Electrocardiographic Response to Intravenous and Intraarterial Injection of Iosimenol (A New, Iodinated, Non-Ionic, Iso-Osmolar Contrast Medium)
Abstract: Serious arrhythmias, sometimes related to the injection of iodinated contrast media, are known complications of cardiac angiography. A new, iodine-based, non-ionic, iso-osmolar x-ray contrast media is in development for use in these procedures. This contrast medium, iosimenol, has a lower viscosity, higher electrolyte content, and higher iodine concentration than other available iso-osmolar contrast media. The present study is a retrospective re-read and centralized analysis of the electrocardiographic response to intravenous and non-cardiac intraarterial injections of iosimenol, placebo, or iodixanol (Visipaque; GE Healthcare, Inc) in a total of 167 healthy subjects and patients enrolled in early clinical trials. No clinically relevant changes in heart rate and rhythm, morphology, atrioventricular conduction, or ventricular repolarization were noted after injection of any of the test articles in these studies. These results, despite the limited number of patients in these trials, suggest that iosimenol can be used safely in larger populations.
J INVASIVE CARDIOL 2013;25(11):573-578
Key words: contrast media, iosimenol
_____________________________
Approximately 1.5 million cardiac angiography procedures using iodinated contrast media (CM) are performed annually in the United States1 and an estimated 80 million diagnostic and interventional cardiovascular and non-cardiovascular procedures are performed worldwide.2
The favorable safety profile of CM with lower osmolality has resulted in an almost global conversion from older ionic CM to low osmolar non-ionic compounds.3,4 However, nephrotoxic5 and thromboembolic events,6 as well as serious arrhythmias,7 remain clinical concerns.
Current data support the use of the iso-osmolar (290 mOsm/kg H2O) non-ionic CM iodixanol (Visipaque; GE Healthcare, Inc) during invasive procedures due to reduced nephrotoxicity8 and because the higher electrolyte content has the potential to reduce the incidence of arrhythmias.9 However, these advantages come at the expense of significantly higher viscosity than other CM, posing some challenges when used in procedures that require catheters of small diameter, such as radial and pediatric applications, and possibly counteracting the improved renal safety profile that accompanies reduced osmolality.10
Iosimenol is an iodine-based, non-ionic, iso-osmolar x-ray CM in development for use during cardiac angiography and percutaneous coronary intervention (PCI). With lower viscosity and higher electrolyte content than iodixanol at equivalent iodine concentrations, iosimenol has the potential to improve cardiac safety. Given the predicted low rate of serious arrhythmias following injection of iosimenol, direct observations of severe events in clinical trials need to be supplemented by studies with surrogate endpoints.
The QT interval of the surface electrocardiogram (ECG) consists of the QRS complex, which represents depolarization within the His-Purkinje system and ventricles and the JT interval, which reflects ventricular repolarization. Prolonged repolarization can lead under the right circumstances to torsade de pointes, a polymorphic ventricular tachyarrhythmia that can result in syncope or progress to ventricular fibrillation and sudden cardiac death. Therefore, current regulatory guidance11 emphasizes evaluation of cardiac repolarization, or more specifically the QT interval and, because of its inverse relationship to heart rate (HR), the corrected QT interval (QTc).
The present study summarizes the ECG response from early clinical trials with iosimenol, particularly effects on ventricular repolarization after intravenous (IV) and non-cardiac intraarterial injection, at various levels of systemic exposure, and compared with effects after injection of iodixanol.
Methods
The study was sponsored by Otsuka Pharmaceutical Development & Commercialization Inc, in Princeton, New Jersey and included re-reading, evaluation, and analyses (at a central ECG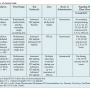 core laboratory; ERT, Philadelphia, Pennsylvania) of ECG recordings from previously conducted clinical trials (Table 1). The original studies were sponsored by Interpharma Praha (Prague, Czech Republic) and were approved by the local ethics committees of the investigating centers. All patients provided signed informed consent and the study followed proper disclosure protocols.
core laboratory; ERT, Philadelphia, Pennsylvania) of ECG recordings from previously conducted clinical trials (Table 1). The original studies were sponsored by Interpharma Praha (Prague, Czech Republic) and were approved by the local ethics committees of the investigating centers. All patients provided signed informed consent and the study followed proper disclosure protocols.
Original ECGs recorded on paper from a total of 167 subjects enrolled in the previous clinical trials were re-analyzed by the core ECG laboratory.12-14 ECGs were bar-coded, scanned into .png files and imported, together with patient demographic information and ECG sample time points (Table 1), into a validated data management system (EXPERT). ECG intervals were measured in a blinded manner by trained analysts using a high-resolution Digipad method (ie, on a digital tablet with magnification and on-screen calipers). Standard definitions for QT duration (tangent method) and morphology changes (ST depression starting at ≥0.5 mm and new negative T-wave changes) were employed. A cardiologist then verified the interval durations, performed the morphology analysis, and recorded any abnormal T-U wave complexes that suggested an effect on cardiac repolarization.
Averages of three measurements of HR, RR, PR, QRS, and QT interval durations were used for analysis; both Fridericia and Bazett corrections (QTcF and QTcB, respectively) were used for QTc data analysis.
Central tendency analysis. Post-dose recordings were compared with baseline to establish the central tendency of all ECG interval parameter deviations (HR, PR, QRS, QT, QTcF, and QTcB). Central tendency was defined as post-dose recordings with baseline values subtracted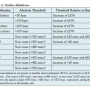 and assessed at each individual time point, as well as averages over time.
and assessed at each individual time point, as well as averages over time.
Outlier analyses. An outlier (categorical) analysis supplemented the central tendency analysis by determining if there were patients who had an exaggerated effect on any ECG interval that would not be revealed in a mean change from baseline central tendency analysis. Outliers were defined as patients who exceeded an absolute and/or relative threshold at least at 1 time point. Outlier criteria are summarized in Table 2.
Morphological analyses. Morphological analyses were performed on ECG waveform interpretations by the cardiologist at the central ECG laboratory. Significant changes from baseline to any time point are provided in Table 3.
Pharmacokinetic-pharmacodynamic analyses. The pharmacokinetic-pharmacodynamic (PK-PD) analysis addressed the relationship between iosimenol plasma concentrations and concomitant ECG/QTc data. Three ascending dose groups, each with 8 healthy male volunteers, were enrolled in study BP 13-4.1 and given IV doses of iosimenol (6 per group) or placebo (saline). Each subject was given only 1 dose. Plasma samples were drawn concomitantly with ECG recordings and iosimenol concentration was indirectly determined based on iodine content measured with x-ray fluorescence.
Results
Results from central tendency analyses are summarized in Table 4.
The ECG data showed no clinically relevant signal of changes in HR, atrioventricular (AV)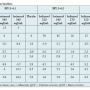 conduction (as measured by PR interval duration), cardiac depolarization (as measured by QRS interval duration), or in cardiac waveform morphology or new rhythms. Data on central tendency by time averaging across the study and by comparison of baseline with each time point revealed no clear signal of effect on cardiac repolarization and no differences between the iosimenol and iodixanol groups. The 3 mL/kg bw of iosimenol 340 mgI/mL showed mean changes from baseline placebo-corrected heart rate of 5 beat per minute increase and a 4.2 ms increase in QTcF (the 2-sided 90% confidence interval for the 3.0 mL/kg dose placebo-corrected QTcF was 3.3 ms, which is below the regulatory concern of 5 ms) that is likely not of clear clinical
conduction (as measured by PR interval duration), cardiac depolarization (as measured by QRS interval duration), or in cardiac waveform morphology or new rhythms. Data on central tendency by time averaging across the study and by comparison of baseline with each time point revealed no clear signal of effect on cardiac repolarization and no differences between the iosimenol and iodixanol groups. The 3 mL/kg bw of iosimenol 340 mgI/mL showed mean changes from baseline placebo-corrected heart rate of 5 beat per minute increase and a 4.2 ms increase in QTcF (the 2-sided 90% confidence interval for the 3.0 mL/kg dose placebo-corrected QTcF was 3.3 ms, which is below the regulatory concern of 5 ms) that is likely not of clear clinical relevance.
relevance.
Results from the outlier analysis are summarized in Table 5. Outlier changes in ECG interval durations such as type and severity, most commonly a minor increase in QTc intervals, were randomly distributed between the iosimenol and iodixanol groups and did not appear to deviate from findings in the placebo controls. Morphologic ECG changes are summarized in Table 6.
There were two patients who had morphologic ECG changes after IV injection of iodixanol: 1 patient had ST-segment depression and 1 patient had a right bundle branch block (RBBB). There were no subjects who had morphologic ECG changes following IV injection of iosimenol. There were 2 subjects who had morphologic ECG changes after intraarterial injection of each of the two compounds. There were no findings of systemic changes in any of the groups.
of the two compounds. There were no findings of systemic changes in any of the groups.
A review of the effect-exposure relationship to analyze further the potential effects on cardiac repolarization is shown in Figure 1. There were no clear dose-dependent effects on QTc observed.
Discussion
The importance of the electrolyte composition of CM formulations in terms of arrhythmogenic potential has been demonstrated repeatedly in many animal models.15-25 Adding proper concentrations of sodium decreases the arrhythmic potential, although too large a quantity has a negative inotropic effect. Alternatively, calcium has a positive chronotropic effect, although it increases the risk of tachyarrhythmias above a certain level. Ideally, it would seem preferable to use an electrolyte composition similar in concentration to circulating blood; this is especially true for sodium.
potential has been demonstrated repeatedly in many animal models.15-25 Adding proper concentrations of sodium decreases the arrhythmic potential, although too large a quantity has a negative inotropic effect. Alternatively, calcium has a positive chronotropic effect, although it increases the risk of tachyarrhythmias above a certain level. Ideally, it would seem preferable to use an electrolyte composition similar in concentration to circulating blood; this is especially true for sodium.
Although non-ionic CM, such as iodixanol, are iso-osmolar compared with plasma and are frequently considered less nephrotoxic than commonly used products with higher osmolality,26,27 there is a pharmaceutical challenge: adding electrolytes to a solution with a fixed concentration of CM will increase the osmolality. Lowering the concentration of the iodine-containing molecule will compensate for this, although it may limit the efficacy of the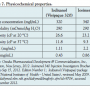 product. Currently, iodixanol is the only marketed iodine-based product formulated to be iso-osmolar, with a high iodine concentration and added electrolytes. However, due to smaller molecular size and higher iodine concentration than iodixanol, iosimenol may be formulated with more electrolytes (sodium) and remain iso-osmolar (Table 7). Additionally, because iosimenol has a lower viscosity than iodixanol, it is easier to inject in smaller-diameter catheters, such as those frequently used for radial and pediatric purposes.10
product. Currently, iodixanol is the only marketed iodine-based product formulated to be iso-osmolar, with a high iodine concentration and added electrolytes. However, due to smaller molecular size and higher iodine concentration than iodixanol, iosimenol may be formulated with more electrolytes (sodium) and remain iso-osmolar (Table 7). Additionally, because iosimenol has a lower viscosity than iodixanol, it is easier to inject in smaller-diameter catheters, such as those frequently used for radial and pediatric purposes.10
Conclusions
In summary, it appears reasonable to anticipate the arrhythmic potential of iosimenol to be as low as, or lower than, iodixanol. However, because serious tachyarrhythmias are rare, a very large sample would be needed to directly demonstrate this in a randomized clinical trial. Almen et al have frequently used a pig model to study the arrhythmogenic effects of CM and have demonstrated that QTc is a reliable predictor of the potential for ventricular fibrillation after intracoronary injection of CM.28 As with any animal model, there are, of course, limitations to the conclusions that may be drawn in terms of application to humans. Pharmaceutical development protocol, therefore, requires the study of surrogate endpoints that are considered predictors of arrhythmogenic potential, typically changes in the depolarization-repolarization process of the ventricular myocardium.
A delay in cardiac repolarization in response to pharmaceuticals other than antiarrhythmics, as measured by prolongation of the QT interval, is an accepted predictor of risk for cardiac arrhythmias, including torsades de pointes, and potentially for other ventricular tachyarrhythmias. Additionally, arrhythmias during cardiac angiography are not necessarily related to pharmacologic effects; rather, they are likely caused by mechanical or other procedure-related events.
This study demonstrated no clinically relevant changes in HR, AV conduction (as measured by QRS interval duration), or cardiac wave form morphology or new rhythms, either by IV or non-cardiac intraarterial injection of iosimenol. There were no differences detected between groups receiving iodixanol and those receiving iosimenol. Recognizing that QT prolongation is an imperfect biomarker for arrhythmic risk and that the sample size is limited, there were no ECG findings indicative of potential adverse arrthymogenic effects following injections of iosimenol that should prevent its usage in larger clinical trial populations.
References
- Riley RF, Don CW, Powell W, et al. Trends in coronary revascularization in the United States from 2001 to 2009: recent declines in percutaneous coronary intervention volumes. Circ Cardiovasc Qual Outcomes. 2011;4(2):1-5.
- Persson PB. Editorial: contrast medium-induced nephropathy. Nephrol Dial Transplant. 2005;20(Suppl 1):i1.
- Davidson CJ, Laskey WK, Hermiller JB, et al. Randomized trial of contrast media utilization in high risk PTCA: the COURT trial. Circulation. 2000;101(18):2172-2177.
- Harrison JK, Hermiller JB, Vetrovec GW, et al. A randomized study of 1276 patients undergoing PCI using iodixanol (Visipaque) vs. iopamidol (Isovue): comparison of in-hospital and 30 day major adverse cardiac events. The results of the VICC trial. Circulation. 2003;108(Suppl S):354-S355.
- Alexopoulos E, Spargias K, Kyrzopoulos S, et al. Contrast-induced acute kidney injury in patients with renal dysfunction undergoing a coronary procedure and receiving non-ionic low-osmolar versus iso-osmolar contrast media. Am J Med Sci. 2010;339(1):25-30.
- Bellemain-Appaix A, Beygui F, Lesty C, et al. Impact of anticoagulation on ionic and nonionic contrast media effect on thrombogenesis and fibrinolysis: the PEPCIT study. Catheter Cardiovasc Interv. 2012;79(5):823-833.
- Missri J, Jeresaty RM. Ventricular fibrillation during coronary angiography: reduced incidence with nonionic contrast media. Catheter Cardiovasc Diagn. 1990;19(1):4-7.
- Aspelin P, Aubry P, Fransson S-G, et al. Nephrotoxic effects in high-risk patients undergoing angiography. New Engl J Med. 2003;348(6):491-499.
- Chai CM, Karlsson JO, Almén T. Incidence of ventricular fibrillation during left coronary angiography in pigs: comparison of a solution of the nonionic dimer iodixanol with solutions of five different nonionic monomers. Acta Radiol. 2008;49(2):150-156.
- Voeltz MD, Nelson MA, McDaniel MC, Manoukian SV. The important properties of contrast media: focus on viscosity. J Invasive Cardiol. 2007;19(3):1A-9A.
- International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. ICH Harmonised Tripartite Guideline E2F. Current Step 4 version. 2010 Aug 17.
- Report of centralized re-evaluation of cardiac ECG strips acquired in the clinical study BP13-4.1 (Otsuka Internal report).
- Report of centralized re-evaluation of cardiac ECG strips acquired in the clinical study BP13-4.2 (Otsuka Internal report).
- Report of centralized re-evaluation of cardiac ECG strips acquired in the clinical study BP13-4.3 (Otsuka Internal report).
- Morris TW. Ventricular fibrillation during right coronary arteriography with ioxaglate, iohexol and iopamidol in dogs. Invest Radiol. 1988;23(3):205-208.
- Hayakawa K, Yamashita K. Low-osmolality contrast media-induced ventricular fibrillation. Invest Radiol. 1989;24(4):298-301.
- Misumi K, Tateno O, Fujiki M, et al. The risk of contrast media-induced ventricular fibrillation is low in canine coronary angiography with ioxilan. J Vet Med Sci. 2000;62(4):421-426.
- Jynge P, Holten T, Öksendal AN. Sodium-calcium balance and cardiac function with isotonic iodixanol. An experimental study in the isolated rat heart. Invest Radiol. 1993;28(1):20-25.
- Jynge P. Sodium-calcium balance in coronary angiography and experimental experience with iodixanol. Eur Radiol. 1996;6(Suppl 2):S8-S12.
- Besjakov J, Chai CM, Bååth L, Almén T. Comparison between oxygenated iohexol solution enriched with electrolytes and other low-osmotic contrast media. Electrophysiologic and hemodynamic effects during perfusion of the left coronary artery in the pig. Invest Radiol. 1994;29 (Suppl 2):S238-S241.
- Bååth L, Almén T. Reduction of the risk of ventricular fibrillation in the isolated rabbit heart by small additions of electrolytes to non-ionic monomeric contrast media. Acta Radiol. 1989;30(3):327-333.
- Bååth L, Almén T, Öksendal A. Effects of sodium addition to non-ionic contrast media on cardiac contractile force. Perfusion of the isolated rabbit heart with iohexol and iopentol containing 0 to 154 mmol Na+/L added as NaCl. Acta Radiol. 1990;31(1):99-104.
- Bååth L, Besjakov J, Öksendal A. Sodium-calcium balance in nonionic contrast media: effects on the risk of ventricular fibrillation in the isolated rabbit heart. Invest Radiol. 1993;28(3):223-227.
- Jacobsen EA, Klöw NE, Mortensen E, Refsum H. Sodium addition to nonionic contrast media. Effects on cardiac monophasic action potentials and hemodynamics in dog model. Invest Radiol. 1991;26(10):888-893.
- Pedersen HK, Jacobsen EA, Refsum H. Contrast media-induced ventricular fibrillation: an experimental study of the effects of dimeric contrast media during wedged catheter injection in dogs. Acad Radiol. 1994;1(2):136-144.
- Jo SH, Youn TJ, Koo BK, et al. Renal toxicity evaluation and comparison between visipaque (iodixanol) and hexabrix (ioxaglate) in patients with renal insufficiency undergoing coronary angiography: the RECOVER study: a randomized controlled trial. J Am Coll Cardiol. 2006;48(5):924-30.
- McCullough PA. Renal safety of iodixanol. Expert Rev Cardiovasc Ther. 2006;4(5):655-661.
- Chai C-M, Almén T, Bååth L, et al. Signs in vector-electrocardiography (VECG) predicting the fibrillatory propensity of iodixanol and mannitol solutions after injection into the left coronary artery of pigs. Acad Radiol. 2007;14(2):162-177.
From 1Otsuka Pharmaceutical Development & Commercialization, Inc, Princeton, New Jersey, and 2ERT (eResearch Technology, Inc), Philadelphia, Pennsylvania.
Funding: Otsuka Pharmaceuticals and Interpharma Praha.
Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. Drs Zhang and Hogstrom are both employees of Otsuka Pharmaceutical Development & Commercialization, Inc. Dr Morganroth is a grant holder and employee of ERT, the company that performed the electrocardiographic analyses.
Manuscript submitted February 26, 2013, provisional acceptance given April 10, 2013, final version accepted July 19, 2013.
Address for correspondence: James J. Zhang, MD, Otsuka Pharmaceutical Development & Commercialization, Inc, 1 University Square Drive, Suite 500 Princeton, NJ 08540. Email: james.zhang@otsuka-us.com











