Complete Heart Block Following Transcatheter Closure of Ruptured Sinus of Valsalva Aneurysm
ABSTRACT: Ruptured sinus of Valsalva aneurysm (RSVA), a rare form of left-to-right shunt, is being increasingly corrected with transcatheter closure (TCC) with promising immediate and mid-term results. Complete heart block (CHB) following TCC of perimembranous ventricular septal defect is a well-described albeit worrisome complication. We describe a hitherto unreported case of development of CHB following TCC of RSVA that responded to high-dose steroids. The patient continues to remain in sinus rhythm without CHB 6 months later.
J INVASIVE CARDIOL 2012;24(12):E314-E317
Key words: interventional, nonsurgical, congenital, fistula
____________________________________________
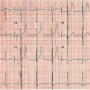 Case Report 1. A 40-year-old man presented with a 12-month history of progressive effort intolerance worsening to New York Heart Association class III over 2 months. He had no associated angina, orthopnea, paroxysmal nocturnal dyspnea, or pedal edema. Physical examination disclosed a collapsing pulse with wide pulse pressure, an elevated jugular venous pressure and a continuous murmur in the third space parasternally. Electrocardiogram (ECG) revealed sinus rhythm, normal QRS axis with a PR interval of 180 ms at heart rate of 70/min, and left ventricular hypertrophy by voltage criteria (Figure 1). The chest x-ray showed cardiomegaly and increased pulmonary vascularity. Transthoracic echocardiography (TTE) and transesophageal echocardiography (TEE) confirmed the diagnosis of a
Case Report 1. A 40-year-old man presented with a 12-month history of progressive effort intolerance worsening to New York Heart Association class III over 2 months. He had no associated angina, orthopnea, paroxysmal nocturnal dyspnea, or pedal edema. Physical examination disclosed a collapsing pulse with wide pulse pressure, an elevated jugular venous pressure and a continuous murmur in the third space parasternally. Electrocardiogram (ECG) revealed sinus rhythm, normal QRS axis with a PR interval of 180 ms at heart rate of 70/min, and left ventricular hypertrophy by voltage criteria (Figure 1). The chest x-ray showed cardiomegaly and increased pulmonary vascularity. Transthoracic echocardiography (TTE) and transesophageal echocardiography (TEE) confirmed the diagnosis of a 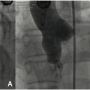 Ruptured sinus of Valsalva aneurysm (RSVA) from non-coronary sinus (NCS) shunting into the right atrium (RA). The RSVA measured 7 mm at the aortic origin and 6 mm at the RA end. There were no associated defects except trivial AR. After informed consent was obtained, the patient was taken up for transcatheter closure (TCC) under general anesthesia with continuous TEE monitoring, antibiotic prophylaxis, and systemic heparinization. Authors have conformed to institutional guidelines and those of the American Physiological Society during this work.
Ruptured sinus of Valsalva aneurysm (RSVA) from non-coronary sinus (NCS) shunting into the right atrium (RA). The RSVA measured 7 mm at the aortic origin and 6 mm at the RA end. There were no associated defects except trivial AR. After informed consent was obtained, the patient was taken up for transcatheter closure (TCC) under general anesthesia with continuous TEE monitoring, antibiotic prophylaxis, and systemic heparinization. Authors have conformed to institutional guidelines and those of the American Physiological Society during this work.
 The patient was started on 150 mg aspirin/day and clopidogrel 75 mg/day. At cardiac catheterization, the aortic and pulmonary pressures were 186/58 mm Hg (mean, 100 mm Hg) and 38/12 mm Hg (mean, 20 mm Hg), respectively. The baseline left-to-right shunt was 3:1. Aortic root angiography confirmed the diagnosis (Figure 2A).
The patient was started on 150 mg aspirin/day and clopidogrel 75 mg/day. At cardiac catheterization, the aortic and pulmonary pressures were 186/58 mm Hg (mean, 100 mm Hg) and 38/12 mm Hg (mean, 20 mm Hg), respectively. The baseline left-to-right shunt was 3:1. Aortic root angiography confirmed the diagnosis (Figure 2A).
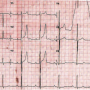
As described previously,1-3 the TCC procedure involves crossing the defect from the aortic side using a 5 Fr Judkin’s right coronary catheter and a 0.035˝ angled-tip glide wire (Terumo Inc), which is exchanged for a 300-cm long noodle wire (AGA Medical Corporation). A stable arteriovenous loop was established by snaring the noodle wire from the superior vena cava by a gooseneck snare (ev3 Endovascular Inc) and exteriorizing it from the femoral vein. Using a 6 Fr delivery sheath through the femoral vein, the 10 x 8 mm first-generation Amplatzer duct occluder (ADO; AGA Medical Corporation) was positioned across the defect. The ADO was released only after confirming on TEE as well as angiography the correct position of the ADO, not causing any significant residual shunting or any increase in aortic regurgitation. The procedure was successful (Figure 2B) and there were no conduction disturbances noticed. The patient was discharged on day 4 with an uneventful course, except for PR prolongation on predischarge ECG (PR interval of 240 ms at heart rate of 60/min) (Figure 3).
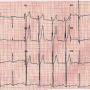
 A week later (on day 8), the patient presented with giddiness and uneasiness. Physical examination revealed bradycardia (pulse rate, 40/min), blood pressure of 90/70 mm Hg, and a grade 2/6 ejection systolic murmur in the second left intercostal space. The ECG showed a CHB with sinus rate of 90/min and a broad QRS ventricular escape rate of 40/min (Figure 4). Fluoroscopy and TTE showed no change in ADO position. There was no residual shunting. On intravenous fluids and temporary transvenous ventricular pacing at 90/min, the blood pressure improved to 120/90 mm Hg. The patient was started on oral prednisolone 20 mg thrice daily (1 mg/kg). After 48 hours of starting steroids, the patient
A week later (on day 8), the patient presented with giddiness and uneasiness. Physical examination revealed bradycardia (pulse rate, 40/min), blood pressure of 90/70 mm Hg, and a grade 2/6 ejection systolic murmur in the second left intercostal space. The ECG showed a CHB with sinus rate of 90/min and a broad QRS ventricular escape rate of 40/min (Figure 4). Fluoroscopy and TTE showed no change in ADO position. There was no residual shunting. On intravenous fluids and temporary transvenous ventricular pacing at 90/min, the blood pressure improved to 120/90 mm Hg. The patient was started on oral prednisolone 20 mg thrice daily (1 mg/kg). After 48 hours of starting steroids, the patient 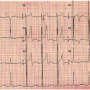 recovered with the ECG revealing a 1:1 atrioventricular (AV) conduction and a PR interval of 320 msec (Figure 5). The temporary pacing wire was removed after 4 days and a 24-hour ambulatory ECG after 7 days of steroid therapy showed a 1:1 AV conduction. The patient was discharged on the 8th day (postprocedure day 15) and predischarge treadmill stress test revealed a good chronotropic response with a maximum heart rate of 150/min (85% of THR) with a 1:1 AV conduction and PR interval shortening to 100 ms at peak exercise. Prednisolone was tapered off over next 3 weeks. Aspirin and clopidogrel were continued during the second admission and on follow-up for 6 months and 6 weeks, respectively. At the last follow-up (6 months since TCC), the ECG revealed sinus rhythm at 83/min with 1:1 AV conduction and a PR interval of 160 ms (Figure 6).
recovered with the ECG revealing a 1:1 atrioventricular (AV) conduction and a PR interval of 320 msec (Figure 5). The temporary pacing wire was removed after 4 days and a 24-hour ambulatory ECG after 7 days of steroid therapy showed a 1:1 AV conduction. The patient was discharged on the 8th day (postprocedure day 15) and predischarge treadmill stress test revealed a good chronotropic response with a maximum heart rate of 150/min (85% of THR) with a 1:1 AV conduction and PR interval shortening to 100 ms at peak exercise. Prednisolone was tapered off over next 3 weeks. Aspirin and clopidogrel were continued during the second admission and on follow-up for 6 months and 6 weeks, respectively. At the last follow-up (6 months since TCC), the ECG revealed sinus rhythm at 83/min with 1:1 AV conduction and a PR interval of 160 ms (Figure 6).
 Discussion. Successful TCC of RSVA is being increasingly reported with excellent immediate and mid-term results.1-3 The potential complications have been failure to deploy, residual shunting, and aortic regurgitation.3 These can be monitored and addressed intraprocedurally with TEE. In the present case, there were no intraprocedural clues to predict the CHB development, like PR interval prolongation or difficulty in tracking the sheath or the device across the defect. Although the occurrence of conduction disturbances, including a 2%-3% incidence of CHB, has been reported following surgical closure of RSVA,4 CHB following TCC is not described. At presentation, occurrence of CHB has been described more often with unruptured SVA than with RSVA,5-7 presumably due to higher pressure exerted by the former on the AV conduction system. Dissection of interventricular septum by the unruptured aneurysm is suggested as a possible mechanism.6,7 In one instance, the CHB due to an unruptured SVA improved following corrective surgery, suggesting mechanical compression of AV conduction system as the likely etiology.8 To our knowledge, this is the first case report of CHB following TCC of RSVA that responded to steroid therapy. It is possible that early steroid therapy for predischarge PR prolongation (Figure 3) may have averted progression to CHB later on day 8 (Figure 4).
Discussion. Successful TCC of RSVA is being increasingly reported with excellent immediate and mid-term results.1-3 The potential complications have been failure to deploy, residual shunting, and aortic regurgitation.3 These can be monitored and addressed intraprocedurally with TEE. In the present case, there were no intraprocedural clues to predict the CHB development, like PR interval prolongation or difficulty in tracking the sheath or the device across the defect. Although the occurrence of conduction disturbances, including a 2%-3% incidence of CHB, has been reported following surgical closure of RSVA,4 CHB following TCC is not described. At presentation, occurrence of CHB has been described more often with unruptured SVA than with RSVA,5-7 presumably due to higher pressure exerted by the former on the AV conduction system. Dissection of interventricular septum by the unruptured aneurysm is suggested as a possible mechanism.6,7 In one instance, the CHB due to an unruptured SVA improved following corrective surgery, suggesting mechanical compression of AV conduction system as the likely etiology.8 To our knowledge, this is the first case report of CHB following TCC of RSVA that responded to steroid therapy. It is possible that early steroid therapy for predischarge PR prolongation (Figure 3) may have averted progression to CHB later on day 8 (Figure 4).
The non-coronary sinus (NCS) of the aortic valve and membranous part of the interventricular septum are in close proximity to the AV conduction system9 and occurrence of CHB may complicate surgical closure or TCC of perimembranous VSD.10,11 Our case can be explained on similar grounds. This complication may be related to direct pressure by the device or local inflammation caused by trauma. Reversal of CHB following TCC of perimembranous VSD by high-dose steroids is well described and possible mechanism of benefit includes control of local inflammation by steroids.11,12 Conduction abnormalities are also seen after conventional aortic valve replacement or transcatheter aortic valve implantation (TAVI).13,14 Predictive models for AV conduction disturbance after TAVI have included the thickness of the non-coronary cusp (NCC), which also highlights the close proximity of the NCC to the AV conduction system.15 Therefore, it is possible that intervention on the NCS is more likely to cause conduction problems than on the right coronary sinus.
In our case, the possibility of inflammation around the device damaging the AV conduction system is more likely, since AV conduction deteriorated gradually and CHB was seen on postprocedure day 8. This is further supported by the patient’s response to steroids. Another possibility includes sudden increased diastolic pressure in the aorta following TCC, which may have led to expansion of the SVA and then compression of the AV conduction system. However, this possibility seems less likely, since it would not be reversible without any intervention.
In conclusion, during TCC of RSVA, AV conduction should be monitored carefully and early empirical steroid therapy should be considered in the rare event of development of AV conduction disturbance.
References
- Kerkar PG. Ruptured sinus of Valsalva aneurysm: Yet another hole to plug! Ann Pediatr Cardiol. 2009;2(1)83-84.
- Kerkar P, Suvarna T, Burkule N, et al. Transcatheter closure of ruptured sinus of Valsalva aneurysm using the Amplatzer duct occluder in a critically ill post-CABG patient. J Invasive Cardiol. 2007;19(6):E169-E171.
- Kerkar PG, Lanjewar CP, Mishra N, et al. Transcatheter closure of ruptured sinus of Valsalva aneurysm using the Amplatzer duct occluder: immediate results and mid-term follow-up. Eur Heart J. 2010;31(23):2881-2887.
- Nicholas TK, Eugene HB, Donald BD, et al. Congenital sinus of Valsalva aneurysm and aortico-left ventricular tunnel. In: Cardiac Surgery. 3rd ed. Philadelphia, PA: Churchill Livingston, Elsevier; 2003:911-927.
- Nitya N, Hitender K. Ruptured sinus of Valsalva with complete heart block. JIACM. 2011;12(3):236-238.
- Choudhary S, Bhan A, Reddy SCB, et al. Aneurysm of sinus of Valsalva dissecting into interventricular septum. Ann Thorac Surg. 1998;65(3):735-740.
- Metras D, Coulibaly AO, Ouattara K, et al. Calcified unruptured aneurysm of sinus of Valsalva with complete heart block and aortic regurgitation. Successful repair of one case. Br Heart J. 1982;48(5):507-509.
- Alasti M, Omidvar B, Mali S, et al. Sinus of Valsalva aneurysm: a reversible cause of complete heart block. Tex Heart Inst J. 2011;38(4):450-451.
- Sutton JP 3rd, Ho SY, Anderson RH. The forgotten interleaflet triangles: a review of the surgical anatomy of the aortic valve. Ann Thorac Surg. 1995;59(2):419-427.
- Smerup M, Hjertholm T, Johnsen SP, et al. Pacemaker implantation after congenital heart surgery: risk and prognosis in a population-based follow-up study. Eur J Cardiothorac Surg. 2005;28(1):61-68.
- Butera G, Carminati M, Chessa M, et al. Transcatheter closure of perimembranous ventricular septal defects: early and long-term tesults. J Am Coll Cardiol. 2007;50(12):1189-1189.
- William CL, Zimmerman F, Hijazi ZM. Heart block and empirical therapy after transcatheter closure of perimembranous ventricular septal defect. Catheter Cardiovasc Interv. 2005;66(3):436-441.
- Rodrigo B, Manazzoni JM, Dumont E, et al. Permanent pacemaker implantation following isolated aortic valve replacement in a large cohort of elderly patients with severe aortic stenosis. Heart. 2011;97(20):1687-1694.
- Maugenest, Anderson RH, Peter P, et al. Early and persistent intraventricular conduction abnormalities and requirements for pacemaking after percutaneous replacement of the aortic valve. JACC Cardiovasc Interv. 2008;1(3):310-316.
- Jilaihawi H, Chin D, Nicotera MV, et al. Predictors for permanent pacemaker requirement after transcatheter aortic valve implantation with the CoreValve bioprosthesis. Am Heart J. 2009;157(5):860-866.
________________________________________
From the Seth GS Medical College and KEM Hospital, Department of Cardiology, Asian Heart Institute, Mumbai, India.
Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. The authors report no conflicts of interest regarding the content herein.
Manuscript submitted May 15, 2012 and accepted June 12, 2012.
Address for correspondence: Prafulla G. Kerkar, Professor and Head, Department of Cardiology, Seth GS Medical College and KEM Hospital, E. Borges Road, Mumbai, Maharashtra, India 400012, Maharashtra 400051, India. Email: prafullakerkar@rediffmail.com












