Percutaneous Focused Force Aortic Valvuloplasty Using the Buddy-Catheter Technique
Abstract: Percutaneous transcatheter aortic valvuloplasty is seriously limited by high restenosis rates. We report a case where the use of a buddy-catheter technique during balloon inflation allowed increased focused force onto calcified stiff commissures with an increase in resultant valve area. This technique may result in further valve area enlargement and decrease the restenosis rate.
J INVASIVE CARDIOL 2012;24(6):287-289
Key words: percutaneous transcatheter aortic valvuloplasty, TAVI, buddy-catheter technique
_____________________________________________
Currently, transcatheter aortic valve implantation (TAVI) is one of the most exciting developments in cardiovascular medicine.1,2 However, TAVI has not been available in Japan yet. Percutaneous transcatheter aortic valvuloplasty (PTAV) still remains one of the therapeutic methods as a “bridge” to surgery in hemodynamically unstable patients or high surgical risk patients with severe senile calcific aortic stenosis in adults.3 However, PTAV is limited by high restenosis rates.4 The pathologic mechanism of valvuloplasty for stenotic aortic valve is known to be splitting or cracking of fused commissures and aortic wall expansion (stretching) at non-fused commissure sites.5 One limitation of valvuloplasty is that balloon inflation at a stenotic aortic valve produces equal force at each of three commissures, resulting in insufficient force at the more calcified commissure. Selective deployment of a buddy catheter onto stiffer commissures during balloon inflation may produce focused force at the commissure and an additional effect to increase valve area. This case demonstrated a further valve area increase with the use of this buddy-catheter technique on two calcified commissures (between the right and left coronary cusps and between the right and non-coronary cusps).
Case Description
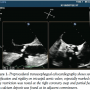 A 73-year-old man was admitted to our hospital with dyspnea. He was diagnosed with congestive heart failure due to severe aortic stenosis. Despite full inotropic and mechanical ventilator support, his condition did not improve and remained Forrester class IV. To improve hemodynamics, PTAV was performed via a retrograde femoral artery approach under mechanical ventilation support and tranesophageal echocardiography (TEE) guidance. TEE showed a severely stenotic tricuspid aortic valve with calcification of each leaflet
A 73-year-old man was admitted to our hospital with dyspnea. He was diagnosed with congestive heart failure due to severe aortic stenosis. Despite full inotropic and mechanical ventilator support, his condition did not improve and remained Forrester class IV. To improve hemodynamics, PTAV was performed via a retrograde femoral artery approach under mechanical ventilation support and tranesophageal echocardiography (TEE) guidance. TEE showed a severely stenotic tricuspid aortic valve with calcification of each leaflet 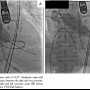 and commissure (Figure 1). Markedly reduced mobility was found at the right coronary cusp due to its extensive calcification and calcified stiffness of the adjacent commissures. Aortic annulus diameter was measured as 20.5 mm by TEE. Simultaneous aortic and left ventricular pressures before valvuloplasty showed peak and mean pressure gradient of 82.6 and 57.7 mm Hg, respectively, and aortic valve area (AVA) was calculated as 0.64 cm2 (Figure 2A). Following two 20 mm balloon inflations (Z-MED II; NuMED), peak and
and commissure (Figure 1). Markedly reduced mobility was found at the right coronary cusp due to its extensive calcification and calcified stiffness of the adjacent commissures. Aortic annulus diameter was measured as 20.5 mm by TEE. Simultaneous aortic and left ventricular pressures before valvuloplasty showed peak and mean pressure gradient of 82.6 and 57.7 mm Hg, respectively, and aortic valve area (AVA) was calculated as 0.64 cm2 (Figure 2A). Following two 20 mm balloon inflations (Z-MED II; NuMED), peak and 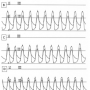 mean pressure gradients were improved to 35.4 and 28.8 mm Hg, respectively (Figure 2B). Because the 20 mm balloon was fluoroscopically undersized and no aortic insufficiency was identified by TEE, the balloon size was changed to 22 mm (TYSHAK; NuMed) and two further inflations were performed resulting in peak and mean gradients of 28.1 and 23.0 mm Hg, respectively (Figure 2C). However, the right coronary cusp was still immobile due to calcified stiffness of the adjacent commissures on TEE. Accordingly, a 5 Fr pigtail catheter with a 0.035˝ Amplatzer super-stiff wire (St Jude Medical) was inserted into the left ventricle and the 22 mm balloon was inflated beside the pigtail catheter, which was maneuvered toward each of the adjacent commissures of the right coronary cusp during balloon inflations under fluoro and TEE guidance (Figure 3). Final hemodynamics demonstrated a significant increase in AVA (peak gradient, 16.4 mm Hg; AVA, 1.41 cm2; Figure 2D) and TEE showed improved aortic valve mobility with mild aortic insufficiency.
mean pressure gradients were improved to 35.4 and 28.8 mm Hg, respectively (Figure 2B). Because the 20 mm balloon was fluoroscopically undersized and no aortic insufficiency was identified by TEE, the balloon size was changed to 22 mm (TYSHAK; NuMed) and two further inflations were performed resulting in peak and mean gradients of 28.1 and 23.0 mm Hg, respectively (Figure 2C). However, the right coronary cusp was still immobile due to calcified stiffness of the adjacent commissures on TEE. Accordingly, a 5 Fr pigtail catheter with a 0.035˝ Amplatzer super-stiff wire (St Jude Medical) was inserted into the left ventricle and the 22 mm balloon was inflated beside the pigtail catheter, which was maneuvered toward each of the adjacent commissures of the right coronary cusp during balloon inflations under fluoro and TEE guidance (Figure 3). Final hemodynamics demonstrated a significant increase in AVA (peak gradient, 16.4 mm Hg; AVA, 1.41 cm2; Figure 2D) and TEE showed improved aortic valve mobility with mild aortic insufficiency.
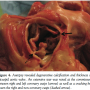 The patient successfully recovered from congestive heart failure after the procedure. However, unfortunately, he died of pneumonia 2 months later. Autopsy revealed heavy calcification of the aortic valve. However, a fifth finger easily slipped through the valve orifice without resistance. Extensive tearing was found at the commissure between the right and left coronary cusps as well as cracking between the right and non-coronary cusps (Figure 4).
The patient successfully recovered from congestive heart failure after the procedure. However, unfortunately, he died of pneumonia 2 months later. Autopsy revealed heavy calcification of the aortic valve. However, a fifth finger easily slipped through the valve orifice without resistance. Extensive tearing was found at the commissure between the right and left coronary cusps as well as cracking between the right and non-coronary cusps (Figure 4).
Discussion
TAVI is safe and effective in high-risk, symptomatic patients with severe aortic stenosis.1,2 PTAV, however, is still useful as a bridge to surgery for hemodynamically unstable cases or as an alternative to TAVI because of local regulations, increased risk of general anesthesia, or issues regarding the arterial access site. Because the Inoue balloon is not available for retrograde PTAV, balloon slipping is sometimes raised as a problem; rapid pacing during balloon inflation is one solution. We first utilized this “buddy-catheter or wire” technique as prevention for balloon slipping. Sheiban et al also reported the buddy wire or balloon technique for increasing deliverability of balloon-expandable aortic valve prosthesis.6 However, we have found that this technique also increases postdilatation AVA. Based on the concept of focused force coronary angioplasty by the use of a cutting balloon or a scoring balloon,7 we have been able to demonstrate a further increase in valve area by balloon inflation with an adjacent catheter positioned selectively at the stiff commissure. Although extensive recoil is one of the main reasons for the high rates of restenosis after PTAV, autopsy performed 2 months after our reported case demonstrated sustained valve area enlargement with extensive tearing at two commissures probably due to focused force by the buddy-catheter technique.
One limitation of this technique is the risk of balloon rupture. It is difficult to cross two wires through the stenotic valve before predilation. Therefore, we first inflate without the buddy catheter several times and then cross another wire to create the buddy catheter. However, this may increase the chance of balloon rupture. One solution to prevent balloon rupture may be a buddy catheter, rather than a buddy wire, because a wire is sharper than a catheter. We believe a combination of 4 or 5 Fr diagnostic catheters and a stiff spring wire may work. A further limitation is the risk of aortic insufficiency. A pathologic mechanism of aortic insufficiency in aortic valvuloplasty is known to be a tear of aortic leaflet.5 However, using this buddy-catheter technique, we found that the adjacent catheter appears to migrate into adjacent commissures upon commencement of balloon inflation. Therefore, we have not encountered severe aortic insufficiency in 13 cases of our experience. Another limitation is that this is only 1 case report. However, we have performed this technique in 13 PTAV cases. Although most cases demonstrated acute hemodynamic effect, 6 months of follow-up were not performed in most cases because most patients were either too high-risk to survive due to their comorbidities at 6 months, or the procedure was performed with the aim of bridge to surgery. We need to study the long-term effect in patients at less risk for surgery.
PTAV has not demonstrated improvement of survival for patients in severe symptomatic aortic stenosis in adults.4 If this is due to the high rate of restenosis, the use of focused force PTAV with the application of a buddy catheter into selective calcified stiff commissures may decrease the rate of restenosis and result in an improvement of survival.
Acknowledgment. We would like to thank Mie Sakurai, MD, for the echocardiography pictures, and Shunpei Mori, MD, and Naoki Uemura, MD, for the autopsy study pictures.
References
- Leon MB, Smith CR, Mack M, et al; the PARTNER trial investigators. Transcatheter aortic-valve implantation for aortic stenosis in patients who cannot undergo surgery. N Engl J Med. 2010;363(17):1597-1607.
- Smith CR, Leon MB, Mack MJ, et al; the PARTNER trial investigators. Transcatheter versus surgical aortic-valve replacement in high-risk patients. N Engl J Med. 2011;364(23):2187-2198.
- Bonow RO, Carabello B, de Leon AC Jr, et al; the ACC/AHA guidelines for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association task force on practice guidelines (committee on management of patients with valvular heart disease). J Am Coll Cardiol. 1998;32(5):1486-1582.
- Otto CM, Mickel MC, Kennedy JW, et al. Three-year outcome after balloon aortic valvuloplasty. Insights into prognosis of valvular aortic stenosis. Circulation. 1994;89(2):642-650.
- Waller BF, McKay C, VanTassel JW, Taliercio C, Howard J, Green F. Catheter balloon valvuloplasty of stenotic aortic valves. Part I: anatomic basis and mechanisms of balloon dilation. Clin Cardiol. 1991;14(10):836-846.
- Sheiban I, Infantino V, Bollati M. Buddy balloon to deliver a percutaneous aortic valve device: a percutaneous shoehorn? Catheter Cardiovasc Interv. 2009;74(5):805-807.
- Solar RJ, Ischinger TA. Focused force angioplasty: theory and application. Cardiovasc Radiat Med. 2003;4(1):47-50.
_____________________________________________
From the Cardiovascular Center, Sendai Kousei Hospital, Aoba, Sendai, Japan.
Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. The authors report no conflicts of interest regarding the content herein.
Manuscript submitted September 19, 2011, provisional acceptance given October 11, 2011, final version accepted November 10, 2011.
Address for correspondence: Norio Tada, MD, Cardiovascular Center, Sendai Kousei Hospital, 4-15 Hirose-machi, Aoba, Sendai, Japan 980-0873. Email: noriotada@hotmail.com














