Successful Management of Lower Extremity Distal Embolization Following Percutaneous Atherectomy with the JetStream G3 Device
ABSTRACT: There has been a resurgence of atherectomy as a strategy to treat peripheral atherosclerosis. Distal embolization occurs frequently during peripheral endovascular interventions but is of particular concern during atherectomy. We describe a case of distal embolization following atherectomy with the Pathway JetStream system successfully managed with aspiration thrombectomy and infrapopliteal angioplasty. A detailed review of the incidence, treatment, and prevention of DE in this context are presented.
J INVASIVE CARDIOL 2012;24(6):E124-E128
_______________________________________
Over the past decade there has been an increased interest in the development of novel endovascular therapies for the treatment of lower extremity peripheral arterial disease (PAD). Plaque excision with atherectomy is an acceptable method of revascularization and is growing in popularity given the increased selection of devices and their relative ease of use. Although the acute angiographic results are often impressive following percutaneous atherectomy, the true efficacy of this approach in comparison to traditional angioplasty and stenting remains to be determined. Underlying effective plaque removal are the potential risks of distal embolization (DE). DE occurs frequently during endovascular procedures throughout the vasculature and can have significant adverse consequences. The purpose of the present review is to outline a case of DE following percutaneous atherectomy of a superficial femoral artery (SFA) and to review the literature concerning the incidence, clinical associations, and prevention of this complication.
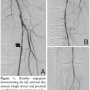 Case Report. A 58-year-old male with a long-standing history of tobacco abuse, diabetes, hypertension, and coronary artery disease with prior coronary artery bypass grafting presented with complaints of lifestyle limiting claudication. After ankle brachial indices confirmed the diagnosis of PAD, the patient underwent a bilateral lower extremity angiogram. The patient had diffuse atherosclerosis involving both legs with a chronic total occlusion of the right SFA. The left external artery demonstrated a focal 90% stenosis and moderately calcified long proximal and distal areas of stenosis (70%) in the left SFA (Figures 1A and 1B). The left infrapopliteal circulation demonstrated 3 patent vessels with a peroneal artery that tapered off at the mid tibia (Figure 1C).
Case Report. A 58-year-old male with a long-standing history of tobacco abuse, diabetes, hypertension, and coronary artery disease with prior coronary artery bypass grafting presented with complaints of lifestyle limiting claudication. After ankle brachial indices confirmed the diagnosis of PAD, the patient underwent a bilateral lower extremity angiogram. The patient had diffuse atherosclerosis involving both legs with a chronic total occlusion of the right SFA. The left external artery demonstrated a focal 90% stenosis and moderately calcified long proximal and distal areas of stenosis (70%) in the left SFA (Figures 1A and 1B). The left infrapopliteal circulation demonstrated 3 patent vessels with a peroneal artery that tapered off at the mid tibia (Figure 1C).
 The decision was made to first treat the left SFA and iliac lesions and then to later stage the right SFA chronic total occlusion. Access was obtained in the right femoral artery and using the peripheral cross over technique, a 7 Fr Ansel sheath was placed in the left external iliac artery. Intravenous heparin was used for anticoagulation to achieve a peak activated clotting time of over 300 seconds. The patient was on chronic aspirin and clopidogrel therapy. A 0.014-in, 300 cm long, 3.0-gram tip weight MiracleBros (Abbott Vascular) wire was
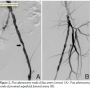
The decision was made to first treat the left SFA and iliac lesions and then to later stage the right SFA chronic total occlusion. Access was obtained in the right femoral artery and using the peripheral cross over technique, a 7 Fr Ansel sheath was placed in the left external iliac artery. Intravenous heparin was used for anticoagulation to achieve a peak activated clotting time of over 300 seconds. The patient was on chronic aspirin and clopidogrel therapy. A 0.014-in, 300 cm long, 3.0-gram tip weight MiracleBros (Abbott Vascular) wire was 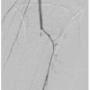 positioned across the left SFA lesions into the left tibioperoneal trunk. A Pathway G3 JetStream atherectomy device (Pathway Medical Technologies, Inc.) was first used to perform atherectomy runs across the left external iliac lesion. Four sequential runs (with the latter 2 runs with the cutting blades extended) was performed and subsequent angiography revealed an excellent result with 20%-30% residual stenosis and no dissection (Figure 2A). The proximal left SFA lesion was then treated with the
positioned across the left SFA lesions into the left tibioperoneal trunk. A Pathway G3 JetStream atherectomy device (Pathway Medical Technologies, Inc.) was first used to perform atherectomy runs across the left external iliac lesion. Four sequential runs (with the latter 2 runs with the cutting blades extended) was performed and subsequent angiography revealed an excellent result with 20%-30% residual stenosis and no dissection (Figure 2A). The proximal left SFA lesion was then treated with the 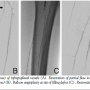 atherectomy system using 8 total runs (3 with blades down and 5 with blades extended). Audible decelerations were noted during the first 3 extended blade runs suggesting highly fibrotic or calcified plaque. Post atherectomy demonstrated significant debulking of plaque (Figure 2B). However, repeat angiography of the below the knee circulation now revealed acute occlusion of the left tibioperoneal trunk (Figure 3). To augment antiplatelet activity, intravenous eptifibatide was started. The 3-gram MiracleBros wire was redirected into the peroneal artery. A second 3-gram MiracleBros wire was directed through the occluded posterior tibial artery. Sequential aspiration thrombectomy was performed using a ProntoLP catheter (Vascular Solutions, Inc.) (Figure 4A), first through the tibioperoneal trunk into the peroneal artery and then similarly into the posterior tibial artery. The aspirate did not reveal any visible thrombus or macroscopic atherosclerotic debris but there was some improvement in flow through the posterior tibial artery (Figure 4B). Focal balloon angioplasty of the tibioperoneal trunk was performed using the 3 mm x 15 mm Maverick balloon (Boston Scientific) for 2 minutes (Figure 4C) resulting in restoration of flow in the infrapopliteal circulation (Figures 4D and 4E). The remaining mid SFA disease was treated with percutaneous transluminal angioplasty (PTA) and stenting. The patient was maintained on a heparin drip overnight. The following day, the patient demonstrated no signs of acute foot ischemia and both the posterior tibial and dorsalis pedis pulses were palpable.
atherectomy system using 8 total runs (3 with blades down and 5 with blades extended). Audible decelerations were noted during the first 3 extended blade runs suggesting highly fibrotic or calcified plaque. Post atherectomy demonstrated significant debulking of plaque (Figure 2B). However, repeat angiography of the below the knee circulation now revealed acute occlusion of the left tibioperoneal trunk (Figure 3). To augment antiplatelet activity, intravenous eptifibatide was started. The 3-gram MiracleBros wire was redirected into the peroneal artery. A second 3-gram MiracleBros wire was directed through the occluded posterior tibial artery. Sequential aspiration thrombectomy was performed using a ProntoLP catheter (Vascular Solutions, Inc.) (Figure 4A), first through the tibioperoneal trunk into the peroneal artery and then similarly into the posterior tibial artery. The aspirate did not reveal any visible thrombus or macroscopic atherosclerotic debris but there was some improvement in flow through the posterior tibial artery (Figure 4B). Focal balloon angioplasty of the tibioperoneal trunk was performed using the 3 mm x 15 mm Maverick balloon (Boston Scientific) for 2 minutes (Figure 4C) resulting in restoration of flow in the infrapopliteal circulation (Figures 4D and 4E). The remaining mid SFA disease was treated with percutaneous transluminal angioplasty (PTA) and stenting. The patient was maintained on a heparin drip overnight. The following day, the patient demonstrated no signs of acute foot ischemia and both the posterior tibial and dorsalis pedis pulses were palpable.
Discussion
Incidence of DE. DE has long been recognized as a potential complication of endovascular therapy for lower extremity PAD. Shrikhande et al examined data from a prospective database to investigate the incidence and predictors of angiographically evident DE (defined as change in any visualized runoff vessel other than vasospasm and dissection at any time during the procedure) during percutaneous intervention of lower extremity lesions.1 In this study, 2137 lesions were treated with PTA with (34.6%) or without (26.7%) stent placement, SilverHawk (ev3, Inc) directional atherectomy (34.4%), DiamondBack 360 (CSI) orbital atherectomy (0.8%), Pathway JetStream (Pathway Medical) rotational atherectomy (0.8%), or Excimer laser atherectomy (2.6%). Overall, there were 34 embolic events resulting in a DE rate of 1.6%. The rates of DE from PTA alone (0.9%), PTA with stent placement (0.7%), SilverHawk atherectomy (1.9%), and Excimer atherectomy (3.6%) were not significantly different. However, DiamondBack (22%) and JetStream (22%) atherectomy resulted in significantly higher rates of DE than the other therapies. When lesion characteristics were analyzed, there were significantly higher rates of DE during treatment of in-stent restenosis and totally occluded lesions compared to de novo lesions. There were also significantly higher rates in TASC C/D lesions compared to TASC A/B lesions. The rates of angiographically detected DE were not different based on preoperative runoff status and lesion calcification was not addressed.
Even without angiographic findings, debris liberation during endovascular therapy appears to be an ubiquitous process. Inspection of distal protection filters reveals macroscopic debris at least 25% of the time following angioplasty (with or without stenting) and up to 90% of the time following atherectomy.2 In addition to macroscopic debris, which can impact the angiographic flow, each stage of therapy from wire passage to balloon dilation and stenting has the potential to liberate smaller particles. Therefore, nearly all endovascular procedures produce some degree of microscopic DE. Using Doppler signals, Lam et al evaluated the incidence of microvascular emboli following varying stages and methods of lower extremity revascularization, including wire crossing, angioplasty, stent deployment, atherectomy with the SilverHawk device, and laser atherectomy.3 Although the subject number was small (n=60), the authors noted a graded increase in embolic signals (ES) from wire crossing to angioplasty to stent deployment. There appeared to be a higher ES count during plaque excision either with the SilverHawk device or with laser versus other therapies, although this finding did not reach statistical significance, likely secondary to a small sample size. Interestingly, ES were also detected between device passages with the atherectomy devices. Taken together these findings suggest that treatments which are highly plaque disrupting/modifying such as stenting and atherectomy are much more likely to result in DE. The clinical significance of DE, particularly of microembolic debris, is difficult to assess in the lower extremity circulation. This is due, in part, to the lack of immediately observable sequelae of embolic complications as compared to the coronary and cerebral vasculature.
 Specific Atherectomy Devices. As interventional techniques and devices have advanced, the spectrum of lesions treated percutaneously has continually grown to include more complex disease. Modern atherectomy devices have shown promise in treating a wide range of lesions, including total occlusions, calcified plaque, and long lesions.4,5 Directional atherectomy (SilverHawk) uses a retractable, rotating blade to excise plaque and pack the material into a nosecone during catheter advancement over a guidewire. Because the debris is removed within a single plane during each pass, the device is rotated as needed based on the circumferential nature of the plaque. These devices have the capability of excising and capturing large segments of plaque intact, which may be examined with clearance of the nosecone after removal of the catheter (Figure 5). In contrast, rotational atherectomy uses a burr on the end of a drive shaft that rotates at a fixed diameter to abrade plaque into matter small enough to pass through the microcirculation, thereby avoiding clinically relevant embolization. Significant advantages have been found in treating calcified lesions in the coronary and peripheral circulations. A newer generation rotational atherectomy device, the JetStream G3 System (Pathway Medical), includes an aspiration port for simultaneous removal of debris as it modifies plaque. The other mechanical device currently on the market utilizes the principle of orbital atherectomy (DiamondBack 360).6 Orbital atherectomy regulates the debulking area by altering the rotational speed of an eccentric crown in order to maximize and control luminal gain. Laser atherectomy attempts to dissolve obstructive plaque into particles that pass through the microcirculation using a focused laser for photoablation. Spectranetics currently markets the Turbo-Elite laser atherectomy system for treatment of infrainguinal peripheral arterial disease. While the DiamondBack, JetStream, and laser atherectomy systems attempt to capture the debris or grind/ablate it to a size that may pass through the microcirculation, the potential for DE still remains as was confirmed by the study by Shrikhande et al discussed above.1
Specific Atherectomy Devices. As interventional techniques and devices have advanced, the spectrum of lesions treated percutaneously has continually grown to include more complex disease. Modern atherectomy devices have shown promise in treating a wide range of lesions, including total occlusions, calcified plaque, and long lesions.4,5 Directional atherectomy (SilverHawk) uses a retractable, rotating blade to excise plaque and pack the material into a nosecone during catheter advancement over a guidewire. Because the debris is removed within a single plane during each pass, the device is rotated as needed based on the circumferential nature of the plaque. These devices have the capability of excising and capturing large segments of plaque intact, which may be examined with clearance of the nosecone after removal of the catheter (Figure 5). In contrast, rotational atherectomy uses a burr on the end of a drive shaft that rotates at a fixed diameter to abrade plaque into matter small enough to pass through the microcirculation, thereby avoiding clinically relevant embolization. Significant advantages have been found in treating calcified lesions in the coronary and peripheral circulations. A newer generation rotational atherectomy device, the JetStream G3 System (Pathway Medical), includes an aspiration port for simultaneous removal of debris as it modifies plaque. The other mechanical device currently on the market utilizes the principle of orbital atherectomy (DiamondBack 360).6 Orbital atherectomy regulates the debulking area by altering the rotational speed of an eccentric crown in order to maximize and control luminal gain. Laser atherectomy attempts to dissolve obstructive plaque into particles that pass through the microcirculation using a focused laser for photoablation. Spectranetics currently markets the Turbo-Elite laser atherectomy system for treatment of infrainguinal peripheral arterial disease. While the DiamondBack, JetStream, and laser atherectomy systems attempt to capture the debris or grind/ablate it to a size that may pass through the microcirculation, the potential for DE still remains as was confirmed by the study by Shrikhande et al discussed above.1
Given its market share, comparatively greater data with respect to DE exist for the SilverHawk device. In 2006, data from the TALON registry was published on 601 patients with 1258 lesions treated with the SilverHawk device and followed prospectively.5 Plaque excision was planned in all subjects and adjunctive therapy was at the discretion of the treating physicians. Moderate/severe calcification was present in 65% of the lesions and 75% of the lesions were above the knee. A low DE event rate of 0.1% was reported. McKinsey et al reported a series of 275 patients with 579 femoral, popliteal, and/or tibial vessels treated with SilverHawk atherectomy.7 There were 291 lesions classified as TASC C/D (50.3%). SilverHawk plaque excision was used alone in 64.8% of lesions and with assisted or adjunctive PTA/stent in 35.2% of lesions. The degree of calcification was not mentioned; however, an embolic protection device was considered in the setting of severely calcified lesions. Procedural embolization was rare, occurring in 0.8% and treated successfully with manual aspiration. The authors commented that a technique of slow passes, frequent packing, and interruption of passes for packing likely contributed to their low embolization rate.
More recently, Minko et al published results from a prospective study of SilverHawk plaque excision in 42 highly calcified lesions.8 Predilation with a 3 mm balloon catheter was required in 2 patients and to achieve technical success (residual stenosis <30%), PTA/stenting was required in 12% of patients. There were 3 (7%) embolic events successfully treated with aspiration. Without commenting on the practice during the study, the authors speculated that frequent clearing of the SilverHawk atherectomy debris collection system might prevent DE during atherectomy of calcified lesions.
The routine use of distal protection in complex lesions undergoing endovascular therapy (PTA ± stenting or atherectomy) was evaluated by Shammas et al in the PROTECT registry.9 In the PROTECT registry the Spider FX (eV3) or EmboShield (Abbott Vascular) embolic protection device (EPD) was deployed during intervention. The lesions were located in the femoral, popliteal, or anterior tibial vessels and had at least one high-risk characteristic (41.1% mod/severe calcification, 26.8% total occlusion, 8.9% filling defect or thrombus, 62.5% irregular/ulcerated lesions at least 30 mm in length, or 17.9% “smooth” lesions at least 50 mm in length). There were 40 patients with 56 lesions; 43 lesions were treated with PTA with or without stenting and 13 lesions were treated with SilverHawk atherectomy and adjunctive PTA (treatment was at the discretion of the operator). Debris larger than dust-like particles (<2mm in length) and clinically significant macroembolization (≥ 2mm in length) occurred in 100% and 90.9% of the SilverHawk treated group, respectively, and in 37.9% and 27.6% in the non-atherectomy group, respectively. The logistic regression analysis identified SilverHawk treatment versus PTA ± stenting as an independent predictor of clinically significant DE (OR 31, 95% CI 3.31-300.34; P<0.003). Restenotic lesions were also predictive (OR 15, 95% CI 2.53-90.90; P<0.003) of significant DE compared to de novo lesions. The authors noted that a significantly higher number of restenotic lesions were treated with atherectomy, possibly contributing to the perceived risk of DE of restenotic lesions.
Given that it is a relatively newer device, there are fewer published reports/data regarding the JetStream Pathway rotational atherectomy. However, as with the SilverHawk, DE remains a potential concern with this technology. In a pilot study from December 2005 to February 2006, 15 patients with lesions in the SFA (47%), popliteal (47%), and posterior tibial (6%) arteries were treated with this rotational atherectomy system.10 Multiple safety endpoints and markers of procedural, clinical, and hemodynamic success were evaluated. One patient (out of the 15) had both a non-flowing limiting dissection treated with stent placement and DE successfully treated with aspiration. Subsequently, the same investigational group enrolled 172 patients for treatment with JetStream rotational atherectomy of SFA (64%), popliteal (27%), and tibial (9%) lesions.11 Moderate to heavy calcium was present in 51% of lesions, and 31% were total occlusions. There were 17 (9.9%) reported DE events, all successfully treated during the interventional procedure. The methods of diagnosis and treatment of the embolic events were not reported in detail. More recently, data has been presented that suggests that DE may be more frequent with the JetStream device as compared with Silverhawk. Boiangiu et al studied 22 patients who underwent atherectomy with the JetStream with distal embolic protection.12 The contents of the filter were analyzed for particle size and compared to data on embolic debris obtained from a separate cohort undergoing SilverHawk atherectomy. Macroscopic debris was retrieved in 21 of the 22 JetStream patients (95.4%). Large embolic debris (≥2 mm in longest axis) was found in 16 of the 22 patients (72.7%). The histological analysis of the debris revealed collagen material in 15 of 16 samples (93.7%), fibrin in 14 (87.5%), macrophages in 13 (81.2%), calcification in 10, (62.5%), and cholesterol-rich material in 6 (37.5%). Compared to the SilverHawk atherectomy cohort, clinically significant emboli were found more frequently with the JetStream device, 72.7% versus 46.7%. Though preliminary, these data coupled with those of Shrikhande1 and our empiric case report suggest that the active aspiration of the JetStream device may be inadequate to reliably prevent DE during plaque modification. Coupling of the JetStream atherectomy with distal embolic protection may be a prudent preventative strategy and may have been helpful in our case. Even fewer data are available at this time for the DiamondBack device in regards to DE, but similar considerations may be made for this technology as well.
Shammas et al examined DE following laser atherectomy using the SpiderFx filter in conjunction with Excimer Laser ablation in the DEEP EMBOLI registry.13 In 20 patients with at least moderate lesion calcification, total occlusions, a filling defect, irregular lesions at least 30 mm in length, or smooth lesions at least 50 mm in length underwent elective laser atherectomy with or without adjunctive angioplasty and stenting in the femoropopliteal or proximal tibial segments. The primary safety endpoints included filter-related complications and the presence of clinically significant macrodebris (classified as ≥ 2 mm in any dimension) in the filter upon retrieval. Macrodebris and clinically significant macrodebris were found in 67% and 22.2% of filters under laser treatment and 35% and 20% of filters during adjunctive therapy, respectively.
Treatment and Prevention Strategies. The characteristics of the embolic material are important to consider when discussing treatment and prevention of DE. Analysis of debris retrieved from distal embolic protection filters has demonstrated particle sizes of greater than 1 mm (major diameter) in more than 50% of cases and greater than 3 mm in 12% of cases.14 The embolic material retrieved from femoropopliteal interventions are composed mainly of platelets, fibrin, inflammatory cells, and extracellular matrix components. Smaller amounts of endothelial cells, foam cells, cholesterol, necrotic components, and calcified material are often also identified.
While there has been no systematic evaluation of treatment strategies for DE, individual reports have focused on mechanical aspiration of debris and/or disruption of thrombus with antiplatelet agents or catheter-directed thrombolytics.3,15,16 In contrast, to the coronary circulation where intra-arterial vasodilators may be helpful in treating and preventing microvascular dysfunction following revascularization, few data are available for their efficacy in the lower extremity vessels. The use of a manual aspiration catheter remains an option for angiographically evident macroembolization and has been shown to be efficacious in cases of DE.15 Given the high prevalence of thrombin and platelet rich material composing embolic debris, the use of focused glycoprotein 2b/3a inhibitors and tPA is a reasonable approach in conjunction with aspiration.16 For cases of large macrovascular obstruction, rheolytic thrombectomy could be considered as well.
Prevention of DE remains focused on protection devices placed distal to the target lesion. There are 2 main types of EPDs: balloon occlusive devices and filter devices. The balloon-based devices are navigated past the lesion and deployed distally to totally occlude the vessels. The intervention must then be performed either with a static contrast column or without real time contrast angiography. Aspiration is then performed to remove any debris in the vessel proximal to the occlusion balloon. It is possible that particles may travel passed the balloon if the occlusion device is not flush against the vessel wall or if the vessel size is altered with the intervention. Filter-based devices are also placed distal to the interventional site, the intervention is then undertaken, and the filter, with collected debris, is retracted back into a catheter for removal, all while maintaining flow. Disadvantages of the filter system include the potential for malapposition of the device against the vessel wall possibly allowing distal embolization around the edges of the device. Furthermore, the ability to deploy filters becomes limited as the target vessel size decreases. Retrieval of filters full of debris may also pose challenges during retrieval, necessitating manual aspiration of the contents prior to capture of the basket. There are also the added financial costs of these devices to consider, which may add at least $1000 per procedure.17,18
Lastly, the preventative use of EPDs is of increased importance in limbs with poor distal runoff, particularly given that particles collected in the filters may range in length from 1 mm-10 mm, and the tibial vessels generally range in size from 1 mm-3 mm, it is conceivable that EPDs may protect against acute occlusion.18,19 Balancing the smaller vessel size with the size of protection devices remains the challenge in below-the-knee circulation.
Conclusions
A review of the literature reveals angiographically evident DE rates as high as 22% during percutaneous atherectomy for lower extremity PAD and microembolic events likely occur in most cases. There is more reported experience with the SilverHawk directional atherectomy system, but the newer devices including the JetStream system may also liberate debris. While prompt treatment with manual aspiration and/or thrombolysis appears effective at treating DE, prevention with the use of an EPD may be a prudent choice. Despite the high rate of captured embolic debris in filters, the exact role of EPDs in preventing complications has yet to be determined. Conclusions from multiple reviews agree that when advanced or calcified disease is present, distal runoff is compromised, or atherectomy is planned, EPDs may prevent against adverse outcomes. Prediction of clinically significant DE for an individual patient or lesion remains challenging and uncertain at this time.
References
- Shrikhande GV, Khan SZ, Hussain HG, Dayal R, McKinsey JF, Morrissey N. Lesion types and device characteristics that predict distal embolization during percutaneous lower extremity interventions. J Vasc Surg. 2011;53(2):347-352.
- Müller-Hülsbeck S, Schäfer PJ, Hümme TH, et al. Embolic protection devices for peripheral application: wasteful or useful? J Endovasc Ther. 2009;16 Suppl 1:I163-169.
- Lam RC, Shah S, Faries PL, McKinsey JF, Kent KC, Morrissey NJ. Incidence and clinical significance of distal embolization during percutaneous interventions involving the superficial femoral artery. J Vasc Surg. 2007;46(6):1155-1159.
- Zeller T, Sixt S, Schwarzwalder U, et al. Two-year results after directional atherectomy of infrapopliteal arteries with the SilverHawk device. J Endovasc Ther. 2007;14(2):232-240.
- Ramaiah V, Gammon R, Kiesz S, et al. Midterm outcomes from the TALON Registry: treating peripherals with SilverHawk: outcomes collection. J Endovasc Ther. 2006;13(5):592-602.
- Korabathina R, Mody KP, Yu J, Han SY, Patel R, Staniloae CS. Orbital atherectomy for symptomatic lower extremity disease. Catheter Cardiovasc Interv. 2010;76(3):326-332.
- McKinsey JF, Goldstein L, Khan HU, et al. Novel treatment of patients with lower extremity ischemia: use of percutaneous atherectomy in 579 lesions. Ann Surg. 2008;248(4):519-528.
- Minko P, Katoh M, Jaeger S, Buecker A. Atherectomy of heavily calcified femoropopliteal stenotic lesions. J Vasc Interv Radiol. 2011;22(7):995-1000.
- Shammas NW, Coiner D, Shammas GA, Dippel EJ, Christensen L, Jerin M. Percutaneous lower-extremity arterial interventions with primary balloon angioplasty versus silverhawk atherectomy and adjunctive balloon angioplasty: randomized trial. J Vasc Interv Radiol. 2011;22(9):1223-1228.
- Zeller T, Krankenberg H, Rastan A, et al. Percutaneous rotational and aspiration atherectomy in infrainguinal peripheral arterial occlusive disease: a multicenter pilot study. J Endovasc Ther. 2007;14(3):357-364.
- Zeller T, Krankenberg H, Steinkamp H, et al. One-year outcome of percutaneous rotational atherectomy with aspiration in infrainguinal peripheral arterial occlusive disease: the multicenter pathway PVD trial. J Endovasc Ther. 2009;16(6):653-662.
- Boiangiu C, Fissha M, Kaid K, et al. Analysis of retrieved particulate debris after superficial femoral artery (SFA) atherectomy using the Pathway Jetstream G3® device. SCAI 2011 Scientific Sessions. Baltimore, Maryland, 2011.
- Shammas NW, Coiner D, Shammas GA, Christensen L, Dippel EJ, Jerin M. Distal embolic event protection using excimer laser ablation in peripheral vascular interventions: results of the DEEP EMBOLI registry. J Endovasc Ther. 2009;16(2):197-202.
- Karnabatidis D, Katsanos K, Kagadis GC, et al. Distal embolism during percutaneous revascularization of infra-aortic arterial occlusive disease: an underestimated phenomenon. J Endovasc Ther. 2006;13(3):269-280.
- Zafar N, Prasad A, Mahmud E. Utilization of an aspiration thrombectomy catheter (Pronto) to treat acute atherothrombotic embolization during percutaneous revascularization of the lower extremity. Catheter Cardiovasc Interv. 2008;71(7):972-975.
- Keo H, Diehm N, Baumgartner R, Husmann M, Baumgartner I. Single center experience with provisional abciximab therapy in complex lower limb interventions. Vasa. 2008;37(3):257-264.
- Mix JW, Stevens SL. Commentary. Lam RC, Shah S, Faries PL, McKinsey JF, Kent KC, Morrissey NJ. Incidence and clinical significance of distal embolization during percutaneous interventions involving the superficial femoral artery. J Vasc Surg. 2007;46:1155-1159. Perspect Vasc Surg Endovasc Ther. 2008;20(4):387-388.
- Wholey M. The role of embolic protection in peripheral arterial atherectomy. Tech Vasc Interv Radiol. 2011;14(2):65-74.
- Suri R, Wholey MH, Postoak D, Hagino RT, Toursarkissian B. Distal embolic protection during femoropopliteal atherectomy. Catheter Cardiovasc Interv. 2006;67(3):417-422.
_______________________________________
From the *University of California San Diego, Department of Medicine, Division of Cardiology, San Diego, California; †North Austin Medical Center, Austin Texas; and the **Department of Medicine, Division of Cardiology at the University of Texas Health Science Center at San Antonio, San Antonio, Texas.
Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. The authors report no conflicts of interest regarding the content herein.
Manuscript submitted November 21, 2011, provisional acceptance given December 28, 2011, final version accepted February 7, 2012.
Address for correspondence: Anand Prasad, MD, FACC, FSCAI, The University of Texas Health Science Center at San Antonio, Department of Medicine, Division of Cardiology, MC 7872, 7703 Floyd Curl Drive, San Antonio, Texas 78229. Email: anandprasadmd@gmail.com












