Persistence of Bilayered Living-Cell Therapy Donor DNA 10 Months after Application: A Case Report
Abstract
Bilayered living-cell therapy has been shown in clinical trials to improve the rate of healing diabetic and venous leg ulcerations. However, literature is conflicted regarding the length of time living-cell therapy persists in acute and chronic wounds. An otherwise healthy 48-year-old man with sepsis from extensive lower-extremity wounds due to ecthyma gangrenosum was admitted to the hospital. Initial treatment consisted of surgical debridement, broad-spectrum antibiotics, and stabilization in the intensive care unit.
After the patient was stabilized, three units of living-cell therapy meshed at a 3:1 ratio were applied to the wounds. Both wounds were healed after 20 weeks. Ten months post grafting, the patient returned with a recurrent ulcer in the area of graft placement. A biopsy of the wound for human leukocyte antigen revealed the presence of donor DNA from the bilayered living-cell therapy. This case suggests that bilayered living-cell therapy may persist for prolonged periods in patients without underlying skin disease or immunosuppression. Research to explicate the mode of action and optimal protocols for use of living-cell therapy is warranted.
Potential Conflicts of Interest: Dr. Serena discloses he has received speaker honoraria from and served as a consultant or paid advisory board member for Organogenesis, Canton, MA. Dr. Bialas has nothing to disclose.
Please address correspondence to: Thomas Serena, MD, FACS, Penn North Centers for Advanced Wound Care, 552 Quaker Hill Rd, Warren, PA 16365; email: serena@healingwounds.com.
Bilayered, living-skin, cell therapy (Apligraf®, Organogenesis, Canton, MA) is composed of human epidermal keratinocytes, human dermal fibroblasts, and an extracellular matrix of bovine collagen. The keratinocytes and fibroblasts are derived from human neonatal foreskin. This bilayered living-cell therapy does not contain antigenic cells such as Langerhans cells, melanocytes, macrophages, lymphocytes, blood vessels, or hair follicles and there is no evidence of an immune response to the cellular components of this cell therapy on immunologic testing.1,2
In multicenter clinical trials,2-4 bilayered living-cell therapy has been shown to improve healing rates in venous leg and diabetic foot ulcerations compared to standard of care. In a randomized, unmasked, multicenter study of 127 patients with venous leg ulcerations >1-month duration, Falanga and the Human Skin Equivalent Investigators Group2 found that, after 6 months, the proportion of patients healed in the bilayered living-cell therapy was higher than that in the control group using compression therapy alone (63% versus 49%; P = 0.02). In a prospective, randomized, multicenter trial5 of diabetic foot ulcers treated with saline-moistened gauze control or bilayered living-cell therapy and conventional care (including extensive surgical debridement and foot offloading), the time to wound closure was 65 days in the treatment versus 90 days in the control group (P = 0.003).
Several theories have been proposed to explain the improved healing rates in chronic wounds after the application of bilayered living-cell therapy. One popular theory is that it facilitates healing by delivering much-needed matrix materials and cytokine growth factors in the appropriate concentrations.4,6 It also has been suggested that bilayered living-cell therapy may act  similarly to an autologous split-thickness skin graft or a biologic dressing, both of which persist within the healed wound bed. However, a study using polymerase chain reaction (PCR) DNA analysis have demonstrated that allogenic DNA does not persist beyond 1 month in venous leg ulcerations or split-thickness donor sites treated with bilayered living-cell therapy.7,8 A growing body of evidence1,9 suggests that in certain conditions, such as epidermolysis bullosa, bilayered living-cell DNA remains present in the wound after 3 months.
similarly to an autologous split-thickness skin graft or a biologic dressing, both of which persist within the healed wound bed. However, a study using polymerase chain reaction (PCR) DNA analysis have demonstrated that allogenic DNA does not persist beyond 1 month in venous leg ulcerations or split-thickness donor sites treated with bilayered living-cell therapy.7,8 A growing body of evidence1,9 suggests that in certain conditions, such as epidermolysis bullosa, bilayered living-cell DNA remains present in the wound after 3 months.
The purpose of this case study was to investigate the presence of DNA in bilayered skin in a nonimmune-compromised patient without underlying skin disease 10 months following application of a bilayered living-cell treatment.
Case Report
History. In November 2000, Mr. N, a 48-year-old man in septic shock, was transferred to the intensive care unit from an outside hospital. The source of his sepsis was multiple necrotic lower-extremity wounds (see Figure 1). His family reported his problem began 1 week prior when several lower-extremity ulcerations developed. The lesions began as small black areas of skin. The patient had no prodromal symptoms and was otherwise healthy. The lesions rapidly increased in size and his legs became erythematous, warm, and swollen. The wound began draining a foul-smelling, tan fluid.
Mr. N was admitted to a rural community hospital. Penicillin (6 g every 6 hours intravenously [IV]) and clindamycin (900 mg IV every 8 hours) were started. Betadine was applied liberally to the ulcers present on both lower extremities. Shortly after admission, Mr. N developed a fever of 39˚ C and became hypotensive. The cellulitis was reported to be progressing rapidly.
Mr. N was transferred to the authors’ institution for surgical debridement and 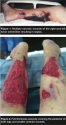 medical management and admitted directly to the ICU. Cultures obtained on admission grew Pseudomonas aeruginosa. He was resuscitated in the ICU by the critical care physician. Fresh frozen plasma was given to reverse the coagulopathy associated with his septic shock. The penicillin was changed to ceftazidime (1 g every 6 hours). He then was taken to the operating room where he underwent extensive debridement of nonviable tissue. At this time, he was diagnosed with ecthyma gangrenosum, a potentially lethal infectious disease resulting from Pseudomonal infection.
medical management and admitted directly to the ICU. Cultures obtained on admission grew Pseudomonas aeruginosa. He was resuscitated in the ICU by the critical care physician. Fresh frozen plasma was given to reverse the coagulopathy associated with his septic shock. The penicillin was changed to ceftazidime (1 g every 6 hours). He then was taken to the operating room where he underwent extensive debridement of nonviable tissue. At this time, he was diagnosed with ecthyma gangrenosum, a potentially lethal infectious disease resulting from Pseudomonal infection.
Mr. N’s initial postoperative course was complicated. Hepatic failure led to a persistent coagulopathy, requiring administration of fresh frozen plasma and vitamin K. His congestive heart failure responded to diuretics. The posterior aspects of both legs were covered with full-thickness wounds; several smaller anterior wounds also were present (see Figure 2). The wounds were treated with saline dressings and repeated sharp bedside debridement.
After 2 weeks, his condition stabilized and the wounds developed a fully granulated base, free of nonviable tissue. The surgical team suggested split-thickness grafting for wound coverage. However, due to ongoing cardiac concerns, including a critically low cardiac ejection fraction, it was decided to forego any surgical intervention that required conscious sedation and bilayered living-cell therapy was applied to the wound bed. The procedure was performed at the bedside in the ICU. Three units of bilayered living-cell therapy were meshed at a 3:1 ratio and applied to the posterior aspects of both legs. The grafts were held in place with petrolatum/bismuth tribromophenate gauze (Xeroform®, Tyco Healthcare/Kendall, Mansfield, MA) and elastic compression wraps. No anesthetic was required. No complications occurred during the procedure.
Follow-up. Mr. N’s left leg wound was completely closed after 5 weeks. The right leg, which had the larger wound, was 90% healed at 12 weeks and completely closed at 20 weeks. No additional living-cell treatment applications were performed.
Mr. N returned 10 months after his original grafting procedure. He had developed two open areas on the posterior aspect of the right leg as a result of trauma several weeks before his visit. At this point, the authors noted that the extremity did not have the typical appearance of a leg treated with a meshed graft — ie, no “pie-crusting” of the grafted skin covering the wounded area was observed (see Figure 3). The unusual appearance of the previous graft site prompted an investigation into the nature of the skin. Mr. N’s human leukocyte antigen (HLA) type was determined by examining  peripheral blood. The HLA type of the living-cell therapy donor, on file with the manufacturer, showed that all three grafts used on Mr. N had come from a single donor. A 6-mm biopsy of skin from his posterior right calf was sent for HLA typing; two loci revealed the persistence of DNA from the living-cell therapy donor 10 months after initial application (see Figure 4). This was the only graft site tested on the patient.
peripheral blood. The HLA type of the living-cell therapy donor, on file with the manufacturer, showed that all three grafts used on Mr. N had come from a single donor. A 6-mm biopsy of skin from his posterior right calf was sent for HLA typing; two loci revealed the persistence of DNA from the living-cell therapy donor 10 months after initial application (see Figure 4). This was the only graft site tested on the patient.
Discussion
It has long been held that cultured allografts do not persist in vivo and that host cells rapidly replace the donor cells. In an open-label study8 of 10 patients with venous leg ulcerations treated with bilayered living cell therapy, the majority of patients showed no evidence of the product at 4 weeks post application. Bilayered living-cell therapy was thought to be rapidly replaced by host fibroblasts and keratinocytes. However, several case studies1,9 note persistence of the DNA of the bilayered living cells in patients with epidermolysis bullosa. Epidermolysis bullosa is a group of disorders characterized by blistering of the skin following minor trauma that may require skin coverage. Bilayered living-cell therapy has been successfully employed for wound coverage in this population, avoiding the need to create another difficult-to-heal wound for autografting. Subsequent biopsies in a small sample of female patients with epidermolysis bullosa revealed the presence of Y chromosomes, indicating the persistence of the bilayered living-cell treatment.1 One theory for the difference noted in living-cell treatment persistence between patients with venous ulcers and epidermolysis bullosa may be that this observation is somehow related to the underlying cutaneous disease of the latter.
In this case report, the presence of bilayered living-cell therapy DNA persisted 10 months after application in a patient without an underlying skin disease. Although it is possible that the persistence of donor DNA is related to Mr. N’s immunocompromised status before and at the time of grafting due to his acute illness and sepsis, his sepsis had resolved at the time of grafting and he was not immune-compromised during the 10 months preceding the biopsy.
To the authors’ knowledge, this is the first published report of long-term persistence of bilayered living-cell therapy in a patient without underlying cutaneous disease. It suggests that in some patients, this graft tissue may “take” and the cells persist for a considerable length of time. The reason for this isolated finding may be locked in the mechanism by which this treatment improves chronic wound healing rates. A decade of experience with this particular bilayered living-cell therapy leads the authors to believe that in the majority of cases, there is no persistence. However, in some wounds, such as the one described in this case study, graft take appears similar to an autograft. In his grading system for ulcers treated with bilayered living-cell therapy in which A grading signifies apparent persistence of the bilayered skin product, Saap et al10 termed this an A grade. Analysis of the venous leg ulcer clinical trial suggested that the A grading occurs infrequently. In the majority of wounds, it is more likely that the bilayered living-cell therapy delivers growth factors to the wound bed.4 Preliminary data11 from a gene chip affermetrix study examining the changes in gene expression in 10 patients with 13 venous leg ulcerations after application of bilayered skin suggest the treatment functions through an upregulation of genes important to the wound healing process.
The possibility that immunosuppression played a role in the persistence of donor DNA observed in this case raises the question as to whether a brief perigraft period of immunosuppression could facilitate graft take in patients receiving bilayered living-cell therapy. Additional research to increase understanding of the mode of action and optimal use of this treatment modality is needed.
Conclusion
The case of a 48-year-old man diagnosed with ecthyma gangrenosum indicates that bilayered living-cell therapy can persist well beyond the previously reported 1 to 2 months in skin tissues of patients without underlying cutaneous disease. The question of how long and under what circumstances bilayered living-cell therapy persists is as yet unanswered. The presence of donor DNA at 10 months post treatment suggests that, in rare cases, bilayered living-cell therapy may function similar to autografting. It will be important to explore whether immunosuppression at the time of graft application influences the persistence of graft donor DNA.
Acknowledgments
The authors thank Laurie LaRusso, MS, ELS, for writing and editorial contributions to this report. In addition, the authors thank Janet Harden-Young, PhD, for the HLA testing.
1. Falabella AF, Valencia IC, Eaglstein WH, Schachner LA. Tissue-engineered skin (Apligraf) in the healing of patients with epidermolysis bullosa wounds. Arch Dermatol. 2000;136(10):1225–1230.
2. Falanga V, Margolis D, Alvarez O, et al. Rapid healing of venous ulcers and lack of clinical rejection with an allogeneic cultured human skin equivalent. Human Skin Equivalent Investigators Group. Arch Dermatol. 1998;134(3):293–300.
3. Falanga V, Sabolinski M. A bilayered living skin construct (APLIGRAF) accelerates complete closure of hard-to-heal venous ulcers. Wound Repair Regen. 1999;7(4):201–207.
4. Shen J, Zhou L, Falanga V. Bioengineered skin. Int J Cosmet Surg Aesth Derm. 2003;5(1):69–75.
5. Veves A, Falanga V, Armstrong DG, Sabolinski ML. Graftskin, a human skin equivalent, is effective in the management of noninfected neuropathic diabetic foot ulcers: a prospective randomized multicenter clinical trial. Diabetes Care. 2001;24(2):290–295.
6. Zaulyanov L, Kirsner RS. A review of a bi-layered living cell treatment (Apligraf) in the treatment of venous leg ulcers and diabetic foot ulcers. Clin Interv Aging. 2007;2(1):93–98.
7. Hu S, Kirsner RS, Falanga V, Phillips T, Eaglstein WH. Evaluation of Apligraf persistence and basement membrane restoration in donor site wounds: a pilot study. Wound Repair Regen. 2006;14(4):427–433.
8. Phillips TJ, Manzoor J, Rojas A, et al. The longevity of a bilayered skin substitute after application to venous ulcers. Arch Dermatol. 2002;138(8):1079–1081.
9. Fivenson DP, Scherschun L, Choucair M, Kukuruga D, Young J, Shwayder T. Graftskin therapy in epidermolysis bullosa. J Am Acad Dermatol. 2003;48(6):886–892.
10. Saap LJ, Donohue K, Falanga V. Clinical classification of bioengineered skin use and its correlation with healing of diabetic and venous ulcers. Dermatol Surg. 2004;30(8):1095–1100.
11. Serena TS, Shultz G. Publication in progress. Data on file.













