What Is This Pink Lesion on the Palm?
Case Report

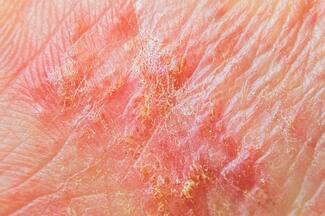
A 71-year-old man with a history of a prostate adeno-carcinoma and chronic lymphocytic leukemia for which he has been properly treated and monitored by the oncology department presented to our clinic with a single pink growth on the palm of his left hand (Figure 1). It appeared 1 year prior to his presentation to the clinic with a slow but steady increase in size. The patient mentioned that he applied Compound W several times over a period of 6 months with no improvement. He had no family or personal history of skin cancer. Cutaneous examination revealed a well-circumscribed, slightly tender, pink, telangiectatic nodule measuring 1.2 cm x 1.1 cm. The patient did not exhibit any other skin lesions and reported no systemic symptoms. An excisional biopsy was performed.
What’s Your Diagnosis?
Continue below for the answer.
Diagnosis
Eccrine Poroma
Poromas are benign adnexal neoplasms characterized by terminal duct differentiation in sweat glands. There are two main types: eccrine and apocrine poroma. Eccrine poroma was first described in 1956 and accounts for 10% of all sweat gland tumors.1 Numerous reports suggest that poroma may originate from either eccrine or apocrine lineage.2 Despite their similar clinical features, differentiation between eccrine and apocrine poroma is achieved through histopathology.
The chief finding of eccrine poroma is a circumscribed proliferation of compact cuboidal keratinocytes with small monomorphous nuclei with scant eosinophilic cytoplasm.3 The aggregates of poroid and cuticular cells in a poroma may show ductal or tubular formations. If the poroma has greater tubular foci lined by columnar cells with holocrine secretions, it strongly suggests an apocrine etiology.4
Some authors used to describe eccrine poroma with the term eccrine acrosprinoma as a synonym. On the other hand, some suggest that the term acrosprinoma is part of a broader group of neoplasms, including nodular hidradenoma, clear cell hidradenoma, hidroacanthoma simplex, and hidradenoma.4
Clinical Presentation
Poromas typically present as solitary normochromic or erythematous papules, plaques, or nodules. Although poromas are generally found on acral locations, they can appear on almost any cutaneous surface. The most common location is on the palmar or plantar surface.5 Pigmented lesions are uncommon. Clinical manifestations can vary widely, and their appearance can mimic both malignant and benign pathologies, such as amelanotic melanoma, basal cell carcinoma, squamous cell carcinoma, and pyogenic granuloma.
The lesion usually grows slowly and is often asymptomatic. Poromatosis is a rare clinical variant characterized by multiple poromas in an acral or widespread distribution, which can occur following chemotherapy and/or radiotherapy.6
Epidemiology
Sweat gland tumors account for roughly 1% of cases in primary skin lesions. Eccrine and apocrine poromas are believed to account for approximately 10% of these tumors.4,7 Poroma is not known to have any ethnic or racial predilection. It affects mostly adults, with an average age of 55 to 57 years, and with an equal sex predilection.8
Although rare, the malignant transformation risk of poroma is present. Primary eccrine porocarcinoma is observed less frequently than poroma. In a meta-analysis of 453 patients with eccrine porocarcinoma, the age of patients ranged from 6 months to 97 years, 49% of cases were men, and 51% were women.9
Etiopathogenesis and Histopathology
The exact cause of the disease remains unclear, although several factors have been proposed to be associated with it, including radiation, trauma, pregnancy, chemotherapy drugs, and human papillomavirus infection.10,11 Histopathologically, poroma involves both the epidermis and dermis. Poroma is categorized into 4 subtypes: hidroacanthoma simplex, poroid hidradenoma, dermal duct tumor, and eccrine poroma.4
The major finding in all subtypes of poroma consists of a circumscribed proliferation of compact cuboidal keratinocytes with small monomorphous nuclei and scant eosinophilic cytoplasm. There is an distinct demarcation between the keratinocytes of the adjacent epidermis and the monomorphic, slightly smaller, cuboidal poroma cells. The tumor cells are united by conspicuous intercellular bridges. Confirmation of the diagnosis is provided by features of maturation, either into ductal lumina with 1 row of luminal cells coated by an eosinophilic lining or, more frequently, into cystic areas empty of any formal lining.12 The degree of ductal differentiation varies greatly in individual neoplasms. Carcinoembryonic antigen immunostaining, which labels the luminal surface of each apocrine and eccrine duct, is utilized as a tool to verify the presence of ductal differentiation.2 The tumor is supported by a fine fibrovascular stroma.
Hidroacanthoma simplex, also known as intraepidermal poroma, is characterized by well-demarcated, uniform poroid cells that may contain cystic or ductal structures in the epidermis. In contrast, poroid hidradenoma and dermal duct tumors are located entirely in the dermal layer.13 Eccrine poroma occurs within the lower portion of the epidermis and extends into the dermis. Occasionally, there may be pigmented variants of eccrine poromas, which can be explained by 2 theories: The first refers to the melanocytes present in the cellular beginnings of the eccrine ducts during the 14th week of intrauterine development, which were not eliminated during the maturation process. The other hypothesis is that the origin of pigmentation comes from epidermal melanocytes.14
Porocarcinoma is composed of anaplastic cells that contain large, irregular, hyperchromatic nuclei and glycogen-rich cytoplasm. Additionally, it shows areas of necrosis and mitotic figures. Porocarcinoma is typically initially characterized by intraepidermal proliferation, which then extends into the dermis. Later on, dermal lymphatics are invaded, and the tumor demonstrates regional and distant metastasis.15
Differential Diagnosis
Poroma is often misdiagnosed, as with other skin neoplasms, because its clinical presentation is nonspecific and variable (eTable). Dermoscopic examination may provide valuable clues for diagnosis of poroma. The white interlacing areas around vessels, yellow structureless areas, milky-red globules, and poorly visualized vessels are the most prominent dermoscopic findings that aid in diagnosis.16 However, histopathologic examination is required for a definitive diagnosis.

Treatment
Poroma is a benign adnexal neoplasm, and superficial or deeper lesions can be treated with excisional surgery. Porocarcinomas have shown a local recurrence rate of about 20%, lymph node metastases in 20%, and solid organ metastases in 10%.17 Given the high rate of local recurrence, wide surgical excision with broad tumor margins or Mohs micrographic surgery is generally considered the treatment of choice.18 Radiotherapy and chemotherapy can be applied in necessary cases.
Our Patient

The primary clinical impression of our patient favored eccrine poroma considering the morphologic appearance and acral location. However, other neoplasms could not be ruled out. Therefore, an excisional biopsy of the lesion was performed.
The evaluation of the biopsy specimen revealed a circumscribed proliferation of compact, cuboidal keratinocytes with small monomorphous nuclei and scant eosinophilic cytoplasm (poroid cells) (Figure 2 and Figure 3). The diagnosis of eccrine poroma was confirmed histopathologically. Because there was no cellular atypia and the surgical margins of the lesions were clear, no additional treatment was required. No recurrence was observed during the patient’s follow-up.

Conclusion
The clinical presentation of poroma can resemble that of various benign and malignant tumors. Therefore, a biopsy is essential to confirm the diagnosis. Additionally, the possibility of malignant transformation in poromas, although minimal, encourages the excision of the lesion.
References
1. Pinkus H, Rogin JR, Goldman P. Eccrine poroma: tumors exhibiting features of the epidermal sweat duct unit. AMA Arch Dermatol. 1956;74(5):511-521.
2. Harvell JD, Kerschmann RL, LeBoit PE. Eccrine or apocrine poroma? Six poromas with divergent adnexal differentiation. Am J Dermatopathol. 1996;18(1):1-9. doi:10.1097/00000372-199602000-00001
3. McCalmont TH. A call for logic in the classification of adnexal neoplasms. Am J Dermatopathol. 1996;18(2):103-109. doi:10.1097/00000372-199604000-00001
4. Sawaya JL, Khachemoune A. Poroma: a review of eccrine, apocrine, and malignant forms. Int J Dermatol. 2014;53(9):1053-1061. doi:10.1111/ijd.12448
5. Johnson RC, Rosenmeier GJ, Keeling JH 3rd. A painful step. Eccrine poroma. Arch Dermatol. 1992;128(11):1530, 1533. doi:10.1001/archderm.128.11.1530
6. Miura T, Yamamoto T. Eruptive poromatosis following radiotherapy. Am J Dermatopathol. 2013;35(5):615-617. doi:10.1097/DAD.0b013e3182743abe
7. Betti R, Bombonato C, Cerri A, Moneghini L, Menni S. Unusual sites for poromas are not very unusual: a survey of 101 cases. Clin Exp Dermatol. 2014;39(2):119-122. doi:10.1111/ced.12185
8. Kang S, Amagai M, Bruckner AL, et al. Fitzpatrick’s Dermatology. 9th ed. McGraw- Hill Education; 2019.
9. Salih AM, Kakamad F, Baba HO, et al. Porocarcinoma; presentation and management, a meta-analysis of 453 cases. Ann Med Surg (Lond). 2017;20:74-79. doi:10.1016/j.amsu.2017.06.027
10. Chen CC, Chang YT, Liu HN. Clinical and histological characteristics of poroid neoplasms: a study of 25 cases in Taiwan. Int J Dermatol. 2006;45(6):722-727. doi:10.1111/j.1365-4632.2006.02741.x
11. Moon J, Lee JS, Park H-s, Yoon H-S, Cho S. Eccrine poroma with rapid growth during pregnancy: a case report and review of the literature. Ann Dermatol. 2018;30(2):222-225. doi:10.5021/ad.2018.30.2.222
12. Eduardo Calonje TB, Alexander J. Lazar, Steven D. Billings Tumors of the sweat glands. In: Calonje E. McKee’s Pathology of the Skin. 5th ed. Elsevier; 2020: 1611- 1679.
13. Obaidat NA, Alsaad KO, Ghazarian D. Skin adnexal neoplasms—part 2: an approach to tumours of cutaneous sweat glands. J Clin Pathol. 2007;60(2):145-159. doi:10.1136/jcp.2006.041608
14. Lacy R, Vega E, Domínguez L, Guevara E. Poroma: a clinico-pathologic study of 45 cases. Med Cutan Iber Lat Am. 2003;31(1):17-22.
15. Jan NA, Masood S. Poroma. StatPearls [Internet]. StatPearls Publishing; 2021.
16. Aydingoz I. New dermoscopic vascular patterns in a case of eccrine poroma. J Eur Acad Dermatol Venereol. 2009;23(6):725-726. doi:10.1111/j.1468- 3083.2009.03182.x
17. Huet P, Dandurand M, Pignodel C, Guillot B. Metastasizing eccrine porocarcinoma: report of a case and review of the literature. J Am Acad Dermatol. 1996;35(5 Pt 2):860-864. doi:10.1016/s0190-9622(96)90105-x
18. Song SS, Wu Lee W, Hamman MS, Jiang SI. Mohs micrographic surgery for eccrine porocarcinoma: an update and review of the literature. Dermatol Surg. 2015;41(3):301-306. doi:10.1097/DSS.0000000000000286