A Novel Method to Deliver the Filtrap Coronary Embolic Protection Filter Using a Manual Thrombectomy Catheter: In Vitro and In Vivo Comparison with the Conventional Method
Abstract: Background. Although several types of distal embolic protection devices have been developed, they are usually difficult to use because of their stiff and bulky characteristics. We present a novel method to deliver the coronary embolic protection filter via a manual thrombectomy catheter. Methods. We modified the Filtrap embolic protection filter (Nipro Corporation) to pass through the aspiration thrombectomy catheter. Three interventional cardiologists performed the Filtrap delivery with in vivo and in vitro coronary arteries using two different methods. The in vitro experiments were performed using a Circuit Cardiac Catheterization Simulator (Just Medical Corporation) with and without coronary artery stenosis. The in vivo experiment was performed using a normal pig coronary artery. The times required for Filtrap delivery in the two different methods were compared. The target lesions were the main and side branch of the left anterior descending, left circumflex, and right coronary arteries. Results. The average Filtrap delivery time when using the thrombectomy catheter was significantly shorter compared to the time using the conventional method (in vitro experiment without coronary stenosis, 39 ± 14 seconds vs 30 ± 6 seconds and P=.019; in vitro experiment with multiple coronary stenosis, 69 ± 55 seconds vs 32 ± 11 seconds and P=.012; in vivo experiment, 169 ± 166 seconds vs 51 ± 12 seconds; P=.019). Conclusion. The study results indicate that delivery of the coronary embolic protection filter via a manual thrombectomy catheter may simplify the procedure and decrease the procedure time.
J INVASIVE CARDIOL 2012;24(4):159-163
Key words: embolic protection filter devices, Filtrap device
_____________________________________________
Recent advancements in imaging technologies suggest that distal embolization is more frequent than previously recognized in coronary and peripheral vascular interventions.1,2 Several types of embolic protection devices have been developed in different vascular territories. Embolic protection devices are expected to retrieve atherothrombotic debris and prevent its embolization, but their effect on clinical outcome has not been established, except for saphenous vein bypass grafts. Although small clinical studies have shown the beneficial effects of distal embolic protection on surrogate endpoints during primary percutaneous coronary interventions (PCI),3,4 large clinical trials have demonstrated that the use of distal embolic protection devices do not improve clinical outcomes.5,6 One possible explanation for the lack of efficacy is that most distal protection devices are difficult to use, promoting embolization while crossing the culprit lesion and extending the door-to-balloon time. The most frequently used device type incorporates a guidewire with a filter. These distal protection devices require guidewire manipulation to cross the culprit lesion, but the devices are usually more stiff and bulky compared to the conventional guidewire, resulting in prolonged procedure times and vascular damage.
On the contrary, aspiration thrombectomy was proved to be beneficial in primary PCI and recommended for use.7–10 We propose a novel method to deliver the distal embolic protection filter using a manual thrombectomy catheter. In this method, aspiration is performed first; thereafter, the thrombectomy catheter is advanced forward, after which the protection filter is advanced via the aspiration lumen of the thrombectomy catheter and deployed. In the present study, the feasibility and efficacy of this method was evaluated by comparing the conventional delivery method with in vitro and in vivo experiments.
Methods
Study design. The study was designed to assess the safety and efficacy of a novel filter delivery method using a manual thrombectomy catheter by comparing it with a conventional delivery method. Three interventional cardiologists, each with the experience of more than 300 coronary interventions, performed the delivery of distal protection filters to in vivo and in vitro coronary arteries using two different methods. The times required for the filter delivery in two different methods were compared. Operator A has 11 years of experience in interventional cardiology, Operator B has 8 years of experience, and Operator C has 7 years of experience. Comparisons of means were made using a paired student’s t-test with P<.05 considered statistically significant. All numeric values were expressed as the mean ± 1 standard deviation.
 Distal embolic protection filter and the delivery system. A modified Filtrap (Nipro Corporation) distal protection filter was used in the present study. The Filtrap was originally developed by Isshiki and colleagues, and is commercially available in Japan.9 The original Filtrap has a 35-mm long distal radiopaque floppy guidewire with a 1.15-mm diameter distal tip fixed in a spindle-shaped spiral wire basket with a porous polyurethane filter membrane (Figure 1A). The original Filtrap is folded in a 3.45 Fr delivery catheter and delivered through the culprit lesion with wire manipulation in the same way as other commercially available embolic protection filters.5,6,11 The distal tip of the Filtrap is modified smaller to 1.10 mm to pass through the aspiration catheter in the present study (Figure 1B).
Distal embolic protection filter and the delivery system. A modified Filtrap (Nipro Corporation) distal protection filter was used in the present study. The Filtrap was originally developed by Isshiki and colleagues, and is commercially available in Japan.9 The original Filtrap has a 35-mm long distal radiopaque floppy guidewire with a 1.15-mm diameter distal tip fixed in a spindle-shaped spiral wire basket with a porous polyurethane filter membrane (Figure 1A). The original Filtrap is folded in a 3.45 Fr delivery catheter and delivered through the culprit lesion with wire manipulation in the same way as other commercially available embolic protection filters.5,6,11 The distal tip of the Filtrap is modified smaller to 1.10 mm to pass through the aspiration catheter in the present study (Figure 1B).
We also developed a low-profile 6 Fr guiding-catheter compatible manual thrombectomy catheter with a large 1.15-mm diameter aspiration lumen which is designed based on the TVAC aspiration catheter (Nipro Corporation). The original TVAC has a unique distal tip in the shape of a duck bill and its efficacy was proven in a prospective randomized trial.9,12 The modified TVAC catheter applied in the present study has a larger aspiration lumen compared to the original one in which the modified Filtrap can be delivered to the target lesion (Figure 1B).
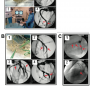 In vitro experiments. The Circuit Cardiac Catheterization Simulator (Just Medical Corporation) was used for in vitro experiments. The circuit simulator is made of a silicone rubber tube that mimics the human cardiovascular system (Figure 2A). An aqueous solution of glycerol of concentration 52% by weight was used as the perfusate. A tubing pump (MP-1983E; As One Corporation) was used to circulate the perfusate with a flow rate of 600ml/min through the system. The original and modified Filtraps were attempted to deliver to the main and side branch of the left descending artery (LAD), the main and side branch of the left circumflex artery (LCX) and the main and side branch of the right coronary artery (RCA) in the circuit simulator using two different methods as described previously. The attempts were made by 3 interventional cardiologists using a 6 Fr guiding catheter. The first experiment was undertaken using normal (non-stenotic) coronary arteries in the circuit system. The same attempt was then repeated after the creation of multiple stenoses by pinching the coronary artery with plastic forceps (Figure 2B). All procedures were undertaken under fluoroscopic guidance (OEC 9800 Plus; General Electric). Filtrap delivery time was defined as the elapsed time from insertion of the Filtrap into the guiding catheter to delivery to the target lesion. The Filtrap delivery time in each attempt was measured as the primary endpoint of the study.
In vitro experiments. The Circuit Cardiac Catheterization Simulator (Just Medical Corporation) was used for in vitro experiments. The circuit simulator is made of a silicone rubber tube that mimics the human cardiovascular system (Figure 2A). An aqueous solution of glycerol of concentration 52% by weight was used as the perfusate. A tubing pump (MP-1983E; As One Corporation) was used to circulate the perfusate with a flow rate of 600ml/min through the system. The original and modified Filtraps were attempted to deliver to the main and side branch of the left descending artery (LAD), the main and side branch of the left circumflex artery (LCX) and the main and side branch of the right coronary artery (RCA) in the circuit simulator using two different methods as described previously. The attempts were made by 3 interventional cardiologists using a 6 Fr guiding catheter. The first experiment was undertaken using normal (non-stenotic) coronary arteries in the circuit system. The same attempt was then repeated after the creation of multiple stenoses by pinching the coronary artery with plastic forceps (Figure 2B). All procedures were undertaken under fluoroscopic guidance (OEC 9800 Plus; General Electric). Filtrap delivery time was defined as the elapsed time from insertion of the Filtrap into the guiding catheter to delivery to the target lesion. The Filtrap delivery time in each attempt was measured as the primary endpoint of the study.
In vivo experiments. In vivo experiments were approved by the institutional animal care and use committee at the Medical School of Kyoto University. One female pig weighing 25 kg was studied in accordance with the Guide for the Care and Use of Laboratory Animals proposed by the Institute of Laboratory Animal Resources. Under general anesthesia, left and right coronary angiographies were performed using a 6 Fr guiding catheter through an introduction sheath percutaneously placed in the right carotid artery. The coronary artery diameter in the target lesion was measured by quantitative coronary angiography (QCA) after the coronary angiographies. The original and modified Filtrap were attempted to deliver to the main and side branch of LAD, the main and side branch of the LCX and the main branch of the RCA by the same 3 interventional cardiologists. The Filtrap delivery time in each attempt was also measured with in vivo experiments.
Results
 The overall results are presented in Figure 3. The average Filtrap delivery time when using the thrombectomy catheter was significantly shorter compared to the time when using the conventional method in in vitro experiments without creating coronary stenosis (39 ± 14 seconds vs 30 ± 6 seconds; P=.019; paired t-test). The results were identical after creating the multiple stenosis in the coronary artery in in vitro experiments; the average delivery time was 69 ± 55 seconds when using the conventional delivery method and 32 ± 11 seconds when using the manual thrombectomy catheter (P=.012; paired t-test).
The overall results are presented in Figure 3. The average Filtrap delivery time when using the thrombectomy catheter was significantly shorter compared to the time when using the conventional method in in vitro experiments without creating coronary stenosis (39 ± 14 seconds vs 30 ± 6 seconds; P=.019; paired t-test). The results were identical after creating the multiple stenosis in the coronary artery in in vitro experiments; the average delivery time was 69 ± 55 seconds when using the conventional delivery method and 32 ± 11 seconds when using the manual thrombectomy catheter (P=.012; paired t-test).
An in vivo experiment using a normal pig coronary artery was also assessed. The coronary artery diameter in the target lesion measured by QCA was 2.88 mm in the LAD main branch, 1.75 mm in the LAD side branch, 3.00 mm in the LCX main branch, 1.88 mm in the LCX side branch and 2.68 mm in the RCA. A total of three attempts using the conventional delivery method were abandoned after 8 minutes of effort. The Filtrap delivery times in these abandoned cases were counted as 8 minutes. The average Filtrap delivery time was 169 ± 166 seconds when using the conventional delivery method and 51 ± 12 seconds when using the manual thrombectomy catheter (P=.015; paired t-test). No vascular injuries occurred during Filtrap delivery with either method.
Discussion
The main finding of the study is that the Filtrap delivery using a manual thrombectomy catheter reduces procedure time compared to the conventional delivery method in in vitro and in vivo experiments. Guidewire manipulation is always required to cross the culprit lesion when using the original Filtrap, which we consider a major drawback in real-world clinical practice. The initial clinical experience reported that Filtrap delivery was abandoned in 7% of cases and the Filtrap delivery required 9.4 ± 3.4 minutes.11 The most frequently used distal embolic protection device type currently on the market in other countries is also an integrated filter guidewire system that requires guidewire manipulation to advance into the coronary artery.13 These devices are usually stiff and bulky, sometimes causing vascular damage or a prolonged procedure time.14 Crossing a floppy guidewire is always the first thing to do in the current PCI. All other devices, including intravascular ultrasound, thrombectomy catheter, and balloon and stent catheter, are advanced over the pre-placed guidewire. Compared to these modern coronary interventional devices, the Filtrap system, which has a distal fixed guidewire and requires careful guidewire handling, seems old fashioned and is not user friendly.
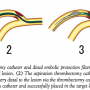 Several recent randomized clinical studies demonstrated that intracoronary aspiration thrombectomy during primary PCI was associated with better clinical outcomes.8,9 Aspiration thrombectomy is currently routinely used in primary PCI. We propose a method using a manual aspiration catheter for the delivery of embolic protection filter in primary PCI. The present study demonstrated that the Filtrap delivery using a manual thrombectomy catheter simplifies the procedure and reduces procedure time for in vitro and in vivo experiments. Door-to-balloon time is extremely important in primary PCI in patients with acute myocardial infarction because the time delay is closely correlated with in-hospital mortality.15 Some would argue that if the filter protection is employed post aspiration when coronary blood flow has already been restored, the door to flow time is the same in both conventional methods and the method described here, and also that predilatation is avoided where much of the initial debris arises, hence rendering the use of a filter less necessary. The VAcuuM asPIration thrombus REmoval (VAMPIRE) trial described that TIMI grade 2 or 3 flow after aspiration was not achieved in 9.3% of patients. Lemesle and colleagues reported that 50% of patients did not achieve TIMI grade 3 flow after aspiration. In the Thrombus Aspiration during Percutaneous coronary intervention in Acute myocardial infarction Study (TAPAS), which assigned 1071 patients to thrombus aspiration followed by primary PCI or conventional primary PCI, balloon dilatation was required after aspiration in 153 patients (28.6%) of the aspiration group. In the Export study, 28.3% of patients required balloon dilatation after aspiration. These study results indicate that thrombus aspiration is not enough to restore sufficient coronary blood flow and balloon dilatation is required in a certain proportion of patients. Thus, improvements to the filter delivery system will potentially improve the clinical outcome in patients with myocardial infarction. Figure 4 depicts our proposed usage of the thrombectomy catheter and distal embolic protection filter in patients with acute myocardial infarction. The method of distal embolic protection filter delivery using a manual thrombectomy catheter can be applicable for patients with stable coronary artery disease, saphenous vein graft disease, and carotid and peripheral arterial diseases.
Several recent randomized clinical studies demonstrated that intracoronary aspiration thrombectomy during primary PCI was associated with better clinical outcomes.8,9 Aspiration thrombectomy is currently routinely used in primary PCI. We propose a method using a manual aspiration catheter for the delivery of embolic protection filter in primary PCI. The present study demonstrated that the Filtrap delivery using a manual thrombectomy catheter simplifies the procedure and reduces procedure time for in vitro and in vivo experiments. Door-to-balloon time is extremely important in primary PCI in patients with acute myocardial infarction because the time delay is closely correlated with in-hospital mortality.15 Some would argue that if the filter protection is employed post aspiration when coronary blood flow has already been restored, the door to flow time is the same in both conventional methods and the method described here, and also that predilatation is avoided where much of the initial debris arises, hence rendering the use of a filter less necessary. The VAcuuM asPIration thrombus REmoval (VAMPIRE) trial described that TIMI grade 2 or 3 flow after aspiration was not achieved in 9.3% of patients. Lemesle and colleagues reported that 50% of patients did not achieve TIMI grade 3 flow after aspiration. In the Thrombus Aspiration during Percutaneous coronary intervention in Acute myocardial infarction Study (TAPAS), which assigned 1071 patients to thrombus aspiration followed by primary PCI or conventional primary PCI, balloon dilatation was required after aspiration in 153 patients (28.6%) of the aspiration group. In the Export study, 28.3% of patients required balloon dilatation after aspiration. These study results indicate that thrombus aspiration is not enough to restore sufficient coronary blood flow and balloon dilatation is required in a certain proportion of patients. Thus, improvements to the filter delivery system will potentially improve the clinical outcome in patients with myocardial infarction. Figure 4 depicts our proposed usage of the thrombectomy catheter and distal embolic protection filter in patients with acute myocardial infarction. The method of distal embolic protection filter delivery using a manual thrombectomy catheter can be applicable for patients with stable coronary artery disease, saphenous vein graft disease, and carotid and peripheral arterial diseases.
Study limitations. The present study has several limitations. First, the Filtrap embolic protection filter is only available in Japan. However, we consider that the proposed delivery method can be applicable to the other commercially available embolic protection filters with small modifications, since the basic structures of the devices are essentially the same. Second, the in vivo study was conducted in normal coronary arteries in the pig and the stenoses were artificially made by plastic forceps in the in vivo study. There seems to be a large difference between these experimental models and diseased human coronary arteries. We calculated the interoperator correlation coefficient of the Filtrap delivery time of each pair of three operators to verify the reproducibility of the study. The correlation coefficients of each pair were 0.85 (Operator A vs B), 0.63 (Operator B vs C), and 0.81 (Operator A vs C). The interoperator correlation coefficients of all possible pairs were larger than 0.6, which indicates that the reproducibility of the study was moderate to good. However, more detailed studies using the pig coronary artery stenosis model are needed before clinical use of this method.
Conclusion
In conclusion, the study results indicate that delivery of the coronary embolic protection filter via a manual thrombectomy catheter may simplify the procedure and decrease the procedure time.
Acknowledgment. The original Filtrap and TVAC were supplied by Nipro Corporation.
References
- Nijveldt R, Beek AM, Hirsch A, et al. Functional recovery after acute myocardial infarction: comparison between angiography, electrocardiography, and cardiovascular magnetic resonance measures of microvascular injury. J Am Coll Cardiol. 2008;52(3):181-189.
- Hirsch A, Nijveldt R, Haeck JDE, et al. Relation between the assessment of microvascular injury by cardiovascular magnetic resonance and coronary Doppler flow velocity measurements in patients with acute anterior wall myocardial infarction. J Am Coll Cardiol. 2008;51(23):2230-2238.
- Duan Y-Y, Zhang H-B, Liu L-W, et al. Effects of distal protection on left ventricular function in acute anterior myocardial infarction: a Doppler echocardiographic study. Int J Cardiovasc Imaging. 2010;26(2):125-133.
- Ito N, Nanto S, Doi Y, et al. Distal protection during primary coronary intervention can preserve the index of microcirculatory resistance in patients with acute anterior ST-segment elevation myocardial infarction. Circulation. 2011;75(1):94-98.
- Guetta V, Mosseri M, Shechter M, et al. Safety and efficacy of the FilterWire EZ in acute ST-segment elevation myocardial infarction. Am J Cardiol. 2007;99(7):911-915.
- Gick M, Jander N, Bestehorn H-P, et al. Randomized evaluation of the effects of filter-based distal protection on myocardial perfusion and infarct size after primary percutaneous catheter intervention in myocardial infarction with and without ST-segment elevation. Circulation. 2005;112(10):1462-1469.
- Kushner FG, Hand M, Smith SC, et al. 2009 focused updates: ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarction (updating the 2004 guideline and 2007 focused update) and ACC/AHA/SCAI guidelines on percutaneous coronary intervention (updating the 2005 guideline and 2007 focused update) a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2009;54(23):2205-2241.
- Vlaar PJ, Svilaas T, van der Horst IC, et al. Cardiac death and reinfarction after 1 year in the thrombus aspiration during percutaneous coronary intervention in acute myocardial infarction study (TAPAS): a 1-year follow-up study. Lancet. 2008;371(9628):1915-1920.
- Ikari Y, Sakurada M, Kozuma K, et al. Upfront thrombus aspiration in primary coronary intervention for patients with ST-segment elevation acute myocardial infarction: report of the VAMPIRE (VAcuuM asPIration thrombus REmoval) trial. JACC Cardiovasc Intervent. 2008;1(4):424-431.
- Chevalier B, Gilard M, Lang I, et al. Systematic primary aspiration in acute myocardial percutaneous intervention: a multicentre randomised controlled trial of the export aspiration catheter. Eurointervention. 2008;4(2):222-228.
- Isshiki T, Kozuma K, Kyono H, Suzuki N, Yokoyama N, Yamamoto Y. Initial clinical experience with distal embolic protection using “Filtrap,” a novel filter device with a self-expandable spiral basket in patients undergoing percutaneous coronary intervention. Cardiovasc Intervent Ther. 2010;26(1):12-17.
- Sakurada M, Ikari Y, Isshiki T. Improved performance of a new thrombus aspiration catheter: outcomes from in vitro experiments and a case presentation. Catheter Cardiovasc Intervent. 2004;63(3):299-306.
- Roffi M, Mukherjee D. Current role of emboli protection devices in percutaneous coronary and vascular interventions. Am Heart J. 2009;157(2):263-270.
- Mauri L, Rogers C, Baim DS. Devices for distal protection during percutaneous coronary revascularization. Circulation. 2006;113(22):2651-2656.
- Rathore SS, Curtis JP, Chen J, et al. Association of door-to-balloon time and mortality in patients admitted to hospital with ST elevation myocardial infarction: national cohort study. Br Med J. 2009;338:B1807-B1807.
_____________________________________________
From the 1Department of Cardiovascular Medicine, Graduate School of Medicine, Kyoto University, Kyoto, Japan and the 2Department of Cardiovascular Medicine, Gifu Heart Center, Gifu, Japan.
Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. The authors report no conflicts of interest regarding the content herein.
Funding. The original Filtrap and TVAC were supplied by Nipro Corporation.
Manuscript submitted August 16, 2011, provisional acceptance given October 6, 2011, final version accepted December 20, 2011.
Address for correspondence: Naritatsu Saito, Department of Cardiovascular Medicine, Graduate School of Medicine, Kyoto University, 54 Shogoin Kawahara-cho, Sakyo-ku, Kyoto 606-8507, Japan. Email: naritatu@kuhp.kyoto-u.ac.jp












