Discordance Between Measured vs Calculated Oxygen Consumption in Adults With Congenital Heart Disease: Limitations and Clinical Implications
Abstract
Introduction. Oxygen consumption (VO2) is frequently estimated using derived formulas for ease of use. We performed a prospective study to assess the correlation and limits of agreement between measured and assumed VO2 (mL/min) in adults with congenital heart disease (CHD). Methods. Consecutive adults with CHD who have undergone cardiac catheterization at Mayo Clinic Rochester from January 2018 to December 2019 were retrospectively enrolled in the study. Expired gas analysis was performed to measure VO2. We estimated VO2 using LaFarge (VO2L), Dehmer (VO2D), and Bergstra (VO2B) formulas. Bland-Altman and linear-regression analyses were used to assess the correlation between measured and calculated VO2. Results. We enrolled 84 patients with a mean age of 43 ± 12 years; 66% were men. Linear correlation analysis showed a moderate correlation of VO2L and VO2B with VO2M (r = 0.58; P<.001 and r = 0.44; P<.01, respectively) and good correlation of VO2D with VO2M (r = 0.79; P<.001). The large spread of limits of agreement assessed using Bland-Altman analysis demonstrates poor agreement of VO2L and VO2B with VO2M (bias, -58; 95% confidence interval [CI], -258 to 48) and bias, 38; 95% CI, -91 to 167, respectively). The limits of agreement for VO2D have a large spread, demonstrating a low degree of agreement with VO2M (bias, 13; 95% CI, -64 to 89). VO2L predominantly misclassified patients as low cardiac index and VO2D and VO2B misclassified patients as normal/high cardiac index. Conclusion. In adults with CHD, assumed VO2 will lead to a significant error while assessing invasive intracardiac hemodynamics.
J INVASIVE CARDIOL 2021;33(2):E100-E107. doi:10.25270/jic/20.00387
Key words: assumed oxygen consumption, measured oxygen consumption
Accurate estimation of cardiac output (CO) is of paramount importance during the invasive hemodynamic evaluation of patients with cardiovascular disorders. Of all the different methods used for CO assessment in clinical practice, the Fick method is considered the gold standard.1,2 The Fick method requires the measurement of oxygen consumption (VO2).3-5 However, its measurement is cumbersome and time consuming, and cannot accurately be measured in patients who are intubated and those requiring supplemental oxygen, hence making it difficult to use in routine clinical practice. As a result, VO2 is frequently estimated using derived formulas from published literature.6-9 The generalizability of the formulas is questionable since they were derived in a small, select population of patients who are similarly aged, ethnically homogenous, and lean, which is unlike the broadly heterogeneous patients encountered in routine clinical practice.10-12 Moreover, some formulas were derived exclusively in children and infants.6,8
In adults, studies have shown poor correlation and agreement between measured VO2 and assumed VO2. Measured VO2 (VO2M) was shown to overestimate the lower values of VO2 and underestimate the higher values of VO2.13,14 This error in VO2 estimation will significantly impact hemodynamic calculations and affect clinical decision making in decompensated patients. The correlation between VO2M and assumed VO2 has never been tested in adults with congenital heart disease (CHD), who commonly have sarcopenia,15 impaired isometric muscle strength and muscle endurance capacity,16-18 high prevalence of obesity,19-21 and anasarca. This would significantly impact the calculation of cardiac output (CO) using VO2M, since VO2M would tend to overestimate systemic oxygen consumption for a given weight, thereby causing a significant error in assessing hemodynamics in this fragile population. Therefore, we performed a prospective study to assess the correlation and limits of agreement between VO2M and assumed VO2¸ CO, and pulmonary vascular resistance (PVR).
Methods
Study population and data collection. Consecutive adults with CHD who have undergone cardiac catheterization at Mayo Clinic Rochester from January 2018 to December 2019 were retrospectively enrolled in the study. Written informed consent was obtained from the patients to authorize the use of their data for the research study. The study was approved by the Mayo Clinic institutional review board. Patients who have undergone isolated percutaneous patent foramen ovale closure were excluded from the study. Demographic, clinical, and echocardiographic characteristics were extracted from the patient’s electronic medical record. Height and weight were measured at the time of cardiac catheterization.
Study design. We hypothesized that there is a poor correlation between VO2M vs calculated VO2 in adults with CHD and that the use of calculated VO2 would result in imprecise CO assessment. The primary study objective was to assess the correlation between VO2M vs assumed VO2 in patients undergoing cardiac catheterization. The secondary study objective was to assess the correlation between CO and PVR derived from VO2M vs assumed VO2.
Cardiac catheterization and expired gas analysis. Patients on chronic medications underwent cardiac catheterization in a fastened state with mild sedation in a supine position. A 7 Fr fluid-filled catheter (Balloon Wedge, Arrow; Teleflex) was used for measurement on intracardiac pressures, including right atrial pressure (RAP), pulmonary artery pressure (PAP), and pulmonary artery wedge pressure (PAWP). Catheter position was confirmed using characteristic pressure tracing/waveforms, fluoroscopy, and/or oximetry. Systemic arterial oxygen saturations and pressures were measured during left heart catheterization (radial/femoral cannulation) using 4 to 6 Fr sheaths. Mixed venous oxygen saturation was obtained from a pulmonary artery (PA) blood sample unless an intracardiac shunt was present. In this case, mixed venous saturation was obtained from the chamber preceding the location of the shunt. All pressure measurements were averaged over at least 5 beats. All hemodynamic tracings obtained during the study procedure were digitized (240 Hz) and stored for offline analysis by an investigator. Computer-averaged PAWP and mean RAP were obtained. Expired gas analysis (MedGraphics) was performed to measure VO2. The difference between PA oxygen content and systemic arterial oxygen content is measured directly to assess the average difference in oxygen consumption (AVO2diff). CO was measured using the Fick equation. The cardiac index (CI) was calculated by the following formula: cardiac output/body surface area. PVR was calculated by the following formula: (mean PAP – left atrial/PAWP)/CO.
Estimated VO2. Body surface area (BSA) was estimated using the Dubois formula22 (BSA [m2] = 0.007184 × weight [kg]0.425 × height [cm]0.725). We estimated VO2 using three published formulas: (1) the Dehmer formula7, 9 (VO2D = 125 × BSA); (2) the LaFarge formula6 (VO2L = 138.1 – [K × lnage] + [0.378 × heart rate] × BSA), where Kwomen = 17.04 and Kmen = 11.49; and (3) the Bergstra formula8 (VO2B = 157.3 × BSA + K – [lnage × 10.5] + 4.8), where Kwomen = 0 and Kmen = 10.
Statistical analysis. Categorical variables were presented as counts (%) and continuous variables were presented as mean ± standard deviation or median (interquartile range [IQR]). Bland-Altman and linear-regression analyses were used to assess the correlation between measured and calculated VO2, CO, and PVR. We calculated the proportion of patients misclassified as a low CI (<2.5 L/min/m2) and normal/high CI when comparing CIs calculated using estimated VO2 with VO2M. A P-value <.05 was considered statistically significant. All statistical analyses were performed with JMP software, version 14.0 (SAS Institute) and Prism, version 7.0 (GraphPad).
Results
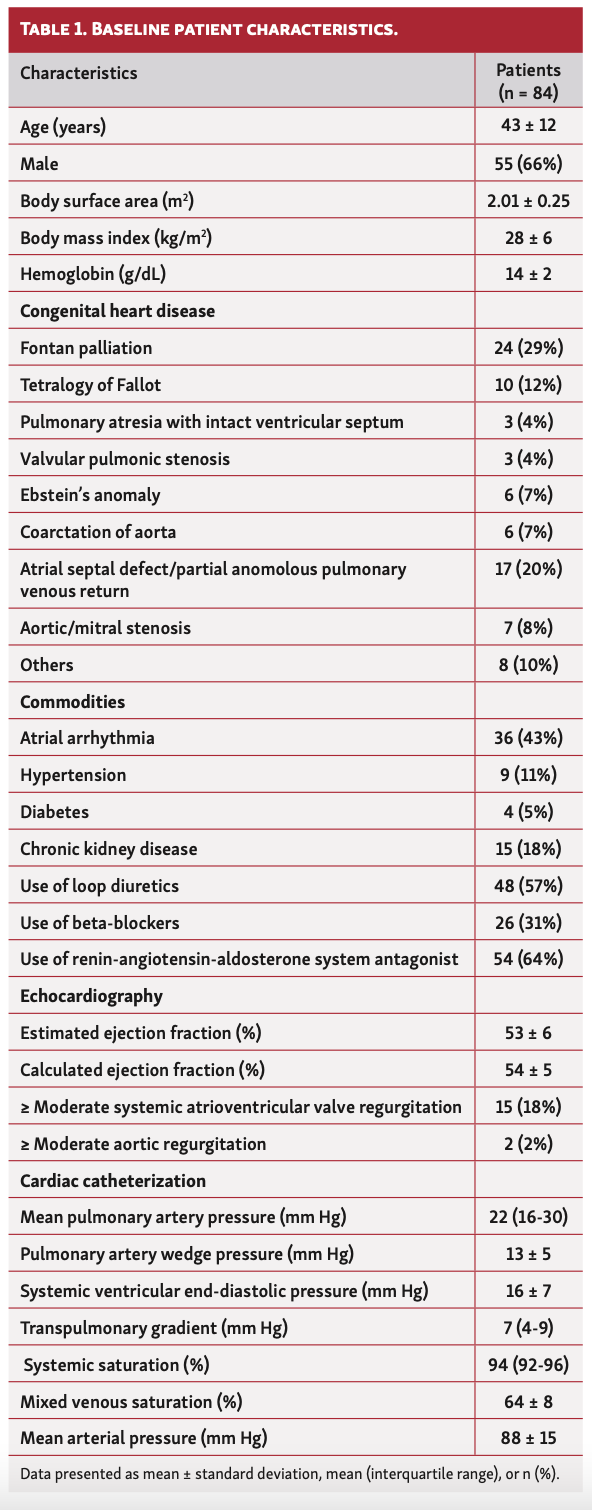
Baseline characteristics. Eighty-four patients were enrolled in the current study, with a mean age of 43 ± 12 years; 66% were men. The mean body mass index and BSA of patients in the cohort were 28 ± 6 kg/m2 and 2.01 ± 0.25 m2, respectively. The cohort comprised patients with history of Fontan palliation (n = 24; 29%), tetralogy of Fallot (n = 10; 12%), atrial septal defect or partial anomalous pulmonary venous return (PAPVR) (n = 17; 20%), pulmonary atresia with ventricular septal defect (n = 3; 4%), valvular pulmonic stenosis (n = 3; 4%), Ebstein’s anomaly (n = 6; 7%), and coarctation of aorta (n = 6; 7%) (Table 1).
Commonly noted comorbidities included atrial arrhythmias (43%), hypertension (11%), diabetes mellitus (5%), and chronic kidney disease (18%). The use of loop diuretics, beta-blockers, and renin-angiotensin-aldosterone system antagonists was seen in 57%, 31%, and 64%, respectively. The mean left ventricular ejection fraction was 54 ± 5% and ≥ moderate systemic atrioventricular valve (AVV) regurgitation was noted in 15 patients (18%). Only 2 patients (2%) had ≥ moderate aortic regurgitation.
Cardiac hemodynamics. The median PA pressure was 22 mm Hg (IQR, 16-30 mm Hg) and mean PAWP was 13 ± 5 mm Hg. The mean arterial pressure was 88 ± 15 mm Hg, mean systemic ventricular end-diastolic pressure (VEDP) was 16 ± 7 mm Hg, and median transpulmonary gradient was 7 mm Hg (IQR, 4-9 mm Hg). The mean mixed venous saturation was 64 ± 8% and the median systemic saturation was 94% (92%-96%).
Measured and assumed oxygen consumption. The mean VO2 calculated using LaFarge (VO2L) and Bergstra (VO2B) formulas was lower (88 ± 9 mL/min vs 122 ± 22 mL/min; P<.05) and higher (155 ± 12 mL/min vs 122 ± 22 mL/min; P<.05), respectively, than the VO2M among all patients in the study cohort. This difference was consistent in the subgroups of patients with Fontan and biventricular physiology. There was no significant difference between VO2 calculated using Dehmer formula (VO2D) with the VO2M (124 ± 17 mL/min vs 122 ± 22 mL/min; P=NS) (Table 2).
Linear correlation analysis showed a moderate correlation of VO2L and VO2B with VO2M (r = 0.58; P<.001 and r = 0.44; P<.01, respectively) (Figures 1A and 1E) and good correlation of VO2D with VO2M (r = 0.79; P<.001) (Figure 1C). The large spread of limits of agreement assessed using Bland-Altman analysis demonstrates poor agreement of VO2L and VO2B with VO2M (bias, -58 [95% confidence interval (CI), -258 to 48]; and bias, 38 [95% CI, -91 to 167], respectively). The VO2L tended to systematically overestimate lower levels of VO2M. In contrast, VO2B tended to underestimate higher VO2M (Figures 1B and 1F). The limits of agreement for VO2D have a large spread, demonstrating a low degree of agreement with VO2M (bias, 13; 95% CI, -64 to +89). A systematic error of underestimating higher VO2 was noted with VO2D (Figure 1D).
Cardiac output. The mean CO calculated using the LaFarge (QL) and Bergstra (QB) formulas was lower (3.4 ± 0.8 L/min vs 4.6 ± 1.6 L/min) and higher (6.0 ± 1.5 L/min vs 4.6 ± 1.6 L/min), respectively, than the CO derived using VO2M (QM). This difference was consistent in the subgroups of patients with Fontan and biventricular physiology. There was no significant difference noted in the CO calculated using the Dehmer formula (QD) compared with QM (Table 2).
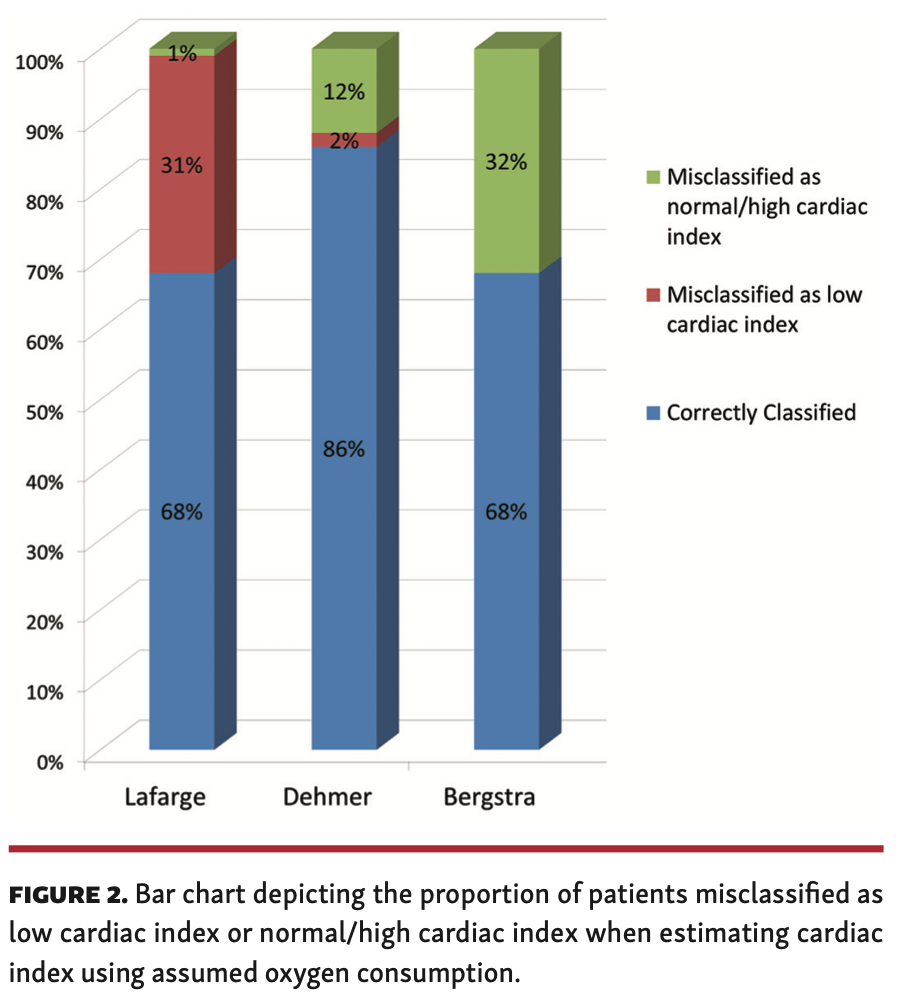
Using VO2M, a total of 38 patients (45%) had low output (CI <2.5 L/min/m2) while 46 patients (55%) had normal/high output. Figure 2 shows the misclassification into low CI vs misclassification into normal/high CI when CI is derived from estimated VO2. The patients were more likely to be misclassified as having low CI using the Lafarge formula (31%). On the other hand, patients were more likely to be misclassified as having normal/high CI using the Bergstra (32%) and Dehmer (12%) formulas. The Dehmer formula had the test tendency for CO misclassification.
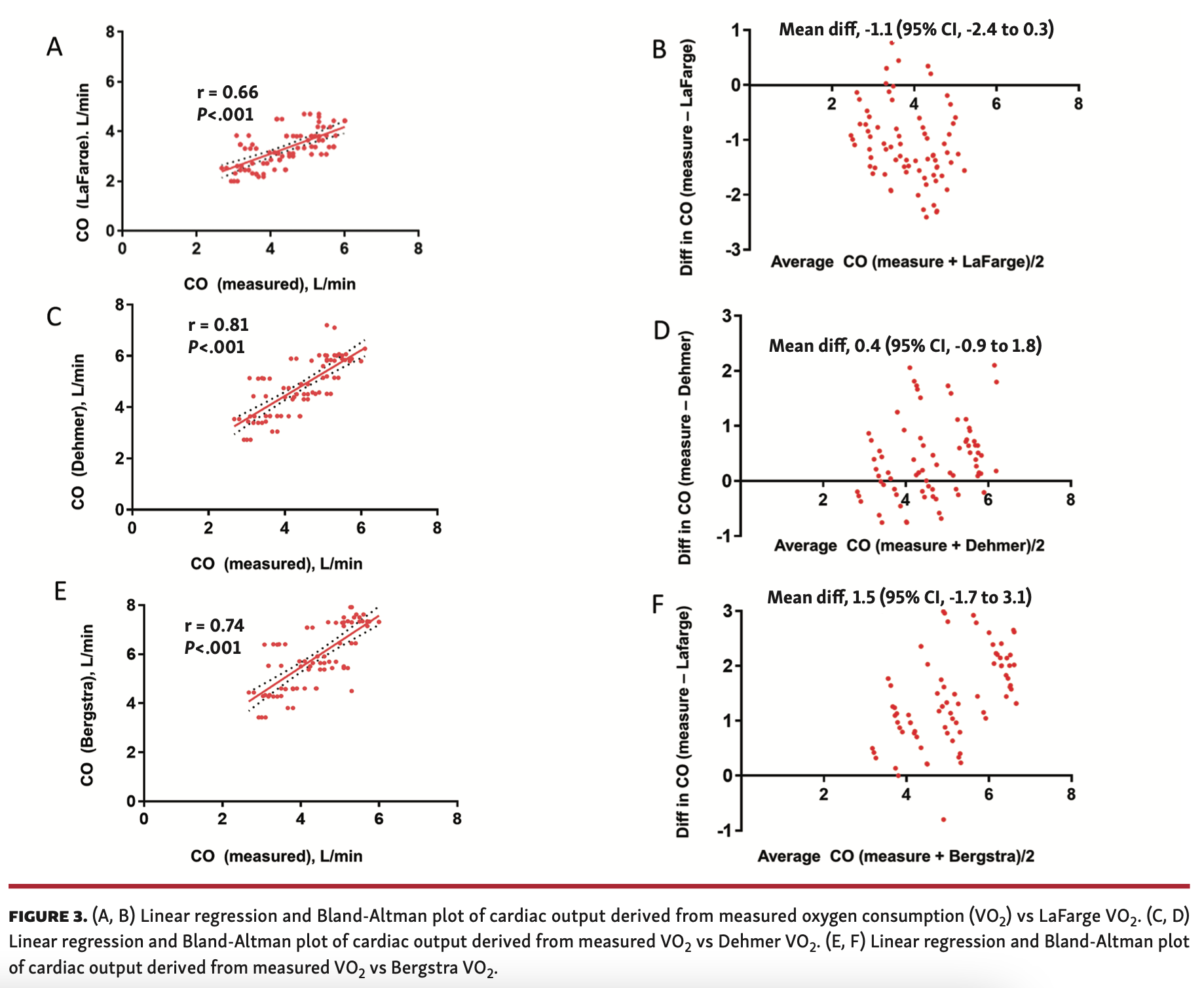
Linear correlation analysis showed a moderate correlation of QL with QM (r = 0.66; P<.001) (Figure 3A) and good correlation of QD & QB with QM (r = 0.81; P<.001 and r = 0.74; P<.001, respectively) (Figures 3C and 3E). The large spread of limits of agreement assessed using Bland-Altman analysis demonstrates poor agreement of QL and QB with QM (bias, -1.1 [95% CI, -2.4 to 0.3] and bias, 1.5 [95% CI, -1.7 to 3.1], respectively). The QL tends to systematically overestimate lower and QB tends to underestimate higher CO values (Figures 3B and 3F). The limits of agreement for QD have a large spread demonstrating a low degree of agreement with QM (bias, 0.4 [95% CI, -0.9 to 1.8]). A systematic error of underestimating higher CO was noted with QD (Figure 3D).
Pulmonary vascular resistance. The median PVR of the cohort calculated using the LaFarge formula (PVRL) was higher than the PVR derived using VO2M (PVRM) (1.3 WU [IQR, 0.7-1.9 WU] vs 0.8 [IQR, 0.4-1.7 WU], respectively; P<.05). In contrast, PVR calculated using the Bergstra formula (PVRB) was lower than PVRM (0.7 WU [IQR, 0.4-0.7 WU] vs 0.8 WU [IQR, 0.4-1.7 WU]; P<.05). Once again, this difference was consistent in the subgroups of patients with Fontan and biventricular physiology. There was no significant difference noted in the calculated PVR using the Dehmer formula (PVRD) with PVRM. This finding was consistent in both the subgroups of patients with Fontan and biventricular physiology (Table 2).
Linear correlation analysis showed a good correlation of PVRL, PVRD, and PVRB with PVRM (r = 0.83, P<001; r = 0.71, P<.001; and r = 0.79, P<.001, respectively) (Figures 4A, 4C, and 4E). The large spread of limits of agreement assessed using Bland-Altman analysis demonstrates poor agreement of PVRL, PVRD, and PVRB with PVRM (bias, 0.3 [95% CI, -0.7 to 1.2]; bias, -0.1 [95% CI, -1.3 to 1.2]; and bias, -0.3 [95% CI, -1.3 to 0.7], respectively) (Figures 4B, 4D, and 4F). The PVRL systematically tends to underestimate at higher PVR and both PVRD and PVRB systematically tend to overestimate at lower PVR.
Discussion
This contemporary study comprising 84 adults with CHD undergoing cardiac catheterization was performed to assess the correlation and limits of agreement between VO2M and assumed VO2 (mL/min), CO (L/min), and PVR (WU). Our observations were: (1) moderate to good correlation of assumed VO2 with VO2M. (2) assumed VO2 overestimated lower values of VO2 and underestimated higher values of VO2; (3) patients were more likely to be misclassified as having low CI using the LaFarge formula (31%); (4) patients were misclassified as having normal/high CI using the Bergstra (32%) and Dehmer (12%) formulas and misclassified as having low CI using the LaFarge formula (31%); (5) there was good correlation of PVR calculated using assumed VO2 with PVR calculated using VO2M; and (6) PVR using assumed VO2 overestimated lower values of PVR and underestimated higher values of PVR.
The present observations are consistent with prior studies that tested a similar hypothesis in both adults,13,14 and children.23,24 Measurement of VO2 has an average error of 6%, with changes in breathing patterns during instrumentation and sedation being the most significant contributors to the error.25 Air leaks from mouthpiece and claustrophobia can occasionally cause an error in measuring VO2. Measuring VO2 can be time intensive, requires special training for personnel, is challenging in babies and children, and is expensive. In patients on supplemental oxygen and intubated, we cannot measure VO2 accurately. To address these difficulties, physicians started incorporating routine use of assumed VO2 in clinical practice.
The three formulas6,8,9 used to calculate assumed VO2 incorporated body weight and BSA without accounting for the degree of adiposity, as adipose tissue has minimal impact on systemic oxygen uptake. In patients with excess adiposity, increased respiratory efforts and the need for physical support influence resting VO2. For estimating maximal VO2, predictive formulas26,27 were developed to adjust for excess adiposity and improve its accuracy; however, its application in estimation of resting VO2 remains to be determined.
Studies have shown that 25%-60% of adults with CHD are overweight or obese.20,21 In patients with Fontan circulation, approximately 50% develop heart failure by adulthood.28 These patients frequently tend to have anasarca and chronic pedal edema,29 which could lead to falsely elevated body mass index/BSA and overestimation of oxygen consumption. Adults with CHD have lower muscle mass than healthy controls, especially in females with higher body mass index due to the chronic effects of CHD.15 This could likely be due to lower participation in structured exercise training and reduced response to exercise training.30,31 In patients with systolic heart failure, skeletal muscle has impaired oxidative adaptation to exercise and reduced oxidative capacity.32 In contrast, adult patients with CHD have chronic exposure to diminished CO, which could negatively affect growth, development, and metabolic activity of skeletal muscles in this select population.15,16,33 Therefore, a combination of impaired metabolic activity and lower mass of skeletal muscle in adults with CHD significantly impacts on the calculation of assumed VO2 using traditional formulas derived from a homogenous group of healthy participants.6,8,9
Hemodynamic calculations using assumed VO2 in our study had a systematic error of overestimating lower values of CO and PVR and underestimating higher values of CO and PVR due to poor agreement noted during Bland-Altman analysis. It is important to understand that limits of agreement are a representation of mean of measured-assumed cardiac output/PVR; therefore, most data will fall within the limits of 2 standard deviations. This does not imply that the agreement between both methods is acceptable. For example, the limits of agreement reach approximately 1 L/min between assumed and calculated CO and approximately 1 WU between assumed and calculated PVR. Patients were more likely to be misclassified as having normal/high CI using the Bergstra (32%) and Dehmer formulas (12%) and misclassified as having low CI using the LaFarge formula (31%). This is a high degree of variability that will impact clinical decision making, especially in decompensated patients, to determine the following: initiation of mechanical ventricular support; titration of inotropic support; initiation and monitoring patients on vasodilator therapy; and candidacy for a heart transplant or surgical repair. Most decompensated patients have extreme values that further increase the degree of error while using assumed VO2 for hemodynamic calculations. In patients with valvular heart disease (stenosis/regurgitation), intracardiac shunt, or pulmonary hypertension, accurate estimation of CO is of paramount importance. Determination of valve area in case of stenosis, as well as determination of regurgitant fraction for valve regurgitation and shunt ratio, systemic regurgitation, and PVR, will determine candidacy for an intervention (percutaneous/surgical).
Extreme care must be undertaken while applying regression-based models to predict biological variables before widespread clinical use. The test population used to generate a model must be heterogeneous and appropriately powered to improve the accuracy of prediction and generalizability. The LaFarge, Dehmer, and Bergstra formulas were generated using regression analysis in either a small homogenous population or children, which explains the wide margin of error when it is applied to a heterogeneous population, such as adults with CHD.
Study limitations. The limitations of the present study include: (1) small sample size; (2) interoperator variability in measuring VO2; (3) assessment of VO2M in the catheterization lab in a steady-state condition is less likely to be a true resting VO2, as the patient’s anxiety falsely elevates VO2M; (4) sedated patients have falsely low resting VO2; and (5) the lack of follow-up data to assess the impact of misclassification of CI on the prognosis of patients. The presence of a standard protocol likely minimized interoperator variability, as all technicians have undergone standard training to maintain consistency in measuring VO2.
Conclusion
In adults with CHD, assumed VO2 will lead to a significant error while assessing invasive intracardiac hemodynamics. This is especially exaggerated in patients who have extremely low or high CI and systemic resistance/PVR. The use of VO2M must be the standard practice in the catheterization laboratory for invasive assessment of hemodynamics in adults with CHD.
From the 1Department of Cardiovascular Medicine, Mayo Clinic Hospital, Phoenix, Arizona; and 2Department of Cardiovascular Medicine, Mayo Clinic, Rochester, Minnesota.
Funding: Dr Egbe is supported by the National Heart, Lung, and Blood Institute (NHLBI) grant K23 HL141448.
Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. The authors report no conflicts of interest regarding the content herein.
Manuscript accepted June 22, 2020.
Address for correspondence: Pradyumna Agasthi, MD, Mayo Clinic Hospital, 5777 East Mayo Boulevard, Phoenix, AZ 85054. Email: pradyumna_agasthi@hotmail.com
- Consolazio CF, Johnson RE, Pecora LJ. Physiological measurements. For use in the study of metabolic functions. Rep US Army Med Res Nutr Lab Denver. 1959:1-416.
- Grossman W. Grossman's Cardiac Catheterization, Angiography, and Intervention: Lippincott Williams & Wilkins; 2006.
- Laszlo G. Respiratory measurements of cardiac output: from elegant idea to useful test. J Appl Physiol (1985). 2004;96:428-437.
- Meriläinen PT. Metabolic monitor. Int J Clin Monit Comput. 1987;4:167-177.
- Webb P, Troutman SJ Jr. An instrument for continuous measurement of oxygen consumption. J Appl Physiol. 1970;28:867-871.
- LaFarge CG, Miettinen OS. The estimation of oxygen consumption. Cardiovasc Res. 1970;4:23-30.
- Summerhill EM, Baram M. Principles of pulmonary artery catheterization in the critically ill. Lung. 2005;183:209-219.
- Bergstra A, van Dijk RB, Hillege HL, Lie KI, Mook GA. Assumed oxygen consumption based on calculation from dye dilution cardiac output: an improved formula. Eur Heart J. 1995;16:698-703.
- Dehmer GJ, Firth BG, Hillis LD. Oxygen consumption in adult patients during cardiac catheterization. Clin Cardiol. 1982;5:436-440.
- Astrand I. The physical work capacity of workers 50-64 years old. Acta Physiol Scand. 1958;42:73-86.
- Robertson JD, Reid DD. Standards for the basal metabolism of normal people in Britain. Lancet. 1952;1:940-943.
- Astrand PO, Ryhming I. A nomogram for calculation of aerobic capacity (physical fitness) from pulse rate during sub-maximal work. J Appl Physiol. 1954;7:218-221.
- Wolf A, Pollman MJ, Trindade PT, Fowler MB, Alderman EL. Use of assumed versus measured oxygen consumption for the determination of cardiac output using the Fick principle. Cathet Cardiovasc Diagn. 1998;43:372-380.
- Narang N, Thibodeau JT, Levine BD, et al. Inaccuracy of estimated resting oxygen uptake in the clinical setting. Circulation. 2014;129:203-210.
- Sandberg C, Johansson K, Christersson C, Hlebowicz J, Thilén U, Johansson B. Sarcopenia is common in adults with complex congenital heart disease. Int J Cardiol. 2019;296:57-62.
- Sandberg C, Thilén U, Wadell K, Johansson B. Adults with complex congenital heart disease have impaired skeletal muscle function and reduced confidence in performing exercise training. Eur J Prev Cardiol. 2015;22:1523-1530.
- Kröönström LA, Johansson L, Zetterström AK, Dellborg M, Eriksson P, Cider Å. Muscle function in adults with congenital heart disease. Int J Cardiol. 2014;170:358-363.
- Diller GP, Dimopoulos K, Okonko D, et al. Exercise intolerance in adult congenital heart disease: comparative severity, correlates, and prognostic implication. Circulation. 2005;112:828-835.
- Andonian C, Langer F, Beckmann J, et al. Overweight and obesity: an emerging problem in patients with congenital heart disease. Cardiovasc Diagn Ther. 2019;9:S360-S368.
- Lerman JB, Parness IA, Shenoy RU. Body weights in adults with congenital heart disease and the obesity frequency. Am J Cardiol. 2017;119:638-642.
- Brida M, Dimopoulos K, Kempny A, et al. Body mass index in adult congenital heart disease. Heart. 2017;103:1250-1257.
- Du Bois D, Du Bois EF. A formula to estimate the approximate surface area if height and weight be known. 1916. Nutrition. 1989;5:303-311; discussion 312-313.
- Li J, Bush A, Schulze-Neick I, Penny DJ, Redington AN, Shekerdemian LS. Measured versus estimated oxygen consumption in ventilated patients with congenital heart disease: the validity of predictive equations. Crit Care Med. 2003;31:1235-1240.
- Fakler U, Pauli C, Hennig M, Sebening W, Hess J. Assumed oxygen consumption frequently results in large errors in the determination of cardiac output. J Thorac Cardiovasc Surg. 2005;130:272-276.
- Guyton AC. Cardiac output and its regulation. Circ Physiol. 1973:353-371.
- Wasserman K, Whipp BJ. Exercise physiology in health and disease. Am Rev Respir Dis. 1975;112:219-249.
- Wasserman K, Hansen JE, Sue DY, Stringer WW, Whipp BJ. Principles of exercise testing and interpretation: including pathophysiology and clinical applications. Med Sci Sports Exerc. 2005;37:1249.
- Stout KK, Broberg CS, Book WM, et al. Chronic heart failure in congenital heart disease: a scientific statement from the American Heart Association. Circulation. 2016;133:770-801.
- Rychik J, Atz AM, Celermajer DS, et al. Evaluation and management of the child and adult with Fontan circulation: a scientific statement from the American Heart Association. Circulation. 2019 July 1 (Epub ahead of print).
- Larsson L, Johansson B, Wadell K, Thilén U, Sandberg C. Adults with congenital heart disease overestimate their physical activity level. Int J Cardiol Heart Vasc. 2019;22:13-17.
- Hedlund ER, Lundell B, Villard L, Sjöberg G. Reduced physical exercise and health-related quality of life after Fontan palliation. Acta Paediatr. 2016;105:1322-1328.
- Southern WM, Ryan TE, Kepple K, Murrow JR, Nilsson KR, McCully KK. Reduced skeletal muscle oxidative capacity and impaired training adaptations in heart failure. Physiol Rep. 2015;3:e12353.
- Kato A. Muscle wasting is associated with reduced exercise capacity and advanced disease in patients with chronic heart failure. Future Cardiol. 2013;9:767-770.