Evaluating the Use of a Skin Failure Indicator Scale in the Diagnosis of Acute Skin Failure
© 2024 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of ePlasty or HMP Global, their employees, and affiliates.
Abstract
Background. Critically ill patients are at increased risk of skin breakdown that may occur in non-pressure locations. Recognition of skin failure as distinct from hospital-acquired pressure injuries (HAPIs) may have significant clinical, financial, and legal implications. Objective. To apply a novel diagnostic scale in the care of patients with pressure injuries to determine its utility in diagnosing skin failure at a quaternary hospital. Materials and Methods. Critically ill patients at a quaternary hospital from September 2022 through November 2023 with a HAPI diagnosis were included. Charts were retrospectively reviewed for demographics, clinical outcomes, and wound parameters. Skin failure was evaluated using the Hill and Petersen Skin Failure Clinical Indicator Scale (SFCIS), with patients with a score of 15 to 21 considered to have high probability of skin failure. Results. During the study period, 12 patients were found to meet criteria for skin failure. For those patients, the mean length of stay was 58 days. All patients scored above 15 on the SFCIS, with 3 patients scoring 21 and 9 patients scoring 18. All patients met criteria for impaired blood flow and sepsis or multisystem organ dysfunction syndrome. Four patients met criteria for moderate or severe protein calorie malnutrition with an albumin level of less than 3.5 g/dL. Ten patients received mechanical ventilation for more than 72 hours. Conclusion. Twelve patients who were identified has having HAPIs were reclassified as having skin failure using the SFCIS. Further work is necessary to validate this scoring system in diagnosing skin failure and the implications associated with its use.
Abbreviations: CMS, Centers for Medicare & Medicaid Services; EMR, electronic medical record; HAPI, hospital-acquired pressure injury; ICU, intensive care unit; LOS, length of stay; MODS, multisystem organ dysfunction syndrome; NPIAP, National Pressure Injury Advisory Panel; PSI, patient safety indicator; SFCIS, Skin Failure Clinical Indicator Scale.
Background
Skin failure as a concept continues to be discussed and defined in the literature. By comparison, pressure injuries are well known and often are included in preventative practices to improve patient outcomes. Recognition of skin failure as a distinct entity from pressure injuries has both clinical and financial implications. HAPIs most often occur in critically ill patients, at rates as high as 45%.1 In 2008, the CMS announced cessation of reimbursements related to stage 3 and 4 HAPI care and in 2014, the Patient Protection and Affordable Care Act enacted a 1% penalty to hospitals with the highest rates of hospital-acquired conditions.1 A HAPI may result in added medical costs in excess of
$17 000 in added incremental charges for a given visit.2 Per the NPIAP, “a pressure injury is localized damage to the skin and underlying soft tissue usually over a bony prominence or related to a medical or other device….The injury occurs because of intense pressure, prolonged pressure or pressure in combination with shear.”3
In 1991, La Puma4 discussed the possibility of skin failing as an organ, similar to other organ systems. Irvine5 later defined skin failure “as a loss of normal temperature control with inability to maintain the core temperature, failure to prevent percutaneous loss of fluid, electrolytes and protein with resulting imbalance and failure of the mechanical barrier to penetration by foreign materials.” Skin failure wounds may initially present similar to HAPIs and are often misidentified as such. Typically, HAPIs present as red to maroon, round lesions over bony prominences. In contrast, skin failure wounds present as irregular, red or black in color, and pear- or butterfly-shaped, and they develop in multiple areas beyond bony prominences and pressure points.6 Skin failure is believed to be related to hypoperfusion due to a systemic imbalance caused by acute illness rather than a physical external force.6,7 Many pathophysiologic factors cause the skin to fail, including hypoperfusion, inflammation, hypoxia, vascular permeability, edema, and comorbid chronic medical conditions.8 It is oftentimes the case that skin failure is a diagnosis of exclusion rather than a primary diagnosis.
PSIs were developed by the Agency for Healthcare Research and Quality to track potentially avoidable safety events during hospitalization. The CMS PSI 90 is a subset of these PSIs and uses International Statistical Classification of Diseases and Related Health Problems, Tenth Revision data in its tracking.9 Stage 3, stage 4, or unstageable HAPIs have the designated CMS PSI 90 measure of PSI 03. Review of PSI 03 occurrences in patients during the COVID-19 pandemic identified many wounds documented as pressure injuries that did not meet NPIAP pressure injury definitions.10 It was noted that despite implementing pressure injury prevention strategies, certain patient populations continued to develop skin breakdown. This population primarily consisted of critically ill patients with common elements such as malnutrition, multisystem organ failure, impaired blood flow, mechanical respiratory ventilation, and vasopressor agent requirements—all factors that play a major role in the development of skin failure.11 Nieto-García et al10,11 found that patients with COVID-19 were 4 times more likely than patients without COVID-19 to develop acute wounds. It has been suggested that perturbations in markers of inflammation and a hypercoagulable phenotype in critically ill COVID-19 patients were associated with skin failure. Based on these findings, skin breakdown in patients may need to be regarded differently and the etiology of pressure injuries questioned. The wounds may not be caused by pressure but rather may be developing skin failure as a result of critical illness.6-8
There is a paucity of literature on the topic of skin failure, and both the definition and diagnosis of skin failure remain controversial.12 Several scoring systems have been proposed to adequately diagnose skin failure. In 2020, Hill and Petersen13 proposed the SFCIS to determine the probability that wounds are related to skin failure as opposed to HAPIs. Using this scale, the more criteria a patient meets, the higher the probability the patient’s wounds are related to skin failure. The aim of the present study was to apply this novel diagnostic scale to a retrospective case series of patients originally identified as having pressure injuries to determine its utility in diagnosing skin failure at a quaternary hospital.
Materials and Methods
Study design
Patient charts were retrospectively reviewed to identify patients who met skin failure criteria based on the Hill and Petersen SFCIS. Following MedStar Washington Hospital Center Institutional Review Board approval for retrospective chart review, patients who presented to a large, quaternary urban level I trauma center and who required ICU care from September 2022 to November 2023 were included for study. Evaluation of charts was conducted by a wound nurse, (H.H.), 3 physicians (J.F., J.S., S.T.), and a PSI committee. Inclusion criteria were patients 18 years of age or older, critically ill as defined by the necessity of ICU level of care, and the presence of a presumed stage 3, stage 4, or unstageable HAPI that fulfilled PSI 03 criteria as determined by the aforementioned PSI committee. Patients were otherwise excluded from chart review. Charts were reviewed for demographic information, physiologic parameters, hospital course, and associated outcomes. Wound parameters collected included location, color, progression, and shape. The treating attending physician was queried for documentation discrepancies. Wounds were evaluated for the presence of a HAPI compared with skin failure using the Hill and Petersen SFCIS (Table 1).13

Defining skin failure
In their multivariate analysis, Hill and Petersen13 demonstrated that 5 variables were significant for the diagnosis of skin failure: serum albumin level less than 3.5 g/dL, impaired blood flow, presence of sepsis or MODS, the use of vasopressors and/or inotropes, and mechanical ventilation. These variables were used to develop the SFCIS scoring system to evaluate the likelihood of skin failure in a patient. Some criteria in this scoring system are weighted more heavily than others, and the probability of skin failure increased as the score increased. To further delineate stringent criteria for skin failure, some criteria were modified. Because many acutely ill patients have a high probability of having an albumin level of less than 3.5 g/dL, patients in the present study were additionally scrutinized to determine if they also met American Society for Parenteral and Enteral Nutrition (ASPEN) guidelines for moderate or severe protein calorie malnutrition.12 To meet the criteria of impaired blood flow, a primary medical diagnosis indicating impaired blood flow to the skin was required. Criteria evaluated to confirm impaired blood flow included clinical statuses suggestive of poor tissue perfusion, such as hypotension, acute myocardial infarction, congestive heart failure, peripheral artery disease, anemia with hemoglobin level less than 7 g/dL, large volume blood loss, or history of radiation therapy.8 To qualify for sepsis or MODS, at least 2 organs—excluding skin—were required to be identified as failing. Regarding vasopressor and inotropic use, there was no consensus in the literature defining the type of agent, dosage, length of use, or the use of multiple vasopressor agents to indicate possible skin failure.14,15 Many studies had inconsistent data on how and which vasopressors affect the skin.16 For the present study, it was decided that norepinephrine use for more than 24 hours, or epinephrine or dopamine infusion rates more than 10 mcg/kg/min for more than 6 hours, would meet criteria for skin failure. Finally, review of several articles showed mechanical respiratory ventilation for 72 hours or more to be standard.7,13,15 Per the Hill and Petersen criteria, patients in the present study were considered to have a high probability of skin failure if they scored 15 or above.
Results
Patient demographics
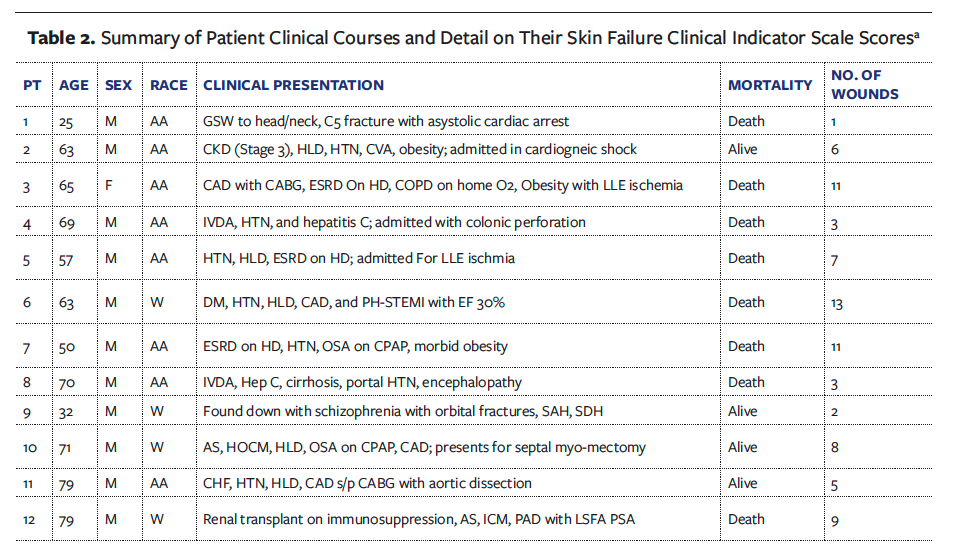
During the study period, 82 charts were reviewed by a PSI committee. Of these, 12 patients were found to meet the Hill and Petersen criteria for skin failure (Figure). These 12 patients were in the ICU due to critical medical illness. Patient ages ranged from 25 years to 79 years, with a mean age of 60.3 years. Eight patients were African American, and 4 were White. Race and sex were obtained from the patients’ EMR, which was recorded on admission and either self-reported or reported by a family member or equivalent legally authorized representative. The LOS ranged from 28 days to 136 days, with a mean LOS of 58 days. Eleven of the 12 patients were male, and 1 was female. Four of the 12 patients were discharged to the next level of care, and 8 patients died. Of the 12 patients, 3 were cared for by surgical services (trauma), 6 were under the cardiovascular service, and 2 were managed by an inpatient medical team.

Wound evaluation
All 12 patients had multiple wounds, with an overall mean of 6.6 wounds (range, 1 wound–13 wounds). Of the 12 patients, 6 had wounds on the sacrum, buttocks, or perineum, and 7 had wounds on the feet.
Skin failure
All 12 patients met the Hill and Petersen criteria, scoring above 15 on the SFCIS. Three patients received a maximum score of 21, while 9 received a score of 18. Of note, all 12 patients met the criteria for impaired blood flow as well as the criteria for sepsis or MODS, with at least 2 organ systems failing. Nine patients had a diagnosis of sepsis, and 3 had MODS. Of the 12 patients, 4 met the criteria for moderate or severe protein calorie malnutrition with an albumin level of less than 3.5 g/dL, all 12 patients required vasopressor agents, and 10 patients were placed on mechanical ventilation for more than 72 hours. Patient clinical courses and SFCIS scores are summarized in Table 2.

Discussion
Unlike when other organs fail, when skin fails there is no diagnostic test to provide definitive results. Skin failure must be visually observed and assessed while considering the patient’s clinical course. For this reason, skin failure is often dismissed or missed. While acute skin failure does develop and progress faster than pressure injuries do, it may not be evident to those unfamiliar with pressure injury timelines or skin conditions in general. In this study’s cohort of patients, the Hill and Petersen criteria proved suitable for identifying skin failure. All 12 patients were admitted to the ICU for either vasopressor or ventilator management. All patients had a primary diagnosis related to ischemia and hypoperfusion as well as a diagnosis of sepsis or MODS. All patients also required a vasopressor in the setting of shock of various etiologies.
While the skin is relatively resistant to hypoperfusion compared with other organs, the skin will ultimately fail without adequate perfusion. It can be assumed that if other organs are showing injury resulting from poor perfusion, then the skin is also at risk of hypoperfusion. Skin mottling is a known sign of hypoperfusion that clinicians often use for assessment. Correlation has been shown between mottling score variations and skin perfusion changes during septic shock resuscitation.17-20 Mottling itself may be an initial sign of skin failure.
It has been well documented that skin breakdown is associated with malnutrition. Munro et al21 demonstrated independent associations between malnutrition and skin tear prevalence. It has also been found that healing of pressure injury is strongly suppressed in malnourished patients. Of the patients evaluated in the present study, 4 met the criteria for moderate or severe protein calorie malnutrition with an albumin level of less than 3.5 g/dL. Despite this, multiple patients qualified for a high probability of skin failure without meeting the malnutrition criteria. The link between malnourishment and acute skin failure is unclear, but most likely, malnourishment can be a contributing factor.21
Of the examined wounds in the present study, wounds over the sacrum developed in 11 patients. These areas are more vulnerable to hypoperfusion and are at increased risk for compromise due to their dependent location. However, sacral wounds are different from pressure injuries due to the timeline of progression, which occurred at a greater rate and with increased severity compared with HAPI rates. In patients with multiple areas of skin breakdown, the mean number of wounds was 6.6, including areas not directly over a bony prominence. In patients who survived, wounds demonstrated evidence of healing and epithelialization as their acute illness resolved.
The Hill and Petersen criteria have implications for quality metrics as well as treatment. Pressure mitigating strategies are commonly used in hospital settings, but these strategies may not necessarily be effective in managing wounds caused by skin failure. Given that various etiologies typically drive skin failure, the underlying etiology should be addressed first. For skin failure cases, unless the driving clinical process is controlled, the skin will continue to worsen and will continue to deteriorate. Despite this, the diagnosis of acute skin failure should only be considered in the acutely critically ill patient if all pressure prevention strategies have been implemented and wounds still develop. If pressure mitigating strategies have not been initiated, doubt may remain that skin failure is the primary cause of skin breakdown and wound development. Incorrect identification of a pressure injury in this context may have a significant effect on hospital reimbursement, quality measures, and PSI 03. Education on skin failure principles and scoring systems like the SFCIS remain paramount to alerting providers and staff about this disease process and to altering pressure injury reporting practices. Pressure injuries are seen as an avoidable adverse event that may have legal ramifications compared with acute skin failure. For example, in 2019, Kim et al6 described a case in which a patient developed acute skin failure that was misdiagnosed as a pressure injury. The patient was a 74-year-old male with sepsis and pneumonia who developed multiple wounds that were labelled as pressure injuries. Although complete healing was achieved, the patient’s caregivers sued the hospital for failing to prevent pressure injury formation. Unlike pressure injury, skin failure may occur despite the implementation of all appropriate preventive measures and could be seen as an unavoidable event.6
Limitations
This study has several limitations. It is a retrospective study with a small sample size. Larger cohorts and prospective observational studies are necessary to fully elucidate the education, diagnosis, and practice management changes necessary to recognize skin failure as distinct from HAPIs and address skin failure accordingly. Additionally, the retrospective nature of this study makes standardized collection of clinical laboratory markers that affect wound healing (eg, vitamin D levels) difficult. As previously mentioned, consensus on a diagnosis for skin failure itself remains mixed. The diagnosis of skin failure may be underreported in the general literature due to the lack of understanding and recognition of this disease process. Furthermore, the present study used the Hill and Petersen SFCIS score to identify skin failure. Some aspects of the scoring system remain vague and required interpretation as discussed above for application and study. More stringent criteria or, in the present study, modification and solidification of definitions may be necessary to achieve definition consensus. Discussion on weighted criteria may also be necessary to account for the significant contribution of hypoperfusion to skin failure. Evaluation and diagnosis were also dependent on chart review and evaluation of wound photography. Other modalities, such as Doppler imaging or thermography, may provide additional information about wound status.
Conclusion
In the present study, 12 patients who were identified as having PSI events using traditional HAPI metrics were instead reclassified as having skin failure using the Hill and Petersen SFCIS. When evaluating wounds, providers must assess all risk factors in determining diagnosis. Distinguishing skin failure from other wound etiologies remains paramount due to the management, financial, and possibly legal implications associated with misdiagnosis. Further work is necessary to further validate the use of scoring systems such as the SFCIS in diagnosing skin failure and the implications associated with its use.
Author and Public Information
Authors: Jessica N. Fields, MD, MHA, MBA1; Hilary Hancock, BSN, RN, CWON1; Jeffrey W. Shupp, MD1-5; and Shawn Tejiram, MD1,2,4,5
Affiliations: 1MedStar Washington Hospital Center, Washington, DC, USA; 2The Burn Center, MedStar Washington Hospital Center, Washington, DC, USA; 3Department of Biochemistry and Molecular & Cellular Biology, Georgetown University Medical Center, Washington, DC, USA; 4Department of Surgery, Georgetown University School of Medicine, Washington, DC, USA; 5Department of Plastic and Reconstructive Surgery, Georgetown University School of Medicine, Washington, DC, USA
Disclosure: The authors disclose no financial or other conflicts of interest.
Ethical Approval: This study was approved by the MedStar Washington Hospital Center’s Institutional Review Board.
Correspondence: Shawn Tejiram, MD; Department of Surgery, MedStar Washington Hospital Center, 110 Irving St NW, Washington, DC 20010 United States; shawn.tejiram@medstar.net
Manuscript Accepted: February 5, 2025
References
1. Holt KR, Short MR, Berger SC, McCalla AW. Evaluation of vasopressor dose and the incidence of pressure injuries in critically ill patients. Adv Skin Wound Care. 2022;35(10):550-554. doi:10.1097/01.ASW.0000833604.12023.71
2. Au Y, Wang SC. Financial and clinical risk evaluation of pressure injuries in US hospitals: a business case for initiating quality improvement. Wounds. 2019;31(5):123-126.
3. National Pressure Injury Advisory Panel (NPIAP). NPIAP DTI evolution timeline. 2016. https://cdn.ymaws.com/npiap.com/resource/resmgr/online_store/npiap_pressure_injury_stages.pdf
4. La Puma J. The ethics of pressure ulcers. Decubitus. 1991;4(2):43-44.
5. Irvine C. "Skin failure"—a real entity: discussion paper. J R Soc Med. 1991;84(7):412-413. doi:10.1177/014107689108400711
6. Kim JH, Shin HK, Jung GY, Lee DL. A case of acute skin failure misdiagnosed as a pressure ulcer, leading to a legal dispute. Arch Plast Surg. 2019;46(1):75-78. doi:10.5999/aps.2018.00087
7. Delmore B, Cox J, Rolnitzky L, Chu A, Stolfi A. Differentiating a pressure ulcer from acute skin failure in the adult critical care patient. Adv Skin Wound Care. 2015;28(11):514-524. doi:10.1097/01.ASW.0000471876.11836.dc
8. Levine JM, Delmore B, Cox J. Skin failure: concept review and proposed model. Adv Skin Wound Care. 2022;35(3):139-148. doi:10.1097/01.ASW.0000818572.31307.7b
9. Bundled Payments for Care Improvement Advanced: Quality Measures Fact Sheet. Centers for Medicare & Medicaid Services. 2020. https://www.cms.gov/priorities/innovation/media/document/bpciadvancedmy123psi90fact-sheet
10. Nieto-García L, Carpio-Pérez A, Moreiro-Barroso MT, Ruiz-Antúnez E, Nieto-García A, Alonso-Sardón M. Are there differences between COVID-19 and non-COVID-19 inpatient pressure injuries? Experiences in internal medicine units. PLoS One. 2022;17(2):e0263900. doi:10.1371/journal.pone.0263900
11. Nieto-García L, Carpio-Pérez A, Moreiro-
Barroso MT, Ruíz-Antúnez E, Nieto-García A, Alonso-Sardón M. Clinical burden of inpatient wound care in internal medicine units during the first wave of COVID-19 outbreak. Adv Skin Wound Care. 2022;35(6):1-7. doi:10.1097/01.ASW.0000824564.25976.c8
12. Dalgleish L, Campbell J, Finlayson K, et al. Understanding skin failure: a scoping review. Adv Skin Wound Care. 2021;34(10):542-550. doi:10.1097/01.ASW.0000755844.87283.20
13. Hill R, Petersen A. Skin failure clinical indicator scale: proposal of a tool for distinguishing skin failure from a pressure injury. Wounds. 2020;32(10):272-278.
14. Cox J. Pressure ulcer development and vasopressor agents in adult critical care patients: a literature review. Ostomy Wound Manage. 2013;59(4):50-54, 56-60.
15. Cox J, Roche S. Vasopressors and development of pressure ulcers in adult critical care patients. Am J Crit Care. 2015;24(6):501-510. doi:10.4037/ajcc2015123
16. Bain M, Hara J, Carter MJ. The pathophysiology of skin failure vs. pressure injury: conditions that cause integument destruction and their associated implications. Wounds. 2020;32(11):319-327.
17. Ait-Oufella H, Lemoinne S, Boelle PY, et al. Mottling score predicts survival in septic shock. Intensive Care Med. 2011;37(5):801-807. doi:10.1007/s00134-011-2163-y
18. Ait-Oufella H, Bourcier S, Alves M, et al. Alteration of skin perfusion in mottling area during septic shock. Ann Intensive Care. 2013;3(1):31. doi:10.1186/2110-5820-3-31
19. Preda G, Bourcier S, Joffre J, et al. Mottling score is associated with 28-day mortality in critically ill patients with sepsis. Minerva
Anestesiol. 2017;83(6):664-666. doi:10.23736/S0375-9393.17.11816-X
20. Dumas G, Lavillegrand JR, Joffre J, et al. Mottling score is a strong predictor of 14-day mortality in septic patients whatever vasopressor doses and other tissue perfusion parameters. Crit Care. 2019;23(1):211. doi:10.1186/s13054-019-2496-4
21. Munro EL, Hickling DF, Williams DM, Bell JJ. Malnutrition is independently associated with skin tears in hospital inpatient settings: findings of a 6-year point prevalence audit. Int Wound J. 2018;15(4):527-533. doi:10.1111/iwj.12893

















