Classification, Diagnosis, and Treatment of Diabetic Foot Ulcers
The diabetic foot is considered one of the most significant complications of diabetes, representing a major worldwide medical, social, and economic problem that greatly affects patient quality of life. The risk for a patient with diabetes to develop a foot ulcer is close to 25%1 leading frequently to disablement and leg amputation.2,3 It has been estimated that every 30 seconds a lower limb is amputated somewhere in the world because of diabetes.4
The link between the diabetic foot ulcers and leg amputations is indisputable, as diabetes is the cause of almost 50% of all nontraumatic lower extremity amputations worldwide.5 Diabetic neuropathy and peripheral vascular disease are the main etiologic factors for the diabetic foot ulceration. Other factors that may act in combination to the above are: the unrecognized trauma, the biomechanical abnormalities, the limited joint mobility, and the increased susceptibility to infection.6
Early diagnosis and efficient treatment of the diabetic foot ulceration is essential in order to avoid limb amputation and preserve the life quality for the patient with diabetes. Accordingly, a specific treatment plan should be designed according to the size, depth, location of the ulcer, and presence/absence of infection or ischemia.6
Foot Ulcer Assessment and Classification
The evaluation and classification of diabetic foot ulcers are essential in order to organize the appropriate treatment plan and follow up. During the past years, several foot-ulcer classification methods have been proposed, however, none of the proposals have been universally accepted. The Wagner-Meggitt classification7 is based mainly on wound depth and consists of 6 wound grades. These include: grade 0 (intact skin), grade 1 (superficial ulcer), grade 2 (deep ulcer to tendon, bone, or joint), grade 3 (deep ulcer with abscess or osteomyelitis), grade 4 (forefoot gangrene), and grade 5 (whole foot gangrene). The University of Texas system7 grades the ulcers by depth and then stages them by the presence or absence of infection and ischemia. More specifically, grade 0 in the Texas System classification represents a pre- or postulcerative site. Grade 1 ulcers are superficial wounds through either the epidermis or the epidermis and dermis, but that do not penetrate to tendon, capsule, or bone. Grade 2 wounds penetrate to tendon or capsule, but the bone and joints are not involved. Grade 3 wounds penetrate to bone or into a joint. Each wound grade is comprised of 4 stages: clean wounds (A), nonischemic infected wounds (B), ischemic wounds (C), and infected ischemic wounds (D). The S(AD) SAD classification8 grades 5 ulcer features (size, depth, sepsis, arteriopathy, and denervation) on a 4-point scale (0–3). Similarly, the International Working Group on the Diabetic Foot has proposed the PEDIS classification,9 which grades the wound on a 5-feature basis: perfusion (arterial supply), extent (area), depth, infection, and sensation. Finally, according to the Infectious Diseases Society of America guidelines,10 the infected diabetic foot is subclassified into the categories of mild (restricted involvement of only skin and subcutaneous tissues), moderate (more extensive or affecting deeper tissues), and severe (accompanied by systemic signs of infection or metabolic instability).6
In a simplified clinical classification approach, diabetic foot ulcers can be characterized as neuropathic, ischemic, or neuroischemic, depending on how complications such as peripheral neuropathy and arterial disease affect the ulcer’s etiology.11
Other important factors that must be considered are: wound size and depth, the presence of sinus tracts or probing to bone, the amount of granulation tissue, the amount of fibrotic or dysvascular tissue, the type and amount of drainage, the amount of hyperkeratotic tissue surrounding the wound, and signs of infection such as erythema, edema, odor, or increased warmth.6
Wound size and depth should be documented at the initial visit and at each subsequent visit.12 Wound area can be measured in several ways and include tracings inked on clear acetate, calibrated digital photographs, or direct measurement of maximum length and width with a small ruler or scalpel blade handle. Wound depth can be determined by using a sterile probe or sterile cotton-tipped applicator in the deepest part of the wound. These measurements provide the basis for estimating the progression of wound contraction and filling, and more importantly, the efficacy of the current treatment.6
The tissue present at the base of the wound can provide a wealth of information regarding the vascularity and the possible presence of infection. Ideally, a healthy, well-perfused wound demonstrates a red granular bed that bleeds well with debridement. A wound with abundant fibrotic or necrotic tissue, or a wound with a dry desiccated appearance may indicate impaired vascular perfusion (Figure 1). 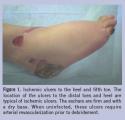 In such a case, effective revascularization surgery is necessary before initiation of any wound care treatment.6
In such a case, effective revascularization surgery is necessary before initiation of any wound care treatment.6
Plantar wounds are usually caused by repetitive, chronic, low-velocity trauma to a neuropathic, insensate diabetic foot. Reduced sensation can substantially impair the patient’s perception of touch, deep pressure, temperature, vibration, and joint position making the foot susceptible to neuropathic ulcer development. Mechanical damage to poorly perfused tissues due to peripheral vascular disease typically causes ischemic ulcers.6 Reducing repetitive trauma is essential to promote proper healing of plantar foot ulcers. Examination of the tissue surrounding the wound can determine the efficacy of wound offloading protocol. The presence of thick, hyperkeratotic tissue around the wound indicates inadequate pressure offloading.6,13
Treatment Cornerstones
Debridement. Debridement is widely recognized as one of the most important techniques in wound bed preparation. Apart from having all nonviable tissues removed, wound debridement also promotes the release of growth factors that contributes to more progressive wound healing. Furthermore, it removes nonviable or infected tissue and stimulates what may have been a previously nonadvancing wound edge.
Wound debridement targets complete removal of all necrotic, dysvascular, and nonviable tissue, to achieve a red, granular wound bed (Figure 2). In a case where ischemia is suspected, aggressive debridement should be postponed until a vascular examination is obtained and, if necessary, a revascularization procedure is completed.6,13
Presently, the most widely used wound debridement methods are: surgical (sharp), autolytic, chemical, mechanical, and biologic. Autolytic, chemical, and surgical methods remove mainly necrotic tissue (selective debridement), whereas mechanical methods remove both necrotic and viable tissue (nonselective debridement). 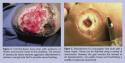 Sharp debridement. Sharp technique for wound debridement is the most efficient and is typically utilized when a wound has a significant amount of necrotic debris or infected tissue. Sharp technique is performed using surgical instruments. Sharp debridement is advantageous to the clinician because he or she can evaluate the size and the severity of the wound. Sharp debridement is considered the gold standard for debriding diabetic foot ulcers.14 Additionally, the physician using this technique is able to obtain tissue for culture when infection of the deep tissues or osteomyelitis is suspected.
Sharp debridement. Sharp technique for wound debridement is the most efficient and is typically utilized when a wound has a significant amount of necrotic debris or infected tissue. Sharp technique is performed using surgical instruments. Sharp debridement is advantageous to the clinician because he or she can evaluate the size and the severity of the wound. Sharp debridement is considered the gold standard for debriding diabetic foot ulcers.14 Additionally, the physician using this technique is able to obtain tissue for culture when infection of the deep tissues or osteomyelitis is suspected.
Using a scalpel blade with the tip pointed in a 45-degree angle, all nonviable tissue can be removed until a healthy bleeding ulcer bed is produced with saucerization of the wound edges. Alternatively, a tissue nipper may be used (Figure 3). Trained individuals who are able to perform it safely and with minimal trauma to vital tissues should perform the sharp method of debridement. Sharp debridement can be performed in the office setting on the insensate foot. However, if debridement is extensive or sensation to the foot is intact, sharp debridement should be carried out in the operating room under appropriate anesthesia and in sterile conditions. Finally, in a case where infection has destroyed foot function or if infection threatens the patient’s life, a guillotine amputation may be necessary to allow prompt control of the infection with subsequent definitive closure.14
Ideally, ulcer treatment should minimize tissue loss, preserve optimal foot function, and avoid deformities that might lead to recurrent ulceration. The optimal treatment should also recognize the potential for revascularization to ensure healing. The surgeon’s experience in this area and vascular surgery support are important to achieve optimal results.14
Enzymatic debridement. Enzymatic debridement involves the use of topical agents that degrade necrotic tissue via proteolytic enzymes such as papain, collagenase, fibrinolysin-DNase, papain-urea, streptokinase-streptodornase, and trypsin. These agents are typically applied once daily on the ulcer that is then covered with an occlusive dressing. Studies on the clinical efficacy of these agents have reported variable results. While the number of large clinical trials completed to date is small, it seems that the use of these agents does not provide additional benefits compared to the standard therapeutic treatment.15,16 Thus, indications for their use are limited and generally indicated to slowly soften large eschars or debride decubitus ulcerations in sensate limbs and in wounds with marginal arterial perfusion. Eschars can be crosshatched with a scalpel blade to increase the effectiveness of these agents. The primary drawbacks for the use of these agents are the lengthy process and their high cost.17
Mechanical debridement gently loosens and removes slough from the wound bed. The simplest form of this technique is the application of wet-to-dry saline gauze. After the wet gauze dressing is applied to the wound bed and allowed to dry, necrotic debris becomes embedded in the gauze and is mechanically stripped from the wound bed when the gauze is removed. Although this is an inexpensive and relatively simple technique, it may remove both viable and nonviable tissue causing pain in the sensate foot; therefore, its use is usually reserved for the treatment of larger, highly exudative wounds. The use of this method in small wounds is minimized in moistening necrotic eschars facilitating their removal.18
Recently, both historical and novel means of wound debridement have been reported. The resurgence of maggot therapy for debridement of necrotic tissue, decreasing bacterial load and stimulation of wound healing regained popularity for use on nonhealing wounds. Although several recent clinical trials have shown maggot therapy to be effective in the treatment of diabetic foot ulcers and pressure ulcers that were unresponsive to conventional therapy,19–21 in the United States, this therapy has not been fully embraced by the medical community mainly due to the high cost of medical grade maggots. The use of low energy ultrasound mist (MIST™, Celleration Inc, Eden Prairie, Minn) has been advocated for the debridement of diabetic foot ulcers.22 This method of wound debridement continues to undergo both clinical and scientific research.
Pressure Offloading
Pressure relief on ulcers, commonly called offloading, should always be a part of the treatment plan for plantar foot ulcers. As discussed previously, ulcerations occur in high-pressure areas of the insensate foot. Many methods have been employed to reduce foot pressures with varying success. The most popular methods include total contact casting, half shoes, short leg walkers, and felted foam dressings.
Total contact casting has been considered as the most effective method for offloading diabetic foot ulcers as measured by wound healing rate.23 This therapeutic method involves the use of a well-molded minimally padded plaster cast, resulting in equal pressure distribution to the entire limb. This method allows the patient to move during the treatment and has been found to be helpful in edema control, which is linked to healing impairment.24 Total contact casts are successful when properly applied and changed at least weekly.25 A factor that contributes to their success is the patient’s inability to remove the cast easily. Disadvantages include the considerable skill and time required for application, the possibility of secondary skin lesions due to cast irritation, and the inability to assess the wound daily (Figure 4). The Scotchcast boot is an alternative approach to the total contact cast technique that is constructed of a well-padded plaster boot cut away at the ankle and is designed to be removable by cutting away the cast over the dorsum of the foot. Windows are cut under the ulcer, and the boot is worn with a cast sandal to increase mobility while also protecting the ulcer from any pressure.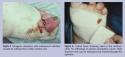 Due to the significant disadvantages associated with the total contact cast, few clinicians use this technique. Commercially available devices, such as the half shoe and short leg walker, are more commonly used. Both devices are relatively inexpensive, easy to use, and readily accepted by the patient. However, pressure reduction is significantly less compared to total contact casting and patient compliance cannot be ensured because the devices are removable.26
Due to the significant disadvantages associated with the total contact cast, few clinicians use this technique. Commercially available devices, such as the half shoe and short leg walker, are more commonly used. Both devices are relatively inexpensive, easy to use, and readily accepted by the patient. However, pressure reduction is significantly less compared to total contact casting and patient compliance cannot be ensured because the devices are removable.26
Felted foam dressings are accommodative offloading devices fashioned from a felt-foam pad with an aperture over the ulceration that allows for customized pressure relief. The pad is generally attached by tape or rubber cement directly applied to the patient’s skin, which prevents pad migration and ensures a degree of patient compliance. Wound care and wound assessment can be performed through the aperture (Figure 5). The felted foam is often used in conjunction with a surgical shoe or half-shoe and must be changed every 10–14 days to ensure integrity of the dressing. Felted foam dressings in combination with a surgical shoe or half-shoe were found to be more effective in pressure reduction when compared to a short leg walker or a half-shoe alone.27
Regardless of the technique used for offloading, patients with diabetic foot ulcers should be advised to reduce their everyday activity until the ulcer has completely healed. Patients are typically less active in total contact casts than in healing shoes,28 presumably due to the bulk and weight of the nonremovable device. Increased activity, with consequent high cumulative pressure load, can delay or prevent ulcer healing.29,30
Treatment of Infection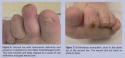 A diabetic foot ulcer serves as a portal of entry for bacteria than can lead to wound infection. Diagnosis of infection is primarily based on clinical appearance and signs such as erythema, edema, pain, tenderness, and warmth. High levels of bacteria (≥ 1 x 106 CFU/g of tissue) impede wound-healing processes and obdurate spontaneous healing and eventual surgical closure of diabetic ulcers.31 The severity of the infection can range from a superficial cellulitis (Figure 6 and 7) to a deep abscess or necrotizing fasciitis (Figure 8) with systemic toxicity. Care must be taken to diagnose and treat infections sufficiently, as mild cellulitis can rapidly progress to a limb threatening infection if left untreated.6
A diabetic foot ulcer serves as a portal of entry for bacteria than can lead to wound infection. Diagnosis of infection is primarily based on clinical appearance and signs such as erythema, edema, pain, tenderness, and warmth. High levels of bacteria (≥ 1 x 106 CFU/g of tissue) impede wound-healing processes and obdurate spontaneous healing and eventual surgical closure of diabetic ulcers.31 The severity of the infection can range from a superficial cellulitis (Figure 6 and 7) to a deep abscess or necrotizing fasciitis (Figure 8) with systemic toxicity. Care must be taken to diagnose and treat infections sufficiently, as mild cellulitis can rapidly progress to a limb threatening infection if left untreated.6
Diagnostic tools such as cultures, radiographs, or other more advanced imaging techniques can be useful in the diagnosis and treatment progression of diabetic foot ulcers. When clinical infection is suspected, cultures taken from the wound area could be useful in determining appropriate antibiotic therapy. Although the most frequent infections are due to aerobic Gram-positive cocci and aerobic Gram-negative organisms, anaerobic organisms are often isolated. Clinically uninfected ulcers should not be cultured, as the recovered organisms will contain only colonizing flora. Empirical antibiotic therapy should be started, and revised if necessary, once culture results are obtained.6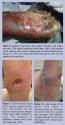 Radiographic imaging of the infected foot can reveal increased density and thickening of the subcutaneous fat along with blurring of the usually visible fat planes.32 Presence of osseous changes, such as periosteal reaction, cortical bone destruction, and focal osteopenia, may suggest the presence of osteomyelitis. However, these changes only become evident after osteomyelitis has been present for 10–14 days, and bone loss may reach almost 50% before it is recognized.33 Advanced imaging techniques, such as magnetic resonance imaging and computed tomography, may aid in the accurate diagnosis of osteomyelitis and detection of abscess formations.
Radiographic imaging of the infected foot can reveal increased density and thickening of the subcutaneous fat along with blurring of the usually visible fat planes.32 Presence of osseous changes, such as periosteal reaction, cortical bone destruction, and focal osteopenia, may suggest the presence of osteomyelitis. However, these changes only become evident after osteomyelitis has been present for 10–14 days, and bone loss may reach almost 50% before it is recognized.33 Advanced imaging techniques, such as magnetic resonance imaging and computed tomography, may aid in the accurate diagnosis of osteomyelitis and detection of abscess formations.
The treatment of infected ulcers requires the appropriate antibiotic therapy along with diligent debridement of all necrotic tissue and drainage of purulent collections (Figure 9 and 10). Antibiotic selection should consider the most likely causative microorganism(s) while also acknowledging the potential toxicity of the agents. In the diabetic foot, the bacteria most likely responsible for non-limb threatening infections are staphylococcus and streptococci, while limb-threatening infections are generally a consequence of a polymicrobial infection.34
Empiric antibiotic selection should be based on the suspected bacterial pathogens as well as the anticipated resistant pathogens that may have been selected during prior hospitalizations (Table 1).  At the authors’ institution, the prevalence of methicillin-resistant Staphylococcus aureus (MRSA) is high. Therefore, patients with an infected diabetic ulcer and history of a resistant staphylococcus infection are empirically placed on antibiotics. Antibiotic selection should minimize toxicity and be cost effective. Broad-spectrum antimicrobial therapy should begin empirically with reassessment after the results of culture and sensitivities have been obtained. Treatment regimens should then be simplified based on culture data.
At the authors’ institution, the prevalence of methicillin-resistant Staphylococcus aureus (MRSA) is high. Therefore, patients with an infected diabetic ulcer and history of a resistant staphylococcus infection are empirically placed on antibiotics. Antibiotic selection should minimize toxicity and be cost effective. Broad-spectrum antimicrobial therapy should begin empirically with reassessment after the results of culture and sensitivities have been obtained. Treatment regimens should then be simplified based on culture data.
Mild cellulitis can be treated with oral antibiotic therapy on an outpatient basis. It has been long been accepted that diabetic foot infections are polymicrobial. However, in mild infections, the most common causative organisms are Gram-positive cocci. Therefore, empirical therapy may be appropriate with a first generation cephalosporin or beta-lactamase inhibitor.
Moderate to more severe infections are characterized by erythema that exceeds the wound margins by 2 cm. Purulent discharge and associated constitutional symptoms of fever and chills may be present. These severe infections are typically polymicrobial and require hospitalization and treatment with intravenous (IV) antibiotics. In the authors’ institution, empirical therapy consists of vancomycin, levofloxacin, and metronidazole. This combination of antibiotics covers the full spectrum of potential infectious organisms and can be adjusted following culture results.
The duration of antimicrobial therapy for severe soft tissue infections of the foot is based on response to the antibiotics and wound care. A 2-week therapy course is commonly used by most physicians; however, recalcitrant infections may require longer courses. Osteomyelitis is best treated by removing the infected bone that is followed with 2–4 weeks of antibiotic therapy.
Once evidence of infection has been resolved, antibiotics can be discontinued even if the ulcer has not fully healed. Continuation of antibiotics beyond this point has not demonstrated any effect on wound healing.35,36
The effectiveness of the antibiotics is closely related to the condition of foot vasculature. Thus, poor blood supply to the foot results in inadequate antibiotic distribution, leading to a significant delay in infection control; therefore, the appropriate antibiotic treatment must be combined with all required revascularization procedures in order to restore blood perfusion to the foot.
Wound Care
Assiduous and effective use of dressings is essential to ensure optimal management of diabetic foot ulcers. The concept of a clean, moist wound-healing environment has been widely accepted. Benefits of this approach include prevention of tissue dehydration and cell death, acceleration of angiogenesis, and facilitation of the interaction of growth factors with the target cells.37 Additionally, it has been found that patients experience less discomfort with moist wound dressings.6 The notion that a moist wound environment increases the risk of developing an infection appears to be unfounded. Although many wound care products are available on the market that promote moist wound healing, wet-to-dry normal saline gauze remains the standard of care.
The concept of wound bed preparation represents a new direction in wound care thinking. Wound bed preparation mainly targeting to the creation of a healthy wound bed through management of bacterial load and moisture levels within the wound. Wound bed preparation practically aims in converting the molecular and cellular environment of a chronic wound to that of an acute healing wound.
Surgical debridement is considered to be the golden standard for wound bed preparation as it adequately removes the necrotic tissue, the bacterial burden and possibly, phenotypically altered cells that may interfere with healing, and reduces edema and exudates, attaining a clean, red and granular wound bed. Many clinicians advocate the use of antimicrobial wound products such as those that contain silver or iodine. Despite the widespread use of these dressings, no randomized, controlled clinical trials to evaluate their clinical effectiveness in preventing or eradicating infection have been performed. Furthermore, recent studies have shown that modern wound dressings including activated charcoal, alginates, hyaluronic acid, hydrofibers, hydrogels, hydrocolloids, collagen, proteolytic enzymes, foams, semipermeable membranes and silver dressings, presented similar results in wound healing progress in comparison to traditional moist gauze technique.38 However, in the authors’ opinion, these products may be helpful in wound bed preparation for the use of advanced wound care products such as the living skin equivalents.
Advanced Wound Care Products
Advanced wound care products have been developed in response to an improved understanding of the impaired wound healing integral in the diabetic foot ulcer. Pathophysiologic defects such as decreased growth factors production and cellular inactivity have led to the development of products that address these deficiencies. Products in this category include recombinant platelet derived growth factor and biological skin substitutes.
Recombinant human platelet-derived growth factor-BB (rhPDGF-BB)(becaplermin) is the only growth factor to date approved by the US Food and Drug Administration for the treatment of diabetic foot ulcers. Levels of PDGF have been shown to be lower in chronic wounds.39 Administered in a gel formulation concurrent with a standardized regimen of good wound care, becaplermin gel increased both the incidence of complete wound closure and decreased the time to achieve complete wound healing.40 Additionally, becaplermin has been anecdotally noted to improve healing times in the patient with renal insufficiency where wound healing is particularly difficult.
Living skin equivalents (LSE) are FDA-approved biological skin substitutes for use in diabetic foot ulcers. While the precise mechanism of action of LSE is not completely understood, it is believed that improved wound healing is because LSE fill the wound with extracellular matrix proteins and subsequently induce and express growth factors and cytokines that are necessary for wound healing. Additionally, the matrix components may further facilitate the recruitment of cells to the wound, improving wound repair.41
Living skin equivalents can be applied in a semi-sterile environment (eg, office setting). The base of the wound bed should be prepared to stimulate granulation; minimal to no fibrotic or necrotic tissue should be present. A moist, nonadherent dressing is then applied and left on for 3–5 days. At the first dressing change, the presence of the LSE may be evident in the wound bed41 (Figure 11).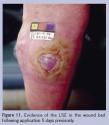 Advanced wound care products have been criticized because they are expensive and are overused when standard wound care treatments, such as normal saline gauze dressings, provide comparable healing rates. It has been demonstrated that percent change in foot ulcer area at a 4-week observation point is a good indicator of wound healing at 12 weeks.42 According to recent studies regarding evidence-based protocols for diabetic foot ulcer treatment, the advanced wound care products should be considered in cases where traditional care does not succeed in 50% wound reduction after a 3-week treatment period.43
Advanced wound care products have been criticized because they are expensive and are overused when standard wound care treatments, such as normal saline gauze dressings, provide comparable healing rates. It has been demonstrated that percent change in foot ulcer area at a 4-week observation point is a good indicator of wound healing at 12 weeks.42 According to recent studies regarding evidence-based protocols for diabetic foot ulcer treatment, the advanced wound care products should be considered in cases where traditional care does not succeed in 50% wound reduction after a 3-week treatment period.43
Negative Pressure Wound Therapy and Hyperbaric Oxygen
The use of negative pressure wound therapy (NPWT) devices may by useful in treatment of nonhealing wounds as they may reduce edema, remove bacterial products, and draw the edges of wound together to promote closure. Thus, their use should be considered when other treatments are not effective.44 Finally, hyperbaric oxygen therapy (HBOT) may be beneficial as it has been proven to reduce amputation rates in patients with ischemic diabetic foot ulcers.44
Conclusion
Diabetic ulcers are chronic wounds that are the result of repetitive trauma in an insensate foot. Furthermore, the unique characteristics of diabetes results in poor resistance to infection and peripheral vascular disease that makes treatment of these wounds difficult. However, adherence to good wound care principles, such as aggressive debridement of all non-viable tissue, adequate pressure offloading, prompt infection treatment, use of moist wound dressings, judicious use of advanced wound care products, and tight glucose control, may provide the best means of treating these challenging wounds. Finally, it is necessary to note that self-assessment and foot inspection, especially in patients with diabetes diagnosed with peripheral neuropathy, is essential in order to detect onset of possible ulceration. Early detection and treatment of a diabetic ulcer can halt the development of complications and save the foot from possible amputation.
















