ADVERTISEMENT
Inapparent Atrial Flutter Due to Atrial Dissociation
ABSTRACT: Percutaneous and surgical procedures intended to potentially cure atrial fibrillation require creating lines of conduction block in specific locations throughout the atrial chambers. In patients presenting with recurrent atrial fibrillation, repeat procedures are often performed, resulting in more extensive regions of conduction block and the potential for regions of dissociated atrial rhythms.1,2 The present case describes a patient post-multiple ablation procedures who presented with a symptomatic atrial arrhythmia, the mechanism of which was hidden by the presence of extensive atrial dissociation. Electrophysiologic study revealed the appropriate mechanism and a beneficial ablative procedure was then successfully accomplished.
J INVASIVE CARDIOL 2012;24(7):E135-E138
______________________________________________
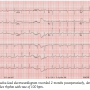 Case Report. A 68-year-old man with history of hypertension and rheumatoid arthritis was admitted for electrophysiologic evaluation. The patient had a prior history of resistant, symptomatic atrial fibrillation with 2 prior pulmonary vein isolation catheter ablation procedures performed at an outside institution. Because of recurrent, persistent atrial fibrillation and disabling symptoms despite medical therapy, he underwent a more recent surgical cryoablation procedure utilizing the Cox Maze III lesion set. He had no history of coronary artery disease and
Case Report. A 68-year-old man with history of hypertension and rheumatoid arthritis was admitted for electrophysiologic evaluation. The patient had a prior history of resistant, symptomatic atrial fibrillation with 2 prior pulmonary vein isolation catheter ablation procedures performed at an outside institution. Because of recurrent, persistent atrial fibrillation and disabling symptoms despite medical therapy, he underwent a more recent surgical cryoablation procedure utilizing the Cox Maze III lesion set. He had no history of coronary artery disease and 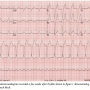 echocardiographic study demonstrated normal left ventricular function and mild left atrial enlargement. Post-surgery ECG demonstrated sinus rhythm with low amplitude P waves evident. Two months later the patient developed episodic palpitations that progressed over the next several weeks. ECGs demonstrated both a narrow-complex rhythm with a rate of 100 bpm (Figure 1) and a wide-complex rhythm with minimally faster rate demonstrating left bundle branch morphology (Figure 2) with the atrial mechanism not clearly evident.
echocardiographic study demonstrated normal left ventricular function and mild left atrial enlargement. Post-surgery ECG demonstrated sinus rhythm with low amplitude P waves evident. Two months later the patient developed episodic palpitations that progressed over the next several weeks. ECGs demonstrated both a narrow-complex rhythm with a rate of 100 bpm (Figure 1) and a wide-complex rhythm with minimally faster rate demonstrating left bundle branch morphology (Figure 2) with the atrial mechanism not clearly evident.
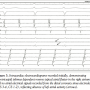 Diagnostic Evaluation. The patient was admitted for electrophysiologic study with multipolar catheters placed near the His bundle, in the coronary sinus, and around the tricuspid annulus. Analysis of the recordings demonstrated evidence of cavotricuspid isthmus (CTI)-dependent reverse typical atrial flutter rotating in the right atrium in a clockwise, caudocranial direction (Figure 3). However, coronary sinus recordings from electrodes reflecting left atrial activity (CS 3-4, CS 1-2) were electrically silent, compatible with atrial dissociation (Figure 3).
Diagnostic Evaluation. The patient was admitted for electrophysiologic study with multipolar catheters placed near the His bundle, in the coronary sinus, and around the tricuspid annulus. Analysis of the recordings demonstrated evidence of cavotricuspid isthmus (CTI)-dependent reverse typical atrial flutter rotating in the right atrium in a clockwise, caudocranial direction (Figure 3). However, coronary sinus recordings from electrodes reflecting left atrial activity (CS 3-4, CS 1-2) were electrically silent, compatible with atrial dissociation (Figure 3).  Presumably this finding was a result of the extensive ablation procedures the patient had undergone, creating interatrial conduction block, which resulted in the absence of electrical activity in the left atrium. This explained the presence of low amplitude atrial activity evident in the surface recordings despite the presence of sinus rhythm and then reverse typical atrial flutter, occurring in only the right atrial chamber. The 2 non-sinus rhythm ECGs were manifestations of atrial flutter with 2:1 AV conduction with and without bundle branch block with the flutter rate slowed by prior ablations and amiodarone therapy. Neither a sawtooth nor a sinusoidal ECG pattern was present, but careful scrutiny of lead II in both tracings did reveal low amplitude atrial activity compatible with this diagnosis. Radiofrequency ablation of the CTI resulted in bidirectional conduction block and restoration of sinus rhythm. Despite conversion, the left atrium recordings remained electrically silent (CS 3-4, CS 1-2) (Figure 4). The patient was discharged in stable condition and remained asymptomatic. Ultrasonography performed 3 months post-CTI ablation with pulsed Doppler examination of the mitral valve inflow demonstrated the absence of an “a” wave during diastolic filling despite continued maintenance of sinus rhythm compatible with atrial dissociation and absence of electromechanical activity in the left atrium. Anticoagulation therapy was continued indefinitely due to this finding.
Presumably this finding was a result of the extensive ablation procedures the patient had undergone, creating interatrial conduction block, which resulted in the absence of electrical activity in the left atrium. This explained the presence of low amplitude atrial activity evident in the surface recordings despite the presence of sinus rhythm and then reverse typical atrial flutter, occurring in only the right atrial chamber. The 2 non-sinus rhythm ECGs were manifestations of atrial flutter with 2:1 AV conduction with and without bundle branch block with the flutter rate slowed by prior ablations and amiodarone therapy. Neither a sawtooth nor a sinusoidal ECG pattern was present, but careful scrutiny of lead II in both tracings did reveal low amplitude atrial activity compatible with this diagnosis. Radiofrequency ablation of the CTI resulted in bidirectional conduction block and restoration of sinus rhythm. Despite conversion, the left atrium recordings remained electrically silent (CS 3-4, CS 1-2) (Figure 4). The patient was discharged in stable condition and remained asymptomatic. Ultrasonography performed 3 months post-CTI ablation with pulsed Doppler examination of the mitral valve inflow demonstrated the absence of an “a” wave during diastolic filling despite continued maintenance of sinus rhythm compatible with atrial dissociation and absence of electromechanical activity in the left atrium. Anticoagulation therapy was continued indefinitely due to this finding.
 Discussion. The presence of atrial dissociation has been described in the experimental and electrocardiographic literature for over 80 years.3,4 This phenomenon has generally been identified by documenting ECG evidence of 2 distinct P wave morphologies depolarizing independently of each other. The postulated mechanisms are described as either interatrial or intra-atrial block, with the original descriptions primarily citing this condition in critically ill patients with decompensated heart failure.
Discussion. The presence of atrial dissociation has been described in the experimental and electrocardiographic literature for over 80 years.3,4 This phenomenon has generally been identified by documenting ECG evidence of 2 distinct P wave morphologies depolarizing independently of each other. The postulated mechanisms are described as either interatrial or intra-atrial block, with the original descriptions primarily citing this condition in critically ill patients with decompensated heart failure.
With the advent of invasive cardiac electrophysiology, more detailed mechanistic analyses described a variety of independent atrial rhythms occurring simultaneously in different locations in the atrial chambers and coronary sinus, including sinus rhythm, focal atrial tachycardia, atrial flutter, and atrial fibrillation.5 In 1975, Wu et al described electrophysiologic documentation of 2 patients with dissimilar atrial rhythms occurring in both atria. One of their conclusions was that, at times, the surface ECG does not always accurately reflect the atrial rhythm, a finding observed in our patient.6 A later report by Friedman et al described 2 patients with atrial flutter who manifested atrial dissociation during electrophysiologic study.7 The first patient presented with atrial flutter initially, but after rapid atrial pacing developed AF localized to the mid and low right atrium while flutter continued in other regions of the left and right atria. The second patient demonstrated left atrial flutter with evidence of 3:2 Wenkebach conduction to the right atrium.
More recently, occurrences of atrial dissociation have been observed in patients who have undergone percutaneous and surgical procedures designed to potentially cure atrial fibrillation, including surgical maze and catheter ablation approaches. Multiple reports described dissociated atrial rhythms in patients with prior surgical maze procedures for AF.8,9 Most commonly, the 2 dissociated rhythms are sinus rhythm present in the majority of atrial locations with an isolated atrial region demonstrating AF. Atrial arrhythmias including isolated AF may not always be recognized in the surface tracing similar to our case, but still pose potential risks. Li et al described a young man post-atrial fibrillation surgery who presented with a cerebrovascular accident due to dissociated AF despite the surface ECG showing normal sinus rhythm.6 Similarly, dissociated atrial rhythms have been documented in patients with history of extensive catheter ablations with segmental rhythms evident in the right and left atria and coronary sinus.1,2 Presumably, these AF procedures create extensive lines of conduction block that isolate the various atrial rhythms from one another.
The presented case was similar to these recent reports in that the patient had previously undergone extensive ablative and surgical procedures in an attempt to cure AF. Low amplitude P waves were present in the discharge ECG and then with the advent of the patient’s rhythm disturbance, atrial activity was not clearly apparent in the surface tracings. The electrophysiologic and echocardiographic data suggest that complete biatrial dissociation was present as a result of these procedures. Considering the anatomic relationships of the atria, the surface ECG in general reflects more left atrial activity than right and when the left atrium is electrically silent, ECG evidence of atrial activity is blunted. A subsequent invasive study documented the presence of CTI-dependent reverse typical atrial flutter localized only to the right atrium and this rhythm was treated with standard ablative approach. Intracardiac recordings reflecting left atrium remained electrically silent, which persisted late post-procedure, as no mechanical atrial function was evident 3 months post-procedure.
The potential for developing this phenomenon certainly must parallel the number of procedures and ablative lines that a patient undergoes in an attempt to improve AF. A resistant, highly symptomatic patient undergoing this number of AF procedures is somewhat atypical in our experience, but considering that AF ablation is somewhat imperfect in terms of outcomes in its present practice, increasing numbers of patients undergoing multiple procedures can be anticipated. Considering this, postablation patients, particularly those who have undergone multiple, extensive procedures, should be monitored for the occurrence of unusual rhythms and consideration given to the diagnosis of atrial dissociation when low-amplitude atrial activity is identified on the surface ECG. Diagnosis may be difficult without invasive electrophysiologic study although ultrasonography may reveal clues in suspicious patients by demonstrating evidence of electromechanical dissociation.10 Similarly, improvement in these echo-Doppler abnormalities may signal resolution of the dissociated rhythms and improved atrial electrical conduction.
Absence of mechanical left atrial function was previously described in patients post-conversion from AF, and in many patients, these stunned atria exhibit gradual recovery of functional activity over time.11 Mantana et al described echocardiographically normal right atrial mechanical function and absent left atrial function 7 months post-conversion to sinus rhythm.12 However, some of these cases may be dissimilar to ours in that electrical activity may be present but the atrial musculature cannot be stimulated resulting in absent atrial mechanical function. The mechanism of mechanical dysfunction in the presented case is unique in that left atrial electrical activity is presumably inhibited by extensive areas of atrial conduction block that were created by the ablative processes.
Although specific guidance is not offered in the literature with respect to the importance of continued anticoagulation in these patients except for isolated case reports, it seems reasonable that anticoagulation should be continued in high-risk patients when the isolated segment is electrically and mechanically inactive due to atrial dissociation as in our patient. In other patients with distinct atrial arrhythmias in different segments of the atria due to this process, anticoagulation should also be considered, although risk stratification guidance seems reasonable utilizing the CHADS2 or CHA2DS2-VASc scoring systems.
In summary, atrial dissociation should be suspected when unusual rhythms occur in patients who have a history of extensive AF procedures, with resulting regions of conduction block in varying atrial locations. The importance of recognizing these processes is highlighted when one considers the potential for embolic phenomenon originating from either isolated atrial arrhythmias or atrial regions with electromechanical dissociation. Therefore continued anticoagulant therapy should be considered in these vulnerable patients until resolution of this electromechanical disorder is documented. In particular, when the left atrium is electrically silent, the surface ECG may not accurately reflect atrial activity, and both ultrasonography and invasive electrophysiologic study may be useful in elucidating the correct rhythm and guiding appropriate therapy.
References
- Rostock T, Rotter M, Sanders P, et al. Fibrillating areas isolated within the left atrium after radiofrequency linear catheter ablation. J Cardiovasc Electrophysiol. 2006;17(8):807-812.
- Matsuo S, Lim KT, Knecht S, Arantes L, Haïssaguerre M. Dual independent atrial tachycardias after ablation of chronic atrial fibrillation. J Cardiovasc Electrophysiol. 2008;19(9):979-981.
- Chung KY, Walsh TJ, Massie E. A review of atrial dissociation, with illustrative cases and critical discussion. Am J Med Sci. 1965;250:72-78.
- Scott ME, Finnegan OC. Atrial dissociation. Brit Heart J. 1975;37(5):539-542.
- Zipes DP, DeJoseph RL. Dissimilar atrial rhythms in man and dog. Am J Cardiol. 1973;32(5):618-628.
- Wu D, Denes P, Leon FA, Chhablani RC, Rosen KM. Limitations of the surface electrocardiogram in diagnosis of atrial arrhythmias. Further observations on dissimilar atrial rhythms. Am J Cardiol. 1975;36(1):91-97.
- Friedman PL, Brugada P, Kuck KH, et al. Inter- and intraatrial dissociation during spontaneous atrial flutter: evidence for a focal origin of the arrhythmia. Am J Cardiol. 1982;50(4):756-761.
- Li YH, Lo HM, Lin FY, Lin JL, Lien WP. Atrial dissociation after atrial compartment operation for chronic atrial fibrillation in mitral valve disease. Pacing Clin Electrophysiol. 1998;21(4 Pt 1):756-759.
- Matsumoto Y, Watanabe G, Endo M, Sasaki H, Kasashima F. Coexistence of sinus rhythm and atrial fibrillation after maze procedure. Ann Thorac Surg. 2002;74(1):249-251.
- Naqvi TZ, Zaky J. Electric dissociation within the left atrial appendage diagnosed by Doppler echocardiography. J Am Soc Echocardiogr. 2004;17(10):1077-1079.
- Manning WJ, Silverman DI, Katz SE, et al. Impaired left atrial mechanical function after cardioversion: relation to the duration of atrial fibrillation. J Am Coll Cardiol. 1994;23(7):1535-1540.
- Matana A, Zaputović L, Mavrić Z, et al. Permanent left atrial electromechanical dissociation after conversion of atrial fibrillation in a patient with interatrial block. Pacing Clin Electrophysiol. 2004;27(8):1158-1160.
______________________________________________
From the 1Cardiac Electrophysiology Section, LSU School of Medicine, New Orleans, Louisiana; and 2Arrhythmia Service, Cardiovascular Institute of the South, Houma, Louisiana.
Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. The authors report no conflicts of interest regarding the content herein.
Manuscript submitted November 21, 2011, provisional acceptance given February 7, 2012, final version accepted March 13, 2012.
Address for correspondence: Richard Abben, MD, 225 Dunn Street, Houma, Louisiana 70360. Email: abben@msn.com
















