Overweight Nonagenarian With Acute Kidney Injury and Not on Glucose-Lowering Therapy
Affiliations: Division of Geriatrics and Post-Acute; Medicine, Baystate Medical Center/TUFTS University School of Medicine, Springfield, MA
Abstract: Fluoroquinolones are widely used in both inpatient and ambulatory settings. They are known to alter glucose homeostasis and insulin production by affecting pancreatic beta-cell adenosine triphosphate–sensitive potassium channel activity; however, fluoroquinolone-induced hypoglycemia remains underreported, especially in elderly patients not on any glucose-lowering therapy. Subsequently, healthcare providers may remain unaware of this complication, which can have significant clinical implications in terms of morbidity, mortality, and adverse drug reactions. The authors present the case of an overweight nonagenarian with diabetes, chronic kidney disease, and acute kidney injury who, due to worsening renal function, became hypoglycemic as a result of being unable to efficiently clear fluoroquinolones. The patient was not on any glucose-lowering therapy. Following the case report, the authors provide an overview of the fluoroquinolones approved for use in the United States and discuss their effects on blood glucose levels.
Key words: Hypoglycemia, hyperglycemia, diabetes mellitus, fluoroquinolones, kidney disease, glucose-lowering therapy.
___________________________________________________________________________________________________________________________________________________
According to the United States Census Bureau, 13% of the total US population in 2010 was aged 65 years and older.1 The percentage of individuals in this age bracket continues to grow. Challenges in caring for this population include a high prevalence of comorbidities and an increase in the use of prescription drugs. In the 1980s, individuals older than 65 years used more than 30% of all prescribed medications.2 A 2013 report indicates that this number has increased to more than 50%, with those older than 65 years taking anywhere between six and eight prescription medications.3 Not only are elderly individuals taking more medications than younger patients, but they are also more sensitive to medications and susceptible to adverse drug reactions (ADRs) due to changes in body composition, drug receptor dynamics, and hepatic, gastrointestinal, and renal function.4 In addition, as the number of medications increases, so does the risk of ADRs. The reported risk of an ADR is 15% with two medications, 58% with five medications, and 82% with seven or more medications.3
Fluoroquinolones, a class of broad-spectrum antibiotics that are effective against both gram-negative and gram-positive bacteria, are widely prescribed to elderly persons in both inpatient and ambulatory settings. They alter glucose homeostasis and insulin production by affecting pancreatic beta-cell adenosine triphosphate–sensitive potassium (KATP) channel activity. Fluoroquinolone use in patients with underlying comorbidities, such as chronic kidney disease (CKD), can increase the risk of hypo- and hyperglycemia.
We report the case of an overweight nonagenarian with diabetes and acute kidney injury who was not on any glucose-lowering medications but became hypoglycemic due to his worsening renal function, which impaired his ability to efficiently clear fluoroquinolones. As this case shows, even patients not on glucose-lowering therapy are at risk of developing hypoglycemia, especially in the setting of underlying comorbid conditions such as CKD. Following this case report, we review the use of fluoroquinolones in the elderly, focusing on their glycemic effects.
Case Report
A 91-year-old man with atrial fibrillation, moderate to severe dementia, type 2 diabetes mellitus, and CKD was admitted to the hospital due to delirium and acute kidney injury following a fall. The patient was overweight with a body mass index (BMI) of 25.91 kg/m2. His outpatient records revealed a baseline albumin level of 3.3 g/dL (reference range, 3.5-5.0 g/dL) and a baseline creatinine level of 0.9 mg/dL (reference range, 0.6-1.2 mg/dL). He tolerated oral intake with an Ensure-supplemented, nectar-thickened diet, and was not on any glucose-lowering therapy. His relevant medication history included metaprolol XL 25 mg once daily for his atrial fibrillation, but this was not a new prescription, and ciprofloxacin, which had been prescribed 1 week prior to his current presentation by his primary care provider for a urinary tract infection (UTI).
On presentation, the patient was alert, only oriented to person, able to follow simple commands, dehydrated with dry mucosa, neurologically nonfocal, hemodynamically stable, and had a grade 2/6 systolic murmur in the aortic area. Blood work on admission revealed a blood urea nitrogen level of 69 mg/dL (reference range, 8-23 mg/dL) and a creatinine level of 4.9 mg/dL (creatinine clearance [CrCl], 9.4 mL/min [reference range, 75-125 mL/min]). A urinalysis was normal and a computed tomography scan of his head showed small vessel ischemia but no acute pathology. At the time of admission, ciprofloxacin was changed to renally dosed levofloxacin.
The patient seemed to do well with conservative management and intravenous (IV) fluids; however, he became unresponsive and was found to have severe hypoglycemia (blood glucose level, 24 mg/dL [reference range, 70-110 mg/dL]) the day after admission. The patient responded to glucose administration with an amp of 50% dextrose, which contained 25 grams of dextrose.
No other etiology for his unresponsiveness was identified on further cardiopulmonary workup. While evaluating his acute kidney injury, the nephrologist noted that the fluoroquinolone therapy could have been the cause of his hypoglycemia, as the patient received ciprofloxacin as outpatient therapy and was only renally dosed with levofloxacin after admission. The Naranjo Adverse Drug Reaction Probability Scale revealed a probable association (score of 6) for the hypoglycemia being caused by fluoroquinolone administration. Quinolone antibiotics were discontinued, and no further hypoglycemia occurred during the course of his hospitalization. The patient was discharged shortly thereafter.
Discussion
The widespread use of fluoroquinolones is well documented across all sites of care and across the age spectrum.5,6 The US Food and Drug Administration (FDA) reports that in 2011, 23.1 million unique patients received a dispensed prescription for an oral fluoroquinolone product from an outpatient retail pharmacy, and that another 3.8 million unique patients were billed for an injectable fluoroquinolone product administered during their hospitalization.7 Thus, it is likely that these agents are often over prescribed, especially in the older adult population. A possible explanation for this is that conditions such as complex urinary tract infections, community- and hospital-acquired pneumonia, and a variety of other bacterial infections disproportionately affect older adults.
Overview of Available Fluoroquinolones
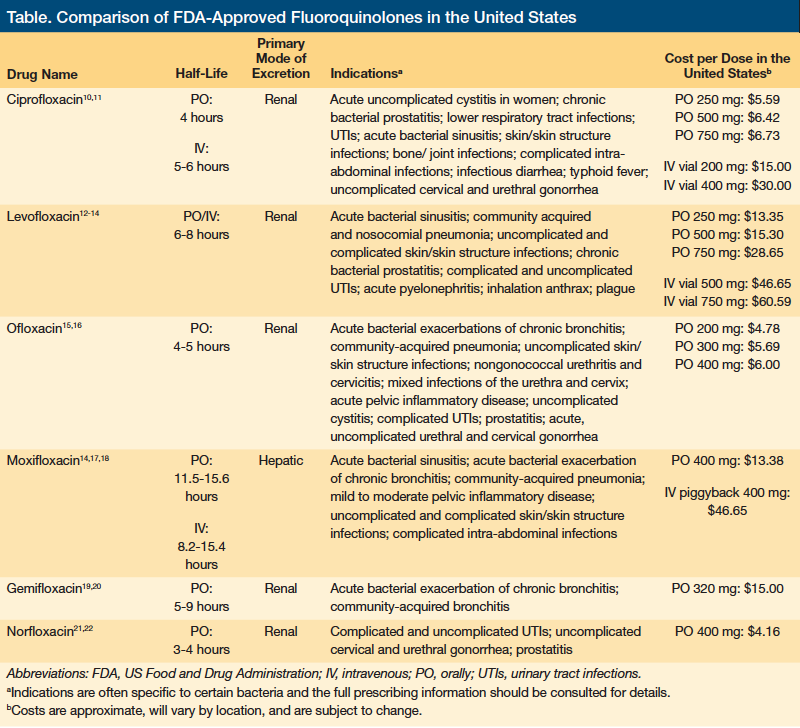
Six fluoroquinolones are approved for use in the United States: ciprofloxacin, levofloxacin, ofloxacin, moxifloxacin, gemifloxacin, and norfloxacin. Of these, ciprofloxacin and levofloxacin, both of which were administered to our patient, are among the most commonly prescribed antibiotics in the United States. An analysis of the Blue Cross Blue Shield of Michigan billing data found ciprofloxacin to be the fourth most commonly prescribed antibiotic for adults between 2007 and 2009. Ciprofloxacin accounted for a staggering 107,295 prescriptions.8 It was also the least expensive of the top 10 antibiotics prescribed during that period, with a treatment regimen costing only $3.66. In comparison, levofloxacin was the eighth most commonly prescribed antibiotic, with 59,525 prescriptions. It was also the most expensive, costing $110.31 per course.8
All fluoroquinolones target bacterial deoxyribonucleic acid (DNA) gyrase and topoisomerase II and IV, which are required for bacterial DNA replication, transcription, repair, and recombination.9,10 Fluoroquinolones primarily target topoisomerase II for gram-positive bacteria and topoisomerase IV for gram-negative bacteria.9 Despite having a similar mechanism of action, the fluoroquinolones vary in terms of their half-life, primary route of elimination, indications, and costs (Table).
• Ciprofloxacin. In patients with normal renal function, ciprofloxacin has an approximate 4-hour serum elimination half-life; 40% to 50% of the orally administered dose is excreted unchanged in urine and 20% to 35% is excreted in feces.10,11 This is prolonged by approximately 20% in those older than 65 years, and clearance is further impaired in the setting of kidney disease or injury.10,11
• Levofloxacin. Levofloxacin has a half-life of 6 to 8 hours.12-14 The half-life in healthy elderly individuals (aged 66-88 years) is closer to 8 hours. Approximately 87% of the drug can be recovered in urine 48 hours after oral administration, while less than 4% is recovered in feces 72 hours after administration. When an individual’s CrCl is less than 50 mL/min, excretion is substantially reduced and plasma elimination half-life is prolonged.12-14
• Ofloxacin. Only generic forms of ofloxacin are available. This agent has a plasma half-life of 4 to 5 hours.15,16 Peak plasma levels occur 0.5 to 1.5 hours after dosing. Approximately 65% to 80% of the administered dose of ofloxacin is excreted in the urine as unchanged drug, with 4% to 8% excreted in the feces. Although the pharmacokinetic properties of ofloxacin do not appear to be affected by advancing age, elderly patients have been found to be more susceptible to developing prolongation of the QTc interval.15,16
• Moxifloxacin. Moxifloxacin is an 8-methoxyquinolone.14,17,18 Fifty-two percent oral or IV moxifloxacin is hepatically metabolized through either glucuronide conjugation (14%) or sulfate conjugation (38%). Neither age nor sex impacts clearance. Furthermore, pharmacologic data indicate that no dose adjustment is needed even in severe renal impairment (CrCl, <30 mL/min).14 Oral moxifloxacin has a half-life ranging from 11.5 hours to 15.6 hours. The IV formulation has a half-life ranging from 8.2 hours to 15.4 hours. Approximately 45% of a dose is excreted unchanged in urine (about 20%) or feces (about 25%).10
• Gemifloxacin. In healthy patients, gemifloxacin and its metabolites have dual routes of excretion, with approximately 61% of the dose excreted in the feces and 36% in the urine as unchanged drug and metabolites.20 The mean plasma elimination half-life at steady state following 320 mg in this population ranged from 4 to 12 hours. The pharmacokinetics of gemifloxacin were not affected by advancing age or sex when bodyweight was accounted for.20
• Norfloxacin. The effective half-life of norfloxacin in serum and plasma is 3 to 4 hours.21,22 Steady-state concentrations of norfloxacin will be attained within 2 days of dosing. In patients with severe renal impairment (CrCl, <30 mL/min), the renal elimination of norfloxacin decreases so that the effective serum half-life is 6.5 hours. Within 24 hours of drug administration, 26% to 32% of the dose is excreted in the urine as unchanged drug, with an additional 5% to 8% being recovered as six active metabolites of lesser antimicrobial potency, whereas 30% is recovered in feces. A pharmacokinetic study of elderly volunteers (aged 65-75 years) with normal renal function for their age showed these patients to be more susceptible to drug-associated effects on the QTc interval.21,22
Adverse Drug Reactions: Focusing on Glucose Homeostasis Abnormalities
Fluoroquinolones have been associated with a variety of ADRs, including skin reactions, musculoskeletal disorders, psychiatric disorders, toxic epidermal necrolysis, Stevens-Johnson syndrome, a prolonged QTc interval, and hyper- and hypoglycemia; however, the only side effects currently warranting a boxed warning by the FDA include the potential for tendinitis, tendon rupture, and permanent peripheral neuropathy.7 These complications can occur in patients of all ages; however, the risk is greater for individuals older than 60 years, those taking corticosteroids, and those with kidney, heart, or lung transplants.7
Although fluoroquinolones are known to alter glucose homeostasis and insulin production, the prevalence of fluoroquinolone-induced hypo- and hyperglycemia is unclear. Documentation of these complications in patients without diabetes and in those with diabetes who are not on any glucose-lowering therapy is sparse. For example, searching PubMedand Ovid using the terms fluoroquinolone, hypoglycemia, and nondiabetic yielded only five published English-language case reports, with fewer than half of these describing this condition in the elderly. Most documentation of quinolone-induced hypoglycemia in patients with diabetes involves individuals on concomitant glucose-lowering therapy.
A case report published in 2000 in the Annals of Emergency Medicine reported a case of resistant hypoglycemia due to the concomitant administration of ciprofloxacin and glyburide.23 A 1993 ciprofloxacin postmarketing surveillance study reported hypoglycemia in 15 of 37,233 patients (0.04%).24 A different phase 4 postmarketing study from 2004 stated that hypoglycemia occurred in 0.1% of patients on levofloxacin for community-acquired pneumonia.25 Wang and Rizvi26 reported a case of levofloxacin-induced hypoglycemia following cardiac bypass graft surgery in a patient without diabetes. A 2005 retrospective study that included 17,108 hospitalized patients determined the rate of dysglycemia to be three times higher in those taking levofloxacin and gatifloxacin versus ceftriaxone.27 A multivariate analysis identified the concomitant use of sulfonylureas and fluoroquinolones as an independent risk factor for the development of hypoglycemia.27
Our literature review further revealed a high prevalence of glucose homeostasis abnormalities associated with gatifloxacin, an agent that was withdrawn from the US market in 2006.28 In 2004, during the 44th Interscience Conference on Antimicrobial Agents and Chemotherapy, Frothingham29 presented data on glucose homeostasis abnormalities from all ADR reports filed with the FDA from 1997 to 2003.Of the 568 reported abnormalities, 80% were associated with gatifloxacin, and 68% of the 25 reported fatalities were linked to gatifloxacin. Yet gatifloxacin accounted for only 6.6% of all fluoroquinolones prescribed.29
In contrast, moxifloxacin seems to be one of the safer quinolones with regard to glucose homeostasis abnormalities, as five postmarketing studies that included 46,130 patients reported no hypoglycemic ADRs and two nondrug-related hyperglycemic episodes.30 In 2012, however, Mandavia and colleagues31 published a case of hypoglycemia in a patient with renal failure but without diabetes who was taking moxifloxacin.
A new fluoroquinolone, finafloxacin, may be on the horizon. It is currently being assessed in a phase II trial for the treatment of complex UTIs and acute pyelonephritis.32 An earlier phase I study in 95 healthy volunteers showed no hypoglycemic reactions.33
Although fluoroquinolones have been observed to cause hypoglycemia and, less commonly, hyperglycemia,34 the exact mechanism causing these effects is still unclear. Studies using gatifloxacin suggest that these agents cause vacuolation of pancreatic beta cells, which may impair insulin secretion and result in hyperglycemia.35 Hypoglycemia has been theorized to be due to the blockage of KATP channels in the beta-cell membranes.36 In addition, ciprofloxacin, unlike levofloxacin and gatifloxacin, inhibits the cytochrome P450 3A4 enzyme that metabolizes most oral hypoglycemic agents.10 Thus, although fluoroquinolones are often thought to be benign medications, they do pose a significant risk for ADRs.
Implications of Adverse Drug Reactions and Impaired Kidney Function in Older Adults
More than 80% of ADRs that occur in the acute care setting or result in a hospital admission are dose-related (type A).37 Type A reactions are predictable, perhaps avoidable, and thought to be more prevalent in the elderly than type B reactions, which are bizarre, unpredictable, and idiosyncratic. In 2004, ADRs ranked among the top four to six leading causes of mortality in the United States, with an ADR resulting in the death of roughly 1 in 300 hospitalized elders.37 Prospective data from a study of 332 nursing home residents reveal that more than 67% of these individuals experienced probable ADRs.38
Underlying comorbidities, such as renal impairment, further increase the risk of type A ADRs. In the United States, CKD prevalence estimates using the continuous National Health and Nutrition Examination Survey (NHANES) dataset from 1999 to 2004 indicate that 13.1% of noninstitutionalized people have stage 1 to 4 CKD, which corresponds to 26.3 million people based on 2000 census estimates.39 When applying 2006 population estimates to the same data set, Snyder and colleagues40 found that prevalence for stage 1 to 4 CKD ranged from 11.7% to 24.9%, a more than twofold difference, resulting in population estimates between 25.8 million and 54.0 million. More recently, when examining NHANES data sets from 1999 to 2004 and from 2005 to 2010 for individuals aged 80 years and older, researchers found that 49.9% and 51.2% of this population, respectively, had an estimated glomerular filtration rate (GFR) of less than 60 mL/min/1.73 m2, whereas 18.6% and 21.7%, respectively, had an estimated GFR of less than 45 mL/min/1.73 m2.41 This points to a rise in prevalence in CKD among people aged 80 years and older. Data from Cooper42 highlight that the CrCl clearance in older adults (>65 years) in long-term care facilities may be assumed to be less than 40 mL/min, even with a serum creatinine of 1 mg/dL to 1.5 mg/dL or less.Given the already high prevalence of renal impairment among the elderly, prescribing renally cleared agents to this population is certainly a high-risk proposition; thus, medication lists must be constantly reviewed to prevent ADRs.
Reviewing Our Patient’s Case
Our case report highlights the importance of recognizing dysglycemia as a fluoroquinolone-related ADR. Based on our patient’s BMI of 25.91 kg/m2 and his intake of a nutritionally supplemented diet, fasting and malnutrition were likely noncontributory. Although beta blockers can stem the epinephrine-mediated release of glucose from hepatic stores, the patient was chronically on metoprolol and was discharged on this without further events. The kidney is an endogenous source of glucose production, especially in fasting states; thus, acute kidney injury can cause hypoglycemia. However, the patient’s renal function took time to normalize, and he experienced no further episodes of hypoglycemia following cessation of fluoroquinolone antibiotics. Therefore, we do not feel that the use metoprolol or his kidney injury alone caused his hypoglycemic event.
Our case is rare in that the great preponderance of published cases mostly report fluoroquinolone-induced hypoglycemia in patients with diabetes on insulin or on oral hypoglycemics. There are few documented cases of hypoglycemia in elderly patients who are not on any glucose-lowering therapy. We suspect that the prevalence in the published literature may underestimate the clinical significance and impact of the problem. This could be due to the fact that some cases are picked up, but not reported. It is also highly likely that many instances of hypoglycemia are ascribed to patients’ concomitant glucose-lowering therapy, malnutrition, or fasting state. Conversely, episodes of hyperglycemia may be attributed to the underlying infection or the physiological stress response to the illness.
Conclusion
When dealing with clinical scenarios necessitating the use of antibiotics in elderly individuals, healthcare providers must take into account patients’ underlying comorbidities and ongoing medical therapy (ie, concomitant use of oral hypoglycemic agents) before prescribing any agent. In particular, many physicians underrecognize the prevalence of renal disease in geriatric patients, which can pose a problem when prescribing agents that are primarily excreted renally. For example, even a creatinine level of 0.9 mg/dL (our patient’s baseline level) can represent renal impairment in a frail elderly person with little muscle mass. If the use of a fluoroquinolone is warranted, such as might be the case when there is a serious systemic infection, moxifloxacin seems to pose the lowest risk of causing dysglycemia. Other drugs, such as finafloxacin, are being investigated in clinical trials and may provide safer options in the future. Further research is undoubtedly needed to delineate the optimal use of fluoroquinolones in the elderly.
References
1. US Census Bureau. 2010 Census Briefs. The older population: 2010. www.census.gov/prod/cen2010/briefs/c2010br-09.pdf. Accessed March 7, 2013.
2. Baum C, Kennedy DL, Knapp DE, Faich A, Annello C. Drug Utilization in the U.S.—1986. Rockville, MD: Department of Health and Human Services, Food and Drug Administration; 1987.
3. Bland CM. Polypharmacy and the elderly. Presented at: 2013 American College of Physicians Georgia Chapter Scientific Meeting; October 4-6, 2013; Savannah, GA. www.acponline.org/about_acp/chapters/ga/13-bland.pdf. Accessed March 18, 2014.
4. Kaysen GA, Myers BD. The aging kidney. Clin Geriatr Med. 1985;1(1):207-222.
5. MacDougall C, Powell JP, Johnson CK, Edmond MB, Polk RE. Hospital and community fluoroquinolone use and resistance in Staphylococcus aureus and Escherichia coli in 17 US hospitals. Clin Infect Dis. 2005;41(4):435-440.
6. Makaryus AN, Byrns K, Makaryus MN, Natarajan U, Singer C, Goldner B. Effect of ciprofloxacin and levofloxacin on the QT interval: is this a significant “clinical” event? South Med J. 2006;99(1):52-56.
7. US Food and Drug Administration Drug Safety Communications. FDA requires label changes to warn of risk for possibly permanent nerve damage from antibacterial fluoroquinolone drugs taken by mouth or by injection. www.fda.gov/downloads/Drugs/DrugSafety/UCM365078.pdf. Released August 15, 2013. Accessed March 18, 2014.
8. Center for Healthcare Research & Transformation. Antibiotic prescribing and use. Accessed March 18, 2014.
9. Mehta A. Mechanism of action of quinolones and fluoroquinolones. https://pharmaxchange.info/press/2011/05/mechanism-of-action-of-quinolones-and-fluoroquinolones. Published May 10, 2011. Accessed March 18, 2014.
10. Bayer HealthCare Pharmaceuticals Inc. CIPRO® (ciprofloxacin hydrochloride) tablets. www.pdr.net/drug-summary/cipro-oral-suspension-and-tablets?druglabelid=2273&id=203. Published 2008. Accessed March 18, 2014.
11. Pham PA, Bartlett JG. Ciprofloxacin. Johns Hopkins Medicine. www.hopkinsguides.com/hopkins/ub/view/Johns_Hopkins_ABX_Guide/540128/all/Ciprofloxacin. Updated August 28, 2012. Accessed March 8, 2013.
12. Physicians Desk Reference. Levaquin. Montvale: Thompson Reuters; 2009. www.pdr.net/drug-summary/levaquin?druglabelid=271&id=993. Accessed March 8, 2014.
13. Pham PA, Bartlett JG. Levofloxacin. Johns Hopkins Medicine. www.hopkinsguides.com/hopkins/ub/view/Johns_Hopkins_ABX_Guide/540312/all/Levofloxacin. Updated August 24, 2012. Accessed March 8, 2013.
14. Rodvold KA, Neuhauser M. Pharmacokinetics and pharmacodynamics of fluoroquinolones. Pharmacotherapy. 2001;21(10 Pt 2):233S-252S.
15. Floxin tablets [prescribing information]. Raritan, NJ: Ortho-McNeil-Janssen Pharmaceuticals, Inc; 2008. Accessed March 18, 2014.
16. Bartlett JG, Auwaerter PG, Pham PA. Ofloxacin. In: Johns Hopkins ABX Guide. Burlington, MA: Jones & Bartlett Publishers; 2012:573.
17. Bayer HealthCare Pharmaceuticals Inc. AVELOX® (moxifloxacin hydrochloride) tablets. Published December 2008. Accessed March 8, 2014.
18. Pham PA, Bartlett JG. Moxifloxacin. Johns Hopkins Medicine. www.hopkinsguides.com/hopkins/ub/view/Johns_Hopkins_ABX_Guide/540355/all/Moxifloxacin. Updated March 14, 2011. Accessed March 8, 2014.
19. Universal Drugstore (UDS). Gemifloxacin. www.universaldrugstore.com/medications/Factive/320mg. Accessed March 10, 2014.
20. Gemifloxacin [prescribing information]. Cary, NC: Cornerstone Therapeutics Inc; 2011. Accessed March 18, 2014.
21. Physicians Desk Reference. Noroxin. Montvale, NJ: Thompson Reuters; 2009. Accessed March 18, 2014.
22. Bartlett JG, Auwaerter PG, Pham PA. Norfloxacin. In: Johns Hopkins ABX Guide. Burlington, MA: Jones & Bartlett Publishers; 2012:571.
23. Roberge RJ, Kaplan R, Frank R, Fore C. Glyburide-ciprofloxacin interaction with resistant hypoglycemia. Ann Emerg Med. 2000;36(2):160-163.
24. Jick SS, Jick H, Dean AD. A follow-up safety study of ciprofloxacin users. Pharmacotherapy. 1993;13(5):461-464.
25. Akpunonu B, Michaelis J, Uy CN, et al. Multicenter, postmarketing assessment of levofloxacin in the treatment of adults with community-acquired pneumonia. Clin Infect Dis. 2004;38(suppl 1):S5-S15.
26. Wang S, Rizvi AA. Levofloxacin-induced hypoglycemia in a nondiabetic patient. Am J Med Sci. 2006;331(6):334-335.
27. Mohr JF, McKinnon PS, Peymann PJ, Kenton I, Septimus E, Okhuysen PC. A retrospective, comparative evaluation of dysglycemias in hospitalized patients receiving gatifloxacin, levofloxacin, ciprofloxacin, or ceftriaxone. Pharmacotherapy. 2005;25(10):1303-1309.
28. US Food and Drug Administration. Notices: determination that Tequin (gatifloxacin) was withdrawn from sale for reasons of safety or effectiveness. Federal Register. 2008;73(175):52357-52358. www.gpo.gov/fdsys/pkg/FR-2008-09-09/pdf/E8-20938.pdf. Accessed March 18, 2014.
29. Frothingham R. Gatifloxacin associated with a 56-fold higher rate of glucose homeostasis abnormalities than comparator quinolones in the FDA spontaneous reporting database. Program and abstracts of the 44th Interscience Conference on Antimicrobial Agents and Chemotherapy; October 30-November 2, 2004; Washington, DC. Abstract A-1092.
30. Gavin JR III, Kubin R, Choudhri S, et al. Moxifloxacin and glucose homeostasis: a pooled-analysis of the evidence from clinical and postmarketing studies. Drug Saf. 2004;27(9):671-686.
31. Mandavia DR, Virpariya MM, Patel TK, Tripathi CB. Moxifloxacin-induced hypoglycemia in a non-diabetic patient. Curr Drug Saf. 2012;7(2):183-185.
32. ClinicalTrials.gov. Finafloxacin for the treatment of cUTI and/or acute pyelonephritis. www.clinicaltrials.gov/ct2/show/NCT01928433?term=finafloxacin&rank=2. Accessed March 18, 2014.
33. Patel H, Andresen A, Vente A, et al. Human pharmacokinetics and safety profile of finafloxacin, a new fluoroquinolone antibiotic, in healthy volunteers. Antimicrob Agents Chemother. 2011;55(9):4386-4393.
34. Saraya A, Yokokura M, Gonoi T, Seino S. Effects of fluoroquinolones on insulin secretion and beta-cell ATP-sensitive K+ channels. Eur J Pharmacol. 2004;497(1): 111-117.
35. Park-Wyllie LY, Juurlink DN, Kopp A, et al. Outpatient gatifloxacin therapy and dysglycemia in older adults. N Engl J Med. 2006;354(13):1352-1361.
36. Friedrich LV, Dougherty R. Fatal hypoglycemia associated with levofloxacin. Pharmacotherapy. 2004;24(12):1807-1812.
37. Routledge PA, O’Mahony MS, Woodhouse KW. Adverse drug reactions in elderly patients. Br J Clin Pharmacol. 2004;57(2):121-126.
38. Cooper JW. Probable adverse drug reactions in a rural geriatric nursing home population: a four-year study. J Am Geriatr Soc. 1996;44(2):194-197.
39. Coresh J, Selvin E, Stevens LA, et al. Prevalence of chronic kidney disease in the United States. JAMA. 2007;298(17):2038-2047.
40. Snyder JJ, Foley RN, Collins AJ. Prevalence of chronic kidney disease in the United States: a sensitivity analysis using the National Health and Nutrition Examination Survey (NHANES) 1999-2004. Am J Kidney Dis. 2009;53(2):218-228.
41. Bowling CB, Sharma P, Fox CS, O’Hare AM, Muntner P. Prevalence of reduced estimated glomerular filtration rate among the oldest old from 1988-1994 through 2005-2010. JAMA. 2013;310(12):1284-1286.
42. Cooper JW. Renal function assessment in nursing home patients. A prospective 6 month study in 282 patients. J Geriatr Drug Ther. 1991;5(3):59-72.
Disclosures: The authors report no relevant financial relationships.
Address correspondence to: Amit Bhargava, MD, Baystate Medical Center/TUFTS University School of Medicine, 759 Chestnut Street, Springfield, MA 01199; abhargava99@yahoo.com










