Vascular Healing Early After Titanium-Nitride-Oxide-Coated Stent Implantation Assessed by Optical Coherence Tomography
Download a PDF of this article.
Abstract: Background. The efficacy and safety of titanium-nitride-oxide-coated bioactive stents (BASs) were demonstrated in prior studies. In a prospective registry, we sought to explore the extent of neointimal coverage of stent struts by optical coherence tomography (OCT) at 30 days following the implantation of BASs in an unselected non-diabetic population. Methods. We enrolled 20 consecutive non-diabetic patients who underwent BAS implantation. OCT images were obtained at 30-day follow-up. Binary stent strut coverage was defined as the number of covered struts as a percentage of all analyzed struts. Results. Patients underwent OCT examination at an average of 30.5 ± 5.7 days following stent implantation. In these, 411 cross-sections were analyzed, including 3780 struts. Binary stent strut coverage was 97.2%, and the prevalence of malapposed struts was 3.2%. Mean neointimal thickness was 109.7 ± 83.6 µm. Conclusions. In the current evaluation by OCT at 30-day follow-up after BAS implantation in an unselected non-diabetic cohort, binary stent strut coverage was satisfactory and the prevalence of malapposed struts was low.
J INVASIVE CARDIOL 2013;25(4):186-189
Key words: optical coherence tomography, titanium, bioactive stents, neointimal coverage
____________________________________________
Recently, the safety of titanium-nitride-oxide-coated bioactive stents (BASs) has been established in several reports from real-life unselected populations.1,2 Interestingly, some reports demonstrated a better outcome with BASs as compared with paclitaxel-eluting stents in high-risk patients with complex lesions,3 and in patients presenting with acute myocardial infarction.4
Neointimal healing response following stent implantation largely depicts long-term clinical and angiographic outcomes. In this respect, recent concerns have focused on incomplete neointimal coverage of stent struts, which may ultimately portend the risk of stent thrombosis (ST).5,6 Neointimal healing response can be evaluated in vivo by means of invasive imaging techniques. The cutting-edge technology of optical coherence tomography (OCT) ensures an axial resolution of 10-15 µm which enables a highly accurate assessment of stent strut coverage. Evidence supports that stent strut coverage measured by OCT correlates well with histological neointimal healing after stenting in animal models.7,8 Reasonably, OCT has become the state-of-the-art modality for evaluation of neointimal coverage in studies comparing different stent types.9,10
In a prospective registry, we sought to explore the extent of neointimal coverage of stent struts by OCT at 30 days after implantation of a titanium-nitride-oxide-coated BAS in an unselected non-diabetic population.
Methods
Patient selection and study design. Prospectively, we enrolled 20 consecutive patients with symptomatic coronary artery disease amenable for percutaneous coronary intervention (PCI), who received the Titan2 titanium-nitride-oxide-coated BAS (Hexacath). We considered patients eligible for enrollment if they were older than 18 years, with at least one significant coronary lesion (defined as at least 50% diameter stenosis by visual estimation) in a native coronary artery. The main exclusion criteria included diabetes mellitus, unprotected left main or aorto-ostial lesions, in-stent restenosis, required stent length >28 mm, and contraindication to aspirin, clopidogrel, or heparin. All patients underwent follow-up coronary angiography with OCT examination of the index vessel at an average of 1 month. Treatment of more than 1 vessel was permissible. Before inclusion, an informed written consent was obtained from each patient after full explanation of the study protocol. The study protocol was reviewed and approved by our Institutional Human Research Committee and conforms to the ethical guidelines of the 1964 Declaration of Helsinki, as revised in 2002.
Optical coherence tomography image acquisition. OCT images were obtained with the C7-XR frequency-domain system (LightLab Imaging, Inc) employing the non-occlusive technique as described elsewhere.11 OCT images were analyzed offline in a core laboratory independently by two experienced investigators blinded to patient baseline, angiographic, and procedural data, employing the proprietary software (OCT system software B.0.1; LightLab Imaging, Inc)
Optical coherence tomography image analysis. Stent strut coverage, strut apposition, neointimal hyperplasia (NIH), and possible thrombosis were evaluated at 1 mm intervals (every fifth frame) in cross-sectional images. All cross-sectional images were initially screened for quality assessment, and if the image quality was insufficient to allow reliable measurements, a subsequent (or preceding) cross-section with adequate quality was evaluated.
The apposition status and tissue coverage of each analyzed strut was classified on a binary scale, thus creating four categories of struts: (a) apposed and covered; (b) apposed and uncovered; (c) malapposed and covered; and (d) malapposed and uncovered. Struts overlying an ostium of a side branch were labeled as non-apposed side-branch struts and excluded from the analysis. Struts were classified as uncovered if any part of the strut was exposed to the lumen with no visible tissue coverage, or covered if a layer of tissue was visible over the reflecting surfaces. For covered struts, the NIH thickness was measured. Binary stent strut coverage was defined as the percentage of covered struts of all analyzed struts. Incomplete stent apposition (ISA) distance was measured for protruding struts as previously described.11 We adopted ISA distance of 110 µm to define malapposition, by adding a margin of 18 µm as a correction for half of the blooming effect12 to the 91 µm strut thickness of BAS. The sum was rounded up to a full 10 microns regarding the axial resolution of frequency-domain OCT.
Lumen and stent contours were traced manually or semi-automatically and the NIH area was calculated by subtracting the lumen cross-sectional area (CSA) from the stent CSA. The percent NIH area was calculated by dividing the NIH area by the stent CSA multiplied by 100. If the lumen or stent CSAs were not measurable, they were omitted.
Statistical analysis. Continuous variables were presented as mean ± standard deviation, while categorical variables were described with absolute and relative (percentage) frequencies. Continuous variables such as stent area, lumen area, NIH area and NIH thickness were estimated as medians which were used for analysis. This has been done because the number of measurements of stent area and lumen area for each stent was rather small (mean, 17 ± 6; median, 17; range, 4 to 29). Pooled analysis of measurements was performed using Meta-analyst Beta 3.13 software (https://tuftscaes.org/meta_analyst/) in order to account for clustering in view of the large number of measurements obtained by OCT, and hence to get a better estimation of binary stent strut coverage, the frequency of malapposed struts, and NIH thickness. The results of pooled analysis were expressed as pooled proportions (%). Because heterogeneity was anticipated in observational studies, it was assessed a priori by a random effects model (DerSimonian-Laird). Inter-observer variability was assessed by evaluating 50 random cross-sectional images by two independent investigators. Statistical analysis was performed using SPSS statistical software (SPSS version 16.0.1; SPSS, Inc).
Results
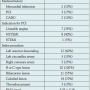 A total of 20 patients underwent OCT examination at an average of 30.5 ± 5.7 days following BAS implantation (Table 1). In these, a total of 411 cross-sections were analyzed, including 3780 struts, an average of 9.2 ± 3.1 struts per cross-section (Table 2). The mean age was 65 ± 13 years, and 85% were male. No clinical events were observed during the period from stent implantation to the time of follow-up.
A total of 20 patients underwent OCT examination at an average of 30.5 ± 5.7 days following BAS implantation (Table 1). In these, a total of 411 cross-sections were analyzed, including 3780 struts, an average of 9.2 ± 3.1 struts per cross-section (Table 2). The mean age was 65 ± 13 years, and 85% were male. No clinical events were observed during the period from stent implantation to the time of follow-up.
Binary stent strut coverage was 97.2%, and the frequency of malapposed struts was 3.2%. No thrombi were detected by OCT at this stage. On average, NIH thickness was 109.7 ±  83.6 µm; NIH area, 0.86 ± 0.46 mm2; percent NIH area, 14.2 ± 8.2%. Pooled analyses showed that the mean NIH thickness was 103 µm (95% confidence interval [CI], 85-125). Inter-observer variability of the same cross-section measurements of NIH thickness was 6 ± 9 µm. In addition, analysis of stent strut apposition and coverage was highly reproducible.
83.6 µm; NIH area, 0.86 ± 0.46 mm2; percent NIH area, 14.2 ± 8.2%. Pooled analyses showed that the mean NIH thickness was 103 µm (95% confidence interval [CI], 85-125). Inter-observer variability of the same cross-section measurements of NIH thickness was 6 ± 9 µm. In addition, analysis of stent strut apposition and coverage was highly reproducible.
Discussion
In the current short-term follow-up by OCT to evaluate neointimal stent strut coverage at 30 days following BAS implantation in an unselected sample of 20 non-diabetic patients, binary stent strut coverage was satisfactory (97.2%), and the prevalence of malapposed struts was quite low (3.2%).
Accumulating evidence supports the prime role of vascular neointimal healing in the prevention of ST following PCI, a fatal complication of real-life clinical practice. Stent struts directly exposed to the blood stream provide a favorable substrate for the occurrence of ST, a fact supported by pathological studies that unveiled deficient tissue healing in cases of late ST associated with drug-eluting stents (DESs).5,6 Additionally, exposed struts, particularly malapposed ones, may probably result in flow disturbance that would, in turn, create a prothrombotic milieu.13 An issue of immense clinical interest is the occurrence of late and very late (after 1 year) ST, well beyond the time covered by dual-antiplatelet therapy as recommended by the most recent guideline updates.14 In this context, a meta-analysis performed by Stone et al pooled data from 9 randomized trials (5261 patients) comparing DES versus bare-metal stents (BMSs). They reported an incidence of ST almost identical between the two stent types during the first year of follow-up (0.6%). Nevertheless, between 1 and 4 years, that incidence was much higher with DES than BMS (0.5% vs 0.1% per year, respectively).15
In previous studies, the binary stent strut coverage has ranged between 84%-99% for DESs at 3-13 months of follow-up,9,10,16-22 and from 98%-99% for BMS at 6-13 months of follow-up.10,21 Long-term OCT follow-up showed excellent binary stent strut coverage for BASs when compared to paclitaxel-eluting stents (99.6 vs 89.2%; P<.001) in patients treated for acute myocardial infarction.23 However, there are few data on very early stent healing. We have previously observed a binary stent strut coverage of 95% after 30 days from implantation of the endothelial progenitor cell (EPC) capturing stent, which is designed to promote endothelialization with antibody-coating.11 Thus, the endothelial coverage of the BAS appears to be more complete when compared to the EPC-capturing stent at this stage. Based on the limited data available, the binary stent strut coverage of BASs as early as 30 days following the implantation would seem far superior than that demonstrated with DESs, and quite comparable with that of BMSs. Yet, the frequency of malapposed struts (3.2%) was somewhat higher than that reported for various DESs (0.2%-1.8%) at 9-13 month follow-up in post hoc analysis of two large randomized controlled trials9,22 or when compared to EPC-capturing stent at 30 days (2.4%).11 This apparent divergence of findings might better be viewed in light of the exceptionally high frequency of patients presenting with acute coronary syndrome (80%) in this rather small-sized cohort in contrast to the lower frequency in the two all-comer randomized trials (40%-55%).9,22 Several mechanisms have been put forth to explain the mysterious occurrence of incomplete stent apposition following DES implantation.5,24 Among these, dissolution of plaque and thrombus behind the stent, leaving a gap between the stent and the vessel wall, especially following PCI for acute ST-segment elevation myocardial infarction. In addition, positive arterial remodelling with an increase in total lumen area might occur, so that the vessel tends to pull away from the stent. On the background of this rather high frequency of malapposed struts, a possible confounding effect on binary stent strut coverage cannot adequately be ruled out in the current registry.
Clinical implications. In view of the recent unsettling reports of ST,25-28 contemporary stent technologies are competitively geared toward negating the root causes of deficient neointimal coverage, the most powerful surrogate of such a grave event. Attempts to quantify ST, a rare-by-nature event, would intuitively require enrolling thousands of patients and protracting the length of follow-up for many years, in order to demonstrate a statistically perceptible difference in clinical outcome. In this realm, surrogate endpoints may be particularly indispensable. The high early binary stent strut coverage demonstrated in the current registry would be an attractive hallmark of BAS, and warrants further emphasis in future research, as well as in real-world practice. Interestingly, some comparative trials showed a better cumulative incidence of major events (and similar restenosis rates) for BASs as compared with paclitaxel-eluting stents in high-risk patients with complex lesions,3 and in patients presenting with acute myocardial infarction.4 In an OCT substudy of the latter trial, the BAS was superior to the paclitaxel-eluting stent with regard to binary stent strut coverage (99.6 vs 89.2%; P<.001) and number of malapposed struts (0.2 vs 13.8%; P<.001).23 In addition to this, small thrombi attached to uncovered struts were seen on 2 patients in the PES but not in the BAS group.23 Achievement of the goal of reducing restenosis rates following stent implantation without taking the risk to suffer a life-threatening “hard” endpoint would be a promising strategy to combat in-stent restenosis. Furthermore, unlike DESs, which need maintenance dual-antiplatelet therapy for at least 12 months (sometimes even for more extended periods), this therapy is required for no more than 1 month with BAS.
Study limitations. Our findings were based on a single-center registry with a relatively small sample size of the cohort, thus the conclusions based on these findings should be taken with caution. Further studies with long-term follow-up are required to evaluate the correlation of early stent endothelialization and clinical events. Despite the high resolution of OCT technique allowing adequate visualization of tissue surrounding the stent struts, the composition of such tissue, however, is far from clearly discernible, particularly around malapposed struts. In this regard, the presence of fibrin and/or thrombotic tissue overlying the struts might be mistaken for neointimal coverage, reducing the specificity of the technique. Alternatively, a thin rim of neointima covering the struts may fall beyond the resolution of the currently commercially available OCT imaging catheters (10-20 µm), and is therefore easily overlooked, compromising the sensitivity.
Finally, images were evaluated at 1 mm longitudinal intervals. Although this methodology has been experimentally validated for the assessment of neointimal strut coverage and showed an excellent reproducibility, it might have a lower sensitivity to detect uncovered struts than shorter-interval protocols.
Conclusion
In the current evaluation by OCT at 30-day follow-up after BAS implantation in an unselected non-diabetic cohort, binary stent strut coverage was satisfactory and the prevalence of malapposed struts was low.
References
- Karjalainen PP, Ylitalo A, Airaksinen KE. Real world experience with the TITAN stent: a 9-month follow-up report from the Titan PORI registry. Eurointervention. 2006;2(2):187-191.
- Mosseri M, Miller H, Tamari I, et al. The titanium-NO stent: results of a multicenter registry. Eurointervention. 2006;2(2):192-196.
- Karjalainen PP, Ylitalo A, Airaksinen JK, Nammas W. Five-year clinical outcome of titanium-nitride-oxide-coated bioactive stent implantation in a real-world population: a comparison with paclitaxel-eluting stents: the PORI registry. J Intervent Cardiol. 2011;24(1):1-8.
- Karjalainen PP, Ylitalo A, Niemela M, et al. Two-year follow-up after percutaneous coronary intervention with titanium-nitride-oxide-coated stents versus paclitaxel-eluting stents in acute myocardial infarction. Ann Med. 2009;21(8):1-9.
- Joner M, Finn AV, Farb A, et al. Pathology of drug-eluting stents in humans: delayed healing and late thrombotic risk. J Am Coll Cardiol. 2006;48(1):193-202.
- Finn AV, Joner M, Nakazawa G, et al. Pathological correlates of late drug-eluting stent thrombosis: strut coverage as a marker of endothelialization. Circulation. 2007;115(118):2435-2441.
- Murata A, Wallace-Bradley D, Tellez A, et al. Accuracy of optical coherence tomography in the evaluation of neointimal coverage after stent implantation. JACC Cardiovasc Imaging. 2010;3(1):76-84.
- Templin C, Meyer M, Muller MF, et al. Coronary optical frequency domain imaging (OFDI) for in vivo evaluation of stent healing: comparison with light and electron microscopy. Eur Heart J. 2010;31(14):1792-1801.
- Barlis P, Regar E, Serruys PW, et al. An optical coherence tomography study of a biodegradable vs. durable polymer coated limus-eluting stent: a LEADERS trial sub-study. Eur Heart J. 2010;31(2):165-176.
- Guagliumi G, Sirbu V, Bezerra H, et al. Strut coverage and vessel wall response to zotarolimus-eluting and bare-metal stents implanted in patients with ST-segment elevation myocardial infarction: the OCTAMI (Optical Coherence Tomography in Acute Myocardial Infarction) study. JACC Cardiovasc Interv. 2010;3(6):680-687.
- Lehtinen T, Kiviniemi TO, Ylitalo A, Mikkelsson J, Airaksinen KEJ, Karjalainen PP. Early vascular healing after endothelial progenitor cell capturing stent implantation. J Invasive Cardiol. 2012;24(12):631-635.
- Gonzalo N, Garcia-Garcia HM, Serruys PW, et al. Reproducibility of quantitative optical coherence tomography for stent analysis. Eurointervention. 2009;5(2):224-232.
- Davies PF, Spaan JA, Krams R. Shear stress biology of the endothelium. Ann Biomed Eng. 2005;33(12):1714-1718.
- Kushner FG, Hand M, Smith SC Jr, et al. focused updates: ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarction (updating the 2004 guideline and 2007 focused update) and ACC/AHA/SCAI guidelines on percutaneous coronary intervention (updating the 2005 guideline and 2007 focused update): a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2009;54(23):2205-2241.
- Stone GW, Moses JW, Ellis SG, et al. Safety and efficacy of sirolimus- and paclitaxel-eluting coronary stents. N Engl J Med. 2007;356(10):998-1008.
- Motreff P, Souteyrand G, Levesque S, et al. Comparative analysis of neointimal coverage with paclitaxel and zotarolimus drug-eluting stents, using optical coherence tomography 6 months after implantation. Arch Cardiovasc Dis. 2009;102(8-9):617-624.
- Kim J-S, Jang I-K, Fan C, et al. Evaluation in 3 months duration of neointimal coverage after zotarolimus-eluting stent implantation by optical coherence tomography. The ENDEAVOR OCT trial. JACC Cardiovasc Interv. 2009;2(12):1240-1247.
- Fan C, Kim J-S, Lee JM, et al. Different vascular healing patterns with various drug-eluting stents in primary percutaneous coronary intervention for ST-segment elevation myocardial infarction: optical coherence tomographic findings. Am J Cardiol. 2010;105(7):972-976.
- Kim J-S, Kim J-S, Kim TH, et al. Comparison of neointimal coverage of sirolimus-eluting stents and paclitaxel-eluting stents using optical coherence tomography at 9 months after implantation. Circ J. 2010;74(2):320-326.
- Li S, Wang Y, Gai L, et al. Evaluation of neointimal coverage and apposition with various drug-eluting stents over 12 months after implantation by optical coherence tomography. Int J Cardiol. 2013;162(3):166-171. 2011 Jun 2 (Epub ahead of print).
- Guagliumi G, Costa MA, Sirbu V, et al. Strut coverage and late malapposition with paclitaxel-eluting stents compared with bare metal stents in acute myocardial infarction: optical coherence tomography substudy of the Harmonizing Outcomes With Revascularization and Stents in Acute Myocardial Infarction (HORIZONS-AMI) Trial. Circulation. 2011;123(3):274-281.
- Gutiérrez-Chico JL, van Geuns RJ, Regar E, et al. Tissue coverage of a hydrophilic polymer-coated zotarolimus-eluting stent vs. a fluoropolymer-coated everolimus-eluting stent at 13-month follow-up: an optical coherence tomography substudy from the RESOLUTE All Comers trial. Eur Heart J. 2011;32(19):2454-2463.
- Lehtinen T, Airaksinen KE, Ylitalo A, Karjalainen PP. Stent strut coverage of titanium-nitride-oxide coated stent compared to paclitaxel-eluting stent in acute myocardial infarction: TITAX-OCT study. Int J Cardiovasc Imaging. 2012;28(8):1859-1866.
- Cook S, Wenaweser P, Togni M, et al. Incomplete stent apposition and very late stent thrombosis after drug-eluting stent implantation. Circulation. 2007;115(18):2426-2434.
- Morice MC, Serruys PW, Barragan P, et al. Long-term clinical outcomes with sirolimus-eluting coronary stents: five-year results of the RAVEL trial. J Am Coll Cardiol. 2007;50(14):1299-1304.
- Lagerqvist B, James SK, Stenestrand U; SCAAR Study Group. Long-term outcomes with drug-eluting stents versus bare-metal stents in Sweden. N Engl J Med. 2007;356(10):1009-1019.
- Nordmann AJ, Briel M, Bucher HC. Mortality in randomized controlled trials comparing drug-eluting vs. bare metal stents in coronary artery disease: a meta-analysis. Eur Heart J. 2006;27(23):2784-2814.
- Garg P, Mauri L. The conundrum of late and very late stent thrombosis following drug-eluting stent implantation. Curr Opin Cardiol. 2007;22(6):565-571.
______________________________________________
*Joint first authors.
From the 1Heart Center, Satakunta Central Hospital, Pori, Finland and 2Department of Medicine, Turku University Hospital, Turku, Finland.
Funding: This study was supported by grants (TL) from the Aarne Koskelo Foundation, Helsinki, Finland and Paavo Nurmi Foundation, Helsinki, Finland.
Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. Dr Kiviniemi reports a grant with Astra Zeneca and travel reimbursement from Boston Scientific, St Jude Medical, and Boehringer Ingelheim. No other authors report conflicts of interest regarding the content herein.
Manuscript submitted October 21, 2012, provisional acceptance given October 29, 2012, final version accepted January 18, 2013.
Address for correspondence: Pasi P. Karjalainen, MD, Heart Center, Satakunta Central Hospital, Sairaalantie 3, Pori, Finland. Email: pasi.karjalainen@satshp.fi












