton RE, Routh WD, Gross GM. Gross hematuria in two patients with ureteral-ileal conduits and double-J stents. J Vasc Interv Radiol 1990;1:69–77.
11. Inoue T, Hioki T, Arai Y, et al. Ureteroarterial fistula controlled by intraluminal ureteral occlusion. Int J Urology 2002;9:120–121.