Pooled Analysis of the CONFIRM Registries: Outcomes in Renal Disease Patients Treated for Peripheral Arterial Disease Using Orbital Atherectomy
Abstract: Background. Patients with renal disease typically have severely calcified peripheral arterial disease. As a result, this population may have worse clinical outcomes following endovascular intervention compared to patients without renal insufficiency. Clinical trials typically exclude this patient population. Methods. Analysis of the CONFIRM I-III registries revealed 1105 patients with renal disease (1777 lesions) and 1969 patients without renal disease (2907 lesions) who underwent orbital atherectomy. This subanalysis compared the composite procedural complication rate including dissection, perforation, slow flow, vessel closure, spasm, embolism, and thrombus formation in patients with and without renal disease. Results. Patients with renal disease had a higher prevalence of diabetes (P<.001), hypertension (P<.001), hyperlipidemia (P<.001), and coronary artery disease (P<.001), Rutherford 5 or 6 lesions (P<.001), as well as more lesions treated (P<.001), more vessels treated (P<.001), and more below-the-knee lesions (P<.001). The renal disease and non-renal disease groups had similar composite procedural complication rates (21.3% vs 22.4%; P=.46), dissection (11.1% vs 11.5%; P=.83), perforation (0.6% vs 0.8%; P=.55), slow flow (5.0% vs 4.2%; P=.19), spasm (6.7% vs 6.2%; P=.40), embolism (1.7% vs 2.6%; P=.12), and thrombus formation (1.4% vs 1.0%; P=.56). The renal disease group had a trend toward decreased vessel closure (1.1% vs 1.6%; P=.08). Conclusion. Plaque modification with orbital atherectomy resulted in similar low rates of procedural complications in the renal disease group compared with the non-renal disease group despite more unfavorable baseline clinical and lesion characteristics in the renal disease group.
J INVASIVE CARDIOL 2014;26(8):350-354
Key words: atherectomy, peripheral vascular disease, peripheral interventions
_______________________________________
End-stage renal disease (ESRD) is a powerful predictor of atherosclerotic vascular disease, cardiovascular morbidity, and mortality.1 The lack of arterial remodeling from uremia results in endothelial dysfunction and decreased arterial distensibility, which predisposes patients to peripheral arterial disease (PAD).2 According to an international observational study of 29,873 hemodialysis (HD) patients, correlates of PAD included age, male sex, diabetes, hypertension, and duration of HD.3 More importantly, the diagnosis of PAD was associated with increased all-cause mortality (hazard ratio: 1.36), cardiac mortality (hazard ratio: 1.43), and all-cause hospitalization (hazard ratio: 1.19). The prevalence of PAD in ESRD patients was reported by the National Institutes of Health as approximately 15%, indicating high disease burden.4
Renal disease significantly predicts adverse outcomes with coronary percutaneous intervention and doubles the risk of death in acute myocardial infarction patients.5,6 Peripheral interventions in patients with ESRD have shown to be feasible, but are often associated with complications.7 The presence of ESRD also predicts early graft failure as well as repeat revascularization in both aortofemoral and infrapopliteal lesions.8-11
The Orbital Atherectomy System (OAS) (Cardiovascular Systems, Inc) is a minimally invasive atherectomy system developed for the treatment of PAD with high atherosclerotic burden.12,13 There are currently no data comparing outcomes of patients with and without kidney disease for the treatment of PAD with the OAS. We sought to compare outcomes between these two groups in the CONFIRM prospective registry of patients treated with OAS.14
Methods
Design. The CONFIRM I, II, and III registries are multi-center, non-randomized, all-comer registries of all patients with PAD who were treated with the OAS in the United States.14 There were no inclusion or exclusion criteria for enrollment. In the CONFIRM registry series, the principal interventionalist recorded that either the patient had or had not been previously diagnosed with renal disease. The patients enrolled in the CONFIRM series were divided into 1105 patients with renal disease (1777 lesions) and 1969 patients without renal disease (2907 lesions) who underwent orbital atherectomy.
Device description. All patients were treated with the OAS manufactured by CSI in St. Paul, Minnesota. Three device iterations were evaluated over the CONFIRM series: CONFIRM I evaluated the Diamondback360° exclusively; CONFIRM II evaluated the Predator360° exclusively; and CONFIRM III evaluated all devices: Diamondback360°, Predator360°, and Stealth360°.
Statistical analysis. Data regarding patient characteristics are reported as frequency counts and percentages. Percentages are computed using available data only, with missing values excluded from analyses. Means and standard deviations are reported for quantitative measurements, whereas minimum and maximum values are reported to indicate data ranges. Relationships between various patient or lesion characteristics and patient outcomes were analyzed by cross-tab analysis. Rarely observed data categories were combined (eg, pretreatment stenosis percentages of less than 70% were collapsed into a single category of stenosis ≤70%) as necessary to allow sufficient counts in cross-tab cells for valid Chi-square analysis. Chi-squared (χ2) values are reported. P-values <.05 are considered statistically significant. Statistical analyses were done using SAS version 9.3.
Results
Baseline clinical and procedural characteristics. The demographics for patients in the study are provided in Table 1. Patients with and without renal disease were similar in age (71.5 ± 10.5 years vs 71.7 ± 10.7 years; P=.41),  with a similar percentage of females in each group (39.7% vs 41.0%; P=.61). The renal disease group had a statistically higher rate of diabetes (73.9% vs 51.6%; P<.001), coronary artery disease (76.2% vs 60.3%; P<.001), hypertension (94.6% vs 90.0%; P<.001), and hyperlipidemia (86.4% vs 77.6%; P<.001). There was a statistically higher rate of current smokers in the no renal disease group (30.6% vs 23.7%; P<.001). At baseline, the renal disease group had more patients classified as Rutherford class 5 and 6 (P<.001) (Table 1).
with a similar percentage of females in each group (39.7% vs 41.0%; P=.61). The renal disease group had a statistically higher rate of diabetes (73.9% vs 51.6%; P<.001), coronary artery disease (76.2% vs 60.3%; P<.001), hypertension (94.6% vs 90.0%; P<.001), and hyperlipidemia (86.4% vs 77.6%; P<.001). There was a statistically higher rate of current smokers in the no renal disease group (30.6% vs 23.7%; P<.001). At baseline, the renal disease group had more patients classified as Rutherford class 5 and 6 (P<.001) (Table 1).
Lesion characteristics are given in Table 2. Preprocedural and postprocedural percent stenosis, final residual stenosis, total run time, maximum inflation pressure, and total inflation time were similar between the two groups (Table 3).
The group with renal disease had a higher likelihood of having a lesion treated below the knee (39.8% vs 34.1% P<.001). Procedural data are available in Table 3. Adjunctive therapies used are shown in Table 4. Compared to 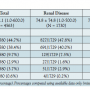 patients with renal disease, patients without renal disease were more likely to have a stent placed (4.5% vs 6.4%; P=.01).
patients with renal disease, patients without renal disease were more likely to have a stent placed (4.5% vs 6.4%; P=.01).
Procedural complications. The composite procedural complication rate including dissection, perforation, slow flow, vessel closure, spasm, embolism, and thrombus formation was similar in patients with and without renal disease (21.3% vs 22.4%; P=.46).
When procedural complications were evaluated, there was no significant difference between the renal disease group vs the no renal disease group in the rate of dissection (11.1% vs 11.5%; P=.83), perforation (0.6% vs 0.8%; P=.55), slow flow (5.0% vs 4.2%; P=.19), vessel closure (1.1% vs 1.6%; P=.08), spasm (6.7% vs 6.2%; P=.40), embolism (1.7% vs 2.6%; P=.12), and thrombus formation (1.4% vs 1.0%; P=.56) (Table 5).
Discussion
In this study comparing patients with and without renal disease treated for PAD with the OAS, we found no statistical difference in acute procedural complication rates, despite more unfavorable baseline and lesion 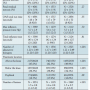 characteristics in the renal disease group.
characteristics in the renal disease group.
Renal disease is more prevalent in PAD patients, and is responsible for significant morbidity and mortality in this patient population.3 The pathophysiology behind endothelial dysfunction in renal disease is not well understood. One theory is based on animal and pathologic studies implicating impairment of the nitric oxide function with chronic uremia.2,15,16 The presence of renal disease predicts poor outcomes in multiple modalities of peripheral interventions across multiple observational studies.9-11
There is a strong correlation between ESRD and arterial calcification, which likely adds to the fibrocalcific burden in PAD.17 The presence of heavy calcium burden is associated with periprocedural complications such as dissections and perforations, which make PAD interventions more challenging.18 However, the OASIS multicenter prospective registry demonstrated a high procedural success rate (90.1% patients had final diameter stenosis 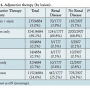 <30%) and a low rate of major adverse events at 6 months (10.4%).19 Eighty-five percent of the patients treated were below the knee and 55% were classified as calcified vessels. CALCIUM 360° was a randomized multicenter trial that compared balloon angioplasty alone versus balloon angioplasty with OAS for treatment of calcified lesions located below the knee.20 Periprocedural success was higher in the OAS group (93.1% vs 82.4%; P=.27), bailout stenting was required with less frequency (6.9% vs 14.3%; P=.14), and both all-cause mortality at 12 months (0% vs 31.6%; P=.01) and the major adverse event rate at 12 months (6.7% vs 42.1%; P=.01) were lower in the OAS group.
<30%) and a low rate of major adverse events at 6 months (10.4%).19 Eighty-five percent of the patients treated were below the knee and 55% were classified as calcified vessels. CALCIUM 360° was a randomized multicenter trial that compared balloon angioplasty alone versus balloon angioplasty with OAS for treatment of calcified lesions located below the knee.20 Periprocedural success was higher in the OAS group (93.1% vs 82.4%; P=.27), bailout stenting was required with less frequency (6.9% vs 14.3%; P=.14), and both all-cause mortality at 12 months (0% vs 31.6%; P=.01) and the major adverse event rate at 12 months (6.7% vs 42.1%; P=.01) were lower in the OAS group.
The CONFIRM registry series is the largest all-comer database of PAD patients, and this is the only study evaluating the differential outcomes in patients with renal disease.14 PAD and renal disease impart significant burden on patients and on the healthcare system, and deserve attention to optimize management and therapeutic options. Chronic kidney dysfunction leads to progressive atherosclerotic disease and calcific burden, and the OAS appears to be feasible and safe in this patient population. In the CONFIRM registry, the renal disease group had a significantly higher comorbidity baseline profile, which is typically not conducive to procedural success and safety;  however, we found that both groups had similar acute procedural and safety outcomes.
however, we found that both groups had similar acute procedural and safety outcomes.
Study limitations. This was an all-comer registry of patients undergoing PAD with the use of the OAS and is exposed to investigator and selection bias. Because of the non-randomized nature, there were differences in demographics and lesion characteristics, which would alter the clinical outcomes. There was no blinding in the CONFIRM registry series, which may also lead to observational bias. The CONFIRM registry series did not include a placebo or comparison group, and was not meant to compare two different treatment modalities. In addition, the CONFIRM series did not differentiate between critical limb ischemic patients and chronic arterial occlusive disease patients, although significant differences may exist between these patient types.
Conclusion
Chronic renal disease begets endothelial dysfunction and calcification, and this group of patients may benefit from orbital atherectomy adjunctive therapy for treatment of PAD. A review of all patients treated with the OAS for PAD in the CONFIRM registry series revealed that both renal disease patients and non-renal disease patients fared well in acute procedural outcomes. However, given the lack of randomized data, further studies are needed to support the use of adjunctive device therapy, such as orbital atherectomy, in the treatment of PAD patients with and without renal disease.
Acknowledgments. The authors thank the CONFIRM Investigators. They would also like to acknowledge the contributions of Brad J. Martinsen, PhD, and Ann Behrens, BS, of Cardiovascular Systems, Inc, with editing and critical review of this manuscript.
References
- Port FK. Morbidity and mortality in dialysis patients. Kidney Int. 1994;46(6):1728-1737.
- Luksha N, Luksha L, Carrero JJ, Hammarqvist F, Stenvinkel P, Kublickiene K. Impaired resistance artery function in patients with end-stage renal disease. Clin Sci (Lond). 2011;120(12):525-536.
- Rajagopalan S, Dellegrottaglie S, Furniss AL, et al. Peripheral arterial disease in patients with end-stage renal disease: observations from the Dialysis Outcomes and Practice Patterns Study (DOPPS). Circulation. 2006;114(18):1914-1922.
- O’Hare A, Johansen K. Lower-extremity peripheral arterial disease among patients with end-stage renal disease. J Am Soc Nephrol. 2001;12(12):2838-2847.
- Parikh PB, Jeremias A, Naidu SS, et al. Impact of severity of renal dysfunction on determinants of in-hospital mortality among patients undergoing percutaneous coronary intervention. Catheter Cardiovasc Interv. 2012;80(3):352-357.
- Jeremias A, Gruberg L, Patel J, Connors G, Brown DL. Effect of peripheral arterial disease on in-hospital outcomes after primary percutaneous coronary intervention for acute myocardial infarction. Am J Cardiol. 2010;105(9):1268-1271.
- Graziani L, Silvestro A, Bertone V, et al. Percutaneous transluminal angioplasty is feasible and effective in patients on chronic dialysis with severe peripheral artery disease. Nephrol Dial Transplant. 2007;22(4):1144-1149.
- Kawarada O, Fujihara M, Higashimori A, Yokoi Y, Honda Y, Fitzgerald PJ. Predictors of adverse clinical outcomes after successful infrapopliteal intervention. Catheter Cardiovasc Interv. 2012;80(5):861-871.
- Kakkos SK, Haurani MJ, Shepard AD, et al. Patterns and outcomes of aortofemoral bypass grafting in the era of endovascular interventions. Eur J Vasc Endovasc Surg. 2011;42(5):658-666.
- Meltzer AJ, Graham A, Connolly PH, et al. Risk factors for early failure after peripheral endovascular intervention: application of a reliability engineering approach. Ann Vasc Surg. 2013;27(1):53-61.
- Abbas AE, Goodman LM, Timmis R, Boura J. Predictors of poor outcome in female patients undergoing endovascular intervention. J Interv Cardiol. 2010;23(4):401-410.
- Adams GL, Khanna PK, Staniloae CS, Abraham JP, Sparrow EM. Optimal techniques with the Diamondback 360° System achieve effective results for the treatment of peripheral arterial disease. J Cardiovasc Transl Res. 2011;4(2):220-229.
- Heuser RR. Treatment of lower extremity vascular disease: the Diamondback 360° Orbital Atherectomy System. Expert Rev Med Devices. 2008;5(3):279-286.
- Das T, Mustapha J, Indes J, et al. Technique optimization of orbital atherectomy in calcified peripheral lesions of the lower extremities: the CONFIRM series, a prospective multicenter registry. Catheter Cardiovasc Interv. 2014;83(1):115-122.
- Morris ST, McMurray JJ, Spiers A, Jardine AG. Impaired endothelial function in isolated human uremic resistance arteries. Kidney Int. 2001;60(3):1077-1082.
- Bagi Z, Hamar P, Antus B, Rosivall L, Koller A. Chronic renal failure leads to reduced flow-dependent dilation in isolated rat skeletal muscle arterioles due to lack of NO mediation. Kidney Blood Press Res. 2003;26(1):19-26.
- Jug B, Kadakia J, Gupta M, et al. Coronary calcifications and plaque characteristics in patients with end-stage renal disease: a computed tomographic study. Coron Artery Dis. 2013;24(6):501-508.
- Fitzgerald PJ, Ports TA, Yock PG. Contribution of localized calcium deposits to dissection after angioplasty. An observational study using intravascular ultrasound. Circulation. 1992;86(1):64-70.
- Safian RD, Niazi K, Runyon JP, et al; for the OASIS Investigators. Orbital atherectomy for infrapopliteal disease: device concept and outcome data for the OASIS trial. Catheter Cardiovasc Interv. 2009;73(3):406-412.
- Shammas NW, Lam R, Mustapha J, et al. Comparison of orbital atherectomy plus balloon angioplasty vs. balloon angioplasty alone in patients with critical limb ischemia: results of the CALCIUM 360 randomized pilot trial. J Endovasc Ther. 2012;19(4):480-488.
______________________________________________
From 1UCLA Medical Center, Los Angeles, California; 2Henry Ford Hospital, Detroit, Michigan; 3University of North Carolina and Rex Health Care, Raleigh, North Carolina; 4Metro Heart and Vascular, Wyoming, Michigan; and 5Cardiology and Interventional Vascular Associates, Dallas, Texas.
Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. Dr Lee, Dr Das, Dr Adams, and Dr Mustapha report consultancies with Cardiovascular Systems, Inc. Dr Yang reports no relevant financial interest.
Manuscript submitted December 16, 2013, provisional acceptance given February 3 2014, final version accepted February 7, 2014.
Address for correspondence: Michael S. Lee, MD, Assistant Clinical Professor of Medicine, UCLA Medical Center, Los Angeles, CA 90095. Email: mslee@mednet.ucla.edu or michaelsblee@gmail.com














