ADVERTISEMENT
Right Pulmonary Artery to Left Atrium Fistula: Transcatheter Closure Without Conventional Venovenous Loop
ABSTRACT: Right pulmonary artery to left atrium fistula is a rare anatomic variation of pulmonary arteriovenous malformation in which the initial connection exists between a pulmonary artery and a pulmonary vein, but during vascular development the pulmonary vein gets incorporated into the left atrium. Though nearly 60 such cases have been reported in literature, only 6 cases have been tackled by transcatheter technique with various types of devices and coils. This is a case where we demonstrate that large fistulae can be closed successfully with an atrial septal occluder without a conventional method of transseptal puncture and venovenous loop formation.
J INVASIVE CARDIOL 2012;24(7):E145-E147
Key words: Pulmonary arteriovenous fistula, arteriovenous malformation, interventional cardiology
____________________________________________________
 Case Report. An 8-year-old boy weighing 19 kgs presented with history of cyanosis and exertional dyspnea. He was saturating 85% in room air. He had a normal second heart sound and there was no murmur, which made us consider pulmonary arteriovenous fistula. On 2D echocardiography, the right pulmonary artery (RPA) appeared larger than left pulmonary artery and the left atrium (LA) was dilated, which were indirectly suggestive of a fistula from RPA to LA. There were no other cardiac anomalies. So we planned to catheterize the patient to delineate the morphology of the fistula and percutaneous closure in the same sitting.
Case Report. An 8-year-old boy weighing 19 kgs presented with history of cyanosis and exertional dyspnea. He was saturating 85% in room air. He had a normal second heart sound and there was no murmur, which made us consider pulmonary arteriovenous fistula. On 2D echocardiography, the right pulmonary artery (RPA) appeared larger than left pulmonary artery and the left atrium (LA) was dilated, which were indirectly suggestive of a fistula from RPA to LA. There were no other cardiac anomalies. So we planned to catheterize the patient to delineate the morphology of the fistula and percutaneous closure in the same sitting.
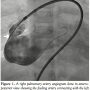
 We did the procedure under conscious sedation. The femoral artery and veins were percutaneously cannulated. Unfractionated heparin (100 units/kg) was administered after vascular access was obtained. A 5 French (Fr) multipurpose catheter with 0.035″Terumo wire was introduced from femoral vein, inferior vena cava, right atrium, right ventricle, main pulmonary artery to RPA. Right pulmonary artery angiography performed in anteroposterior view showed a large feeding artery from distal, prebranching portion of RPA communicating
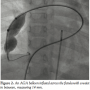
We did the procedure under conscious sedation. The femoral artery and veins were percutaneously cannulated. Unfractionated heparin (100 units/kg) was administered after vascular access was obtained. A 5 French (Fr) multipurpose catheter with 0.035″Terumo wire was introduced from femoral vein, inferior vena cava, right atrium, right ventricle, main pulmonary artery to RPA. Right pulmonary artery angiography performed in anteroposterior view showed a large feeding artery from distal, prebranching portion of RPA communicating 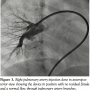 with a large aneurysmal sac of the left atrium (Figure 1, Video 1). The fistula was crossed with the multipurpose catheter and 0.035-inch Terumo wire and this was exchanged with 0.035-inch Amplatz extrastiff exchange length wire that was parked with a good loop in the left atrium. We introduced a 24 mm AGA sizing balloon to occlude the vessel, rule out additional fistula, and to measure the narrowest portion of the feeder as it entered the left atrium (Figure 2, Video 2). The communication was
with a large aneurysmal sac of the left atrium (Figure 1, Video 1). The fistula was crossed with the multipurpose catheter and 0.035-inch Terumo wire and this was exchanged with 0.035-inch Amplatz extrastiff exchange length wire that was parked with a good loop in the left atrium. We introduced a 24 mm AGA sizing balloon to occlude the vessel, rule out additional fistula, and to measure the narrowest portion of the feeder as it entered the left atrium (Figure 2, Video 2). The communication was 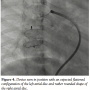 single and the narrow segment measured 14 mm with the proximal portion measuring 26 mm. Considering the morphology of the fistula, we decided to close the defect antegradely. As the fistula was large and roomy we decided to close the feeding vessel with an atrial septal occluder. The left atrial disc would easily be accommodated in the aneurysmal sac and the right atrial disc was expected to configure into the shape of the feeding artery without protruding into the lumen of RPA. The constricted segment was well away from the branches of the RPA as well as the opening of the pulmonary veins into the left atrium. The balloon was removed and an 8 Fr long Cook sheath was introduced antegradely over the Amplatz wire through RPA, across the fistula into the LA. As the constricted segment measured 14 mm, we deployed a 16 mm Cardiofix atrial septal occluder (Starway Medical Corporation) across the narrow portion of the feeding vessel. While the left atrial disc assumed the expected shape, the right atrial disc was rounded due to comparatively less space in the feeding artery. We did RPA injections in multiple views to be sure that there was no obstruction to RPA flow as well as that there is no residual flow across the fistula (Figure 3, Video 3). After checking the stability of the device, it was released (Figure 4, Video 4). The patient’s saturation improved to 100% and there was no residual fistula on follow-up. He was put on oral aspirin for 6 months and is doing well on follow-up without any complications.
single and the narrow segment measured 14 mm with the proximal portion measuring 26 mm. Considering the morphology of the fistula, we decided to close the defect antegradely. As the fistula was large and roomy we decided to close the feeding vessel with an atrial septal occluder. The left atrial disc would easily be accommodated in the aneurysmal sac and the right atrial disc was expected to configure into the shape of the feeding artery without protruding into the lumen of RPA. The constricted segment was well away from the branches of the RPA as well as the opening of the pulmonary veins into the left atrium. The balloon was removed and an 8 Fr long Cook sheath was introduced antegradely over the Amplatz wire through RPA, across the fistula into the LA. As the constricted segment measured 14 mm, we deployed a 16 mm Cardiofix atrial septal occluder (Starway Medical Corporation) across the narrow portion of the feeding vessel. While the left atrial disc assumed the expected shape, the right atrial disc was rounded due to comparatively less space in the feeding artery. We did RPA injections in multiple views to be sure that there was no obstruction to RPA flow as well as that there is no residual flow across the fistula (Figure 3, Video 3). After checking the stability of the device, it was released (Figure 4, Video 4). The patient’s saturation improved to 100% and there was no residual fistula on follow-up. He was put on oral aspirin for 6 months and is doing well on follow-up without any complications.
Discussion. First described by Churton in 1897, pulmonary arteriovenous malformations can be congenital or acquired and represent a fistulous connection between the pulmonary arterial system and pulmonary venous system in the form of dilated vascular trunks, thin aneurysmal sacs, or a tangle of distended tortuous vessels. They can be solitary or multiple, unilateral or bilateral, or minute and diffuse throughout both lungs.1,2 Direct connection of the RPA to left atrium is a rare congenital variant first reported in 1950 by Freidlich et al. Central cyanosis with clubbing of fingers and toes, polycythemia, exertional dyspnea, and decreased arterial oxygen saturation usually accompany the lesion but the patient is apparently normal on cardiovascular examination. The second heart sound in particular is normal. Often there is no murmur and the diagnosis is delayed. The murmur is often audible when there is accompanying pulmonary arterial hypertension. Complications are cerebral and systemic emboli, cerebral abscesses, and rupture of the fistula.3,4 This patient also had a long interval from the onset of symptoms to diagnosis. Our case highlights the awareness and the gratifying results of intervention.
There have been 6 reported cases of transcatheter closure of RPA to LA fistula. While a Gianturco coil was used to close the communication in a sick neonate5 and an atrial septal occluder was used in an adult,6 duct occluders have been used in all other cases.7-10 All the cases used a standard venovenous loop formation for the procedure. We used an atrial septal occluder because the communication was relatively large and could accommodate the larger discs of the atrial septal occluder. Our case is unique because the communication was closed in an antegrade fashion without transseptal puncture and conventional venovenous loop because the feeder vessel was easily accessible antegradely and this simplified the whole procedure.
References
- Perloff JK. The Clinical Recognition of Congenital Heart Disease. 5th ed. Philadelphia, Pa: WB Saunders; 2003;561-571.
- Grifka RG, Preminger TJ. Vascular Anomalies. In: Allen HD, Driscoll DJ, Shaddy RE, Feltes TF, eds. Moss and Adams’ Heart Disease in Infants, Children and Adolescents; Including the Fetus and Young Adult. 7th ed. Philadelphia, Pa: Lippincott Williams & Wilkins; 2007:720-725.
- Lucas RV Jr, Lund GW, Edwards JE. Direct communication of a pulmonary artery with the left atrium: an unusual variant of pulmonary arteriovenous fistula. Circulation. 1961;24:1409-1414.
- Verel D, Grainger RG, Taylor DG. Direct communication of a pulmonary artery with the left atrium. Heart. 1964;26;856-858.
- Slack MC, Jedeikin R, Jones JS. Transcatheter coil closure of a right pulmonary artery to left atrial fistula in an ill neonate. Catheter Cardiovasc Interv. 2000;50(3):330-333.
- Uthaman B, Al-Qbandi M, Abushaban L, Rathinasamy J. Transcatheter closure of large pulmonary arteriovenous fistula including pulmonary artery to left atrial fistula with Amplatzer septal occluder. Catheter Cardiovasc Interv. 2007;70(3):422-428.
- Duke C, Alwi M. Transcatheter closure of direct communication between right pulmonary artery and left atrium using Amplatzer device. Heart. 2003;89(10):1210.
- Zanchetta M, Rigatelli G, Pedon L, Zennaro M, Maiolino P, Onorato E. Transcatheter Amplatzer duct occluder closure of direct right pulmonary to left atrium communication. Catheter Cardiovasc Interv. 2003;58(1):107-110.
- Francis E, Sivakumar K, Kumar RK. Transcatheter closure of fistula between the right pulmonary artery and left atrium using the Amplatzer duct occluder. Catheter Cardiovasc Interv. 2004;63(1):83-86.
- Ramakrishnan S, Shivdas S, Kothari SS. Trans-septal closure of a right pulmonary artery to left atrial communication. Catheter Cardiovasc Interv. 2010;75(7):1096-1099.
____________________________________________________
From the Department of Pediatric Cardiology, Apollo Health City, Jubilee Hills, Hyderabad, India.
Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. The authors report no conflicts of interest regarding the content herein.
Manuscript submitted January 20, 2012 and accepted February 7, 2012.
Address for correspondence: Dr. Anurakti Srivastava, Department of Pediatric Cardiology, Apollo Health City, Jubilee Hills, Hyderabad, India. Email: anurakti07@yahoo.co.in
















