ADVERTISEMENT
Assessment of Inflammatory Response to Transfemoral Transcatheter Aortic Valve Implantation Compared to Transapical and Surgical Procedures: A Pilot Study
Abstract: Aims. Surgical aortic valve replacement (AVR) has been associated with systemic inflammatory reactions. Yet, the role of inflammation following transcatheter aortic valve implantation (TAVI) has not been fully elucidated. Methods and Results. In a total of 40 patients evaluated by the ‘heart team,’ this retrospective study assessed levels of high-sensitive C-reactive protein (hs-CRP) and leukocyte counts following ‘uneventful’ AVR and TAVI. Four groups of matched patients were compared (AVR; transapical and transfemoral Edwards SAPIEN [TA ES and TF ES, respectively]; and transfemoral Medtronic CoreValve [TF CV]). A postprocedural increase of both hs-CRP levels and leukocyte counts was observed (P<.001) with peak levels 48 hours after the procedures. Comparing treatment groups, hs-CRP levels at 48 hours were significantly higher following AVR and TA ES compared to TF ES and TF CV (P<.04). Leukocyte counts at 48 hours were higher following TA ES compared to TF ES and TF CV (P<.03). Multivariate analysis incorporating both hs-CRP levels and leukocyte counts confirmed significant differences for all measurements over time (P<.001). Furthermore, the treatment group significantly influenced postprocedural hs-CRP levels and leukocyte counts (P<.001). Conclusion. Both AVR and TAVI evoke a postprocedural inflammatory response. Higher hs-CRP levels and leukocyte counts following AVR and apical TAVI suggest less inflammation following femoral procedures.
J INVASIVE CARDIOL 2012;24(8):407-411
Key words: inflammation, aortic stenosis, transcatheter aortic valve implantation
_________________________________________________
Degenerative aortic stenosis (AS) is the most common isolated valve disease in Western communities.1 Without any treatment, patients with severe symptomatic AS usually die within 2 to 3 years.2,3 Surgical aortic valve replacement (AVR) is the standard treatment for these patients.3,4 However, in recent years, transcatheter aortic valve implantation (TAVI) has emerged as a less invasive treatment option for elderly high-risk individuals not amenable to open heart surgery.5-7 Moreover, TAVI is of value in high-risk patients, as demonstrated in a most recent randomized trial with comparable 1-year survival rates to AVR.8 Yet, there appeared to be essential differences in the periprocedural risks of these two therapies.
Systemic inflammatory responses of different severity have been observed following major surgical procedures including heart surgery.9-11 Indeed, elevated levels of inflammatory markers such as high-sensitive C-reactive protein (hs-CRP), interleukins, and neutrophil elastase have been noted in patients after AVR.11,12 The extent of this inflammatory response has been correlated to postoperative complications and worse short- and long-term outcomes in many cases,10,13-15 and attempts in minimizing these reactions with the use of different extracorporeal circulatory support systems or perioperative steroid treatment have been undertaken in adult and pediatric patients.10,12,13,16 Hence, elevated levels of inflammatory markers might exhibit a sort of postprocedural ‘alert system’ with the potential to identify patients at risk for adverse events.
Yet, the occurrence, severity, and variation in time of postinterventional systemic inflammatory responses following TAVI have not been elucidated. Hence, this study aims to characterize postprocedural inflammation following transfemoral and transapical TAVI in comparison to AVR focusing on hs-CRP levels and leukocyte counts.
Methods
Patients. Out of a cohort of 130 consecutive patients undergoing TAVI at our institution between May 2008 and October 2010, thirty cases could be matched to 10 consecutive cases evaluated for TAVI by the heart team, but finally elected for AVR. Matching criteria were age, gender, normal renal function, normal left ventricular ejection fraction, coronary artery disease, atrial fibrillation, and other baseline characteristics. Only patients with uneventful procedures were included in the study. Uneventful procedures were defined according to the combined safety endpoint definition of the valve academic research consortium (VARC).17 Accordingly, patients with death, major stroke, life-threatening or disabling bleeding, acute kidney injury (stage 3 including renal replacement therapy), periprocedural myocardial infarction, major vascular complications, or repeat procedures for valve-related dysfunction (surgical or interventional therapy) were excluded from the study. Four groups of matched patients were compared (AVR; transapical and transfemoral Edwards SAPIEN [TA ES, and TF ES, respectively]; and transfemoral Medtronic CoreValve [TF CV]).
All patients were evaluated by the heart team, a multidisciplinary team consisting of at least one interventional and clinical cardiologist, at least one cardio-thoracic surgeon, one cardiac anesthesiologist, and one imaging specialist for possible TAVI. Severe AS was defined as either a mean transaortic systolic pressure gradient of ≥40 mm Hg or an aortic valve area of <1.0 cm2 or <0.6 cm2/m2. All procedures were performed under general anesthesia. Patients not amenable to open heart surgery were selected for TAVI in accordance with the recommendations of the European Association of Cardio-Thoracic Surgery (EACTS), the European Society of Cardiology (ESC), and the European Association of Percutaneous Cardiovascular Interventions (EAPCI).18 Written informed consent was obtained. Transapical access was used in patients with small iliofemoral arteries due to atherosclerosis or small body size. The Medtronic CoreValve (26 and 29 mm; Medtronic), and the Edwards SAPIEN TFX 9000 (23 and 26 mm; Edwards Lifesciences) prostheses were utilized.19 Both transfemoral and transapical procedures were performed in the cardiac catheterization laboratory as previously described.20
Blood sampling and laboratory methods. Levels of hs-CRP and leukocytes at baseline, as well as 24, 48, and 72 hours after AVR and TAVI were retrospectively assessed. For hs-CRP measurements, peripheral blood samples were harvested in serum tubes. Serum was obtained by centrifugation at 2800 xG for 10 minutes at 17 °C. Serum hs-CRP levels were immediately assessed using a commercially available test from Roche Diagnostic Systems (CRPL3) on a Roche/Hitachi modular clinical chemistry analyzer (Roche Diagnostic Systems) with maximal coefficients of variation of 5.5%. High sensitive CRP concentrations ≤5 mg/L were considered to be within the reference range.
For leukocyte quantification, peripheral blood was collected in EDTA tubes. Leukocyte counts were assessed automatically using the ADVIA 2120 analyzer (Siemens Healthcare Diagnostics GmbH) with maximal coefficients of variation of 2.4%. Leukocyte counts between 3.0-9.6 1000/µL were considered to be within the reference range. All analyses were performed at the University Hospital Zürich, Zürich, Switzerland.
Statistical analysis. To assess inflammatory responses among groups over time, doubly multivariate repeated measures analyses (Hotelling’s Trace test and Roy’s Largest Root test) were utilized. When multivariate analysis of variance (MANOVA) revealed significant differences, univariate analysis of variance (ANOVA) and repeated measures ANOVA were conducted for each inflammatory marker separately. In case of significant results, within- and between-subjects effects were identified by additional t-testing subjected to Bonferroni’s adjustment for multiple comparisons. The assumptions of normality and homoscedasticity were evaluated using Shapiro-Wilk’s goodness of fit test and Mauchly’s tests for sphericity, respectively. The Greenhouse-Geisser correction was applied when necessary.
Relationships among groups are visualized by profile plots (interaction plots), which display the estimated marginal means (estimates of predicted mean values for the cells) in the model. Continuous variables are presented either as mean ± standard deviation (SD) or as median and interquartile range (IQR), as appropriate. Categorical variables are given as frequencies and percentages. Differences among categorical variables were analyzed by the Pearson’s chi-square test and the Fisher’s exact test, respectively. A two-sided P-value of <.05 was considered statistically significant. All statistical analyses were performed using JMP version 8.0.1 (SAS Institute Inc).
Results
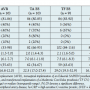 Baseline characteristics. Baseline characteristics are summarized in Table 1. Median age of the patients (43% male) was 83 years (IQR, 81-87). There were no differences among the groups except for median logistic EuroSCORE (7.7 [IQR, 6.8-12.0] for AVR vs 22.5 [IQR, 17.0-27.4] for TA ES, 29.0 [IQR, 19.5-32.9] for TF ES, and 16.6 [IQR, 12.4-28.1] for TF CV, respectively; P=.0005) and peripheral vascular disease, which was more frequent in patients with transapical procedures (P=.0002). Median baseline creatinine level was 83 µmol/L [IQR, 64-105]. Regarding baseline anti-inflammatory medication, 12 patients (30%) were on statins and 1 patient (2.5%) was on steroids due to chronic polyarthritis. Acetylsalicylic acid was administered in 28 patients (70%), and clopidogrel in 5 patients (13%).
Baseline characteristics. Baseline characteristics are summarized in Table 1. Median age of the patients (43% male) was 83 years (IQR, 81-87). There were no differences among the groups except for median logistic EuroSCORE (7.7 [IQR, 6.8-12.0] for AVR vs 22.5 [IQR, 17.0-27.4] for TA ES, 29.0 [IQR, 19.5-32.9] for TF ES, and 16.6 [IQR, 12.4-28.1] for TF CV, respectively; P=.0005) and peripheral vascular disease, which was more frequent in patients with transapical procedures (P=.0002). Median baseline creatinine level was 83 µmol/L [IQR, 64-105]. Regarding baseline anti-inflammatory medication, 12 patients (30%) were on statins and 1 patient (2.5%) was on steroids due to chronic polyarthritis. Acetylsalicylic acid was administered in 28 patients (70%), and clopidogrel in 5 patients (13%).
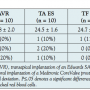 Periprocedural characteristics. Mean prosthetic valve size was smaller in patients with AVR compared to those treated with TAVI (22.8 ± 2.0 mm for AVR vs 24.5 ± 1.6 mm for TA ES, 24.7 ± 1.6 mm for TF ES, and 27.4 ± 1.6 mm for TF CV; P<.03). Among TAVI patients, largest diameters were observed with TF CV (P<.004). Otherwise, periprocedural characteristics did not differ, in particular packed red blood cell transfusions, new permanent pacemaker implantation within 72 hours, and need for reintubation (Table 2). Creatinine levels did not significantly increase following the procedures (P=.1) without significant differences among the 4 groups at 48 hours (P=.16). Postprocedurally, 27 patients (90%) following TAVI had dual anti-platelet therapy, 2 patients (7%) oral anti-coagulation and clopidogrel, and 1 patient (3%) oral anti-coagulation and acetylsalicylic acid. Following AVR, besides oral anti-coagulation for 3 months, 9 patients (90%) had acetylsalicylic acid and 1 patient (10%) had clopidogrel. All-cause mortality at 30 days was 2.5%. One patient died suddenly during the third postprocedural (TF CV) day after an initially uneventful course.
Periprocedural characteristics. Mean prosthetic valve size was smaller in patients with AVR compared to those treated with TAVI (22.8 ± 2.0 mm for AVR vs 24.5 ± 1.6 mm for TA ES, 24.7 ± 1.6 mm for TF ES, and 27.4 ± 1.6 mm for TF CV; P<.03). Among TAVI patients, largest diameters were observed with TF CV (P<.004). Otherwise, periprocedural characteristics did not differ, in particular packed red blood cell transfusions, new permanent pacemaker implantation within 72 hours, and need for reintubation (Table 2). Creatinine levels did not significantly increase following the procedures (P=.1) without significant differences among the 4 groups at 48 hours (P=.16). Postprocedurally, 27 patients (90%) following TAVI had dual anti-platelet therapy, 2 patients (7%) oral anti-coagulation and clopidogrel, and 1 patient (3%) oral anti-coagulation and acetylsalicylic acid. Following AVR, besides oral anti-coagulation for 3 months, 9 patients (90%) had acetylsalicylic acid and 1 patient (10%) had clopidogrel. All-cause mortality at 30 days was 2.5%. One patient died suddenly during the third postprocedural (TF CV) day after an initially uneventful course.
High-sensitive CRP levels following AVR and TAVI. Mean hs-CRP level at baseline was 4.2 ± 0.8 mg/L without any differences among the 4 groups (P=.8). After the interventions, a rise of hs-CRP levels was observed peaking at 48 hours (P<.0001). Analyzing the 4 treatment groups separately, different time courses were observed. Following AVR and TA ES, significant increases in hs-CRP levels were observed at 24 hours (54.8 ± 6.6 mg/L vs 4.1 ± 1.4 mg/L for AVR [P<.0001] and 44.9 ± 5.8 mg/L vs 3.3 ± 1.6 mg/L for TA ES [P=.001]). Peak levels of up to 50-fold were reached at 48 hours (162.9 ± 19.5 mg/L vs 4.1 ± 1.4 mg/L for AVR [P<.0001] and 177.0 ± 21.5 mg/L vs 3.3 ± 1.6 mg/L for TA ES [P<.001]). Following TF ES and TF CV, responses of hs-CRP were less rapid reaching maximal levels of up to 20-fold after 72 hours (81.1 ± 13.6 mg/L vs 4.2 ± 1.9 mg/L for TF ES [P=.012] and 81.1 ± 18.1 mg/L vs 5.2 ± 1.7 mg/L for TF CV [P=.31]). Comparing treatment groups, hs-CRP levels at 48 hours were significantly higher following AVR and TA ES compared to TF ES and TF CV (P<.04) with a similar trend at 72 hours (P=.07 for AVR vs TF ES, and P=.09 for AVR vs TF CV; P=.03 for TA ES vs TF CV, and P=.045 for TA ES vs TF ES).
 Leukocyte counts following AVR and TAVI. Mean leukocyte count at baseline was 7.6 ± 0.4 1000/µL without significant differences among the 4 groups (P=.4). A significant postprocedural increase was observed with peak levels at 48 hours (P=.001). Similar to changes in hs-CRP levels, significant increases in leukocyte counts were observed after 48 hours in patients with AVR and TA ES (11.4 ± 0.9 1000/µL vs 6.8±0.4 1000/µL for AVR [P<.0001] and 12.7 ± 1.2 1000/µL vs 8.7 ± 1.2 1000/µL for TA ES [P=.03]). In patients with TF ES and TF CV, postprocedural changes in leukocyte counts were not significant over 72 hours in univariate analysis. Between-groups analysis at 48 hours showed higher leukocyte counts following TA ES compared to TF ES and TF CV (P<.03).
Leukocyte counts following AVR and TAVI. Mean leukocyte count at baseline was 7.6 ± 0.4 1000/µL without significant differences among the 4 groups (P=.4). A significant postprocedural increase was observed with peak levels at 48 hours (P=.001). Similar to changes in hs-CRP levels, significant increases in leukocyte counts were observed after 48 hours in patients with AVR and TA ES (11.4 ± 0.9 1000/µL vs 6.8±0.4 1000/µL for AVR [P<.0001] and 12.7 ± 1.2 1000/µL vs 8.7 ± 1.2 1000/µL for TA ES [P=.03]). In patients with TF ES and TF CV, postprocedural changes in leukocyte counts were not significant over 72 hours in univariate analysis. Between-groups analysis at 48 hours showed higher leukocyte counts following TA ES compared to TF ES and TF CV (P<.03).
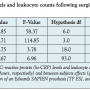 Multivariate analysis of postprocedural inflammatory responses. Multivariate analysis incorporating both, hs-CRP levels and leukocyte counts, revealed significant differences for all measurements over time (P<.001). Furthermore, the procedure group significantly influenced hs-CRP levels and leukocyte counts (P<.001; Table 3). Estimated marginal means of both hs-CRP levels and leukocyte counts were calculated to visualize these differences (Figure 1).
Multivariate analysis of postprocedural inflammatory responses. Multivariate analysis incorporating both, hs-CRP levels and leukocyte counts, revealed significant differences for all measurements over time (P<.001). Furthermore, the procedure group significantly influenced hs-CRP levels and leukocyte counts (P<.001; Table 3). Estimated marginal means of both hs-CRP levels and leukocyte counts were calculated to visualize these differences (Figure 1).
Discussion
This study demonstrates that both AVR and TAVI evoke a postprocedural inflammatory response. High-sensitive CRP levels and leukocyte counts were higher following AVR and transapical TAVI compared to transfemoral procedures.
Systemic inflammatory reactions are frequently observed following heart surgery.14,21 The pathogenesis of postoperative inflammation appears to be multifactorial. Indeed, direct mechanical trauma, ischemia-reperfusion injury, and contact of blood components to foreign surfaces of cardio-pulmonary bypass systems have been implicated.22 However, as CRP levels did not differ between variable cardio-pulmonary bypass types, between procedures with and without use of a cardio-pulmonary bypass, and even between coronary artery bypass grafting and surgery for non-specific thoracic masses, the exposure of blood to non-physiological surfaces as it occurs with the use of a cardio-pulmonary bypass may be less important as previously expected.12,16,21,23 A comparable increase of hs-CRP levels following transapical TAVI and AVR further supports the hypothesis that direct local trauma and myocardial damage is the principal activator of inflammation, and transfemoral procedures may be less traumatic in this regard.
The time course of the postprocedural increase in inflammatory markers observed in this study correlates with previously reported postoperative hs-CRP elevations occurring between the first and third day following heart surgery.12,21,23 High-sensitive CRP and leukocyte elevations at later points in time may not only be affected by the procedure itself, but also by varying complications such as infections or bleedings. Furthermore, as levels of inflammatory markers remain relatively low following transfemoral procedures, even small alterations may cause substantial relative increases. Furthermore, an increased need for permanent pacemaker implantation within 72 hours after the procedure may at least in part account for a sustained increase in hs-CRP levels and leukocyte counts in transfemorally treated patients. In our patient cohort, permanent pacemaker implantation was needed in 36% of patients following deployment of a TF CV prosthesis, while it was not observed in patients treated with surgical AVR. The majority of patients undergoing AVR or TAVI was on anti-inflammatory medication such as statins and acetylsalicylic acid as coronary artery and peripheral artery disease are frequent comorbidities in this patient population. Due to their anti-inflammatory properties, these drugs may attenuate postprocedural inflammation and thereby favorably impact the postprocedural course.
So far, the clinical value of increased postprocedural inflammatory markers has not fully been elucidated. However, severe inflammation may complicate the postprocedural course, resulting in respiratory failure, renal dysfunction, coagulation disorders, and multiple organ failure. Besides organ dysfunction, increased inflammatory responses observed following AVR and transapical TAVI may further comprise the patient’s self-perceived well-being and lead more frequently to postoperative delirium. Yet, extensive and sustained inflammatory responses can be observed during uncomplicated courses, and might interfere with the diagnosis of infection.23
Study limitations. The limitations of this hypothesis-generating pilot study are its retrospective nature limited to single-center experience, and the relatively small number of patients in each group. The relatively small number of patients was counterbalanced by: (1) the optimal matching for pre- and postprocedural patient characteristics; and (2) the restriction to patients with completely ‘uneventful’ procedures attempting optimal comparison of inflammatory reactions among different procedure types and avoiding the bias of variable comorbidities and periprocedural complications. However, multivariate analyses may be compromised in small study populations.
Conclusion
In conclusion, this study demonstrates that TAVI as previously shown for AVR provokes postprocedural systemic inflammation. This inflammatory response is diminished following transfemoral procedures.
References
- Nkomo VT, Gardin JM, Skelton TN, et al. Burden of valvular heart diseases: a population-based study. Lancet. 2006;368(9540):1005-1011.
- Iung B, Cachier A, Baron G, et al. Decision-making in elderly patients with severe aortic stenosis: why are so many denied surgery? Eur Heart J. 2005;26(24):2714-2720.
- Bach DS, Cimino N, Deeb GM. Unoperated patients with severe aortic stenosis. J Am Coll Cardiol. 2007;50(20):2018-2019.
- Leon MB, Kodali S, Williams M, et al. Transcatheter aortic valve replacement in patients with critical aortic stenosis: rationale, device descriptions, early clinical experiences, and perspectives. Semin Thorac Cardiovasc Surg. 2006;18(2):165-174.
- Webb JG, Chandavimol M, Thompson CR, et al. Percutaneous aortic valve implantation retrograde from the femoral artery. Circulation. 2006;113(6):842-850.
- Cribier A, Eltchaninoff H, Bash A, et al. Percutaneous transcatheter implantation of an aortic valve prosthesis for calcific aortic stenosis: first human case description. Circulation. 2002;106(24):3006-3008.
- Leon MB, Smith CR, Mack M, et al. Transcatheter aortic-valve implantation for aortic stenosis in patients who cannot undergo surgery. N Engl J Med. 2010;363(17):1597-1607.
- Smith CR, Leon MB, Mack MJ, et al. Transcatheter versus surgical aortic-valve replacement in high-risk patients. N Engl J Med. 2011;364(23):2187-2198.
- Levy JH, Tanaka KA. Inflammatory response to cardiopulmonary bypass. Ann Thorac Surg. 2003;75(2):S715-S720.
- Weis F, Beiras-Fernandez A, Schelling G, et al. Stress doses of hydrocortisone in high-risk patients undergoing cardiac surgery: effects on interleukin-6 to interleukin-10 ratio and early outcome. Crit Care Med. 2009;37(5):1685-1690.
- Kastellanos SS, Toumpoulis IK, Aggeli C, et al. Time course of c-reactive protein, tumour necrosis factor-alpha and monocyte chemoattractant protein-1 following the surgical treatment of patients with aortic valve stenosis. Hellenic J Cardiol. 2007;48(1):5-14.
- Bical OM, Fromes Y, Gaillard D, et al. Comparison of the inflammatory response between miniaturized and standard cpb circuits in aortic valve surgery. Eur J Cardiothorac Surg. 2006;29(5):699-702.
- Kilger E, Heyn J, Beiras-Fernandez A, et al. Stress doses of hydrocortisone reduce systemic inflammatory response in patients undergoing cardiac surgery without cardiopulmonary bypass. Minerva Anestesiol. 2011;77:268-274.
- Clarizia NA, Manlhiot C, Schwartz SM, et al. Improved outcomes associated with intraoperative steroid use in high-risk pediatric cardiac surgery. Ann Thorac Surg. 2011;91(4):1222-1227.
- Prat C, Ricart P, Ruyra X, et al. Serum concentrations of procalcitonin after cardiac surgery. J Card Surg. 2008;23(6):627-632.
- Fromes Y, Gaillard D, Ponzio O, et al. Reduction of the inflammatory response following coronary bypass grafting with total minimal extracorporeal circulation. Eur J Cardiothorac Surg. 2002;22(4):527-533.
- Leon MB, Piazza N, Nikolsky E, et al. Standardized endpoint definitions for transcatheter aortic valve implantation clinical trials: a consensus report from the valve academic research consortium. Eur Heart J. 2011;32(2):205-217.
- Vahanian A, Alfieri O, Al-Attar N, et al. Transcatheter valve implantation for patients with aortic stenosis: a position statement from the European Association of Cardio-Thoracic Surgery (EACTS) and the European Society of Cardiology (ESC), in collaboration with the European Association of Percutaneous Cardiovascular Interventions (EAPCI). Eur Heart J. 2008;29(11):1463-1470.
- Patel JH, Mathew ST, Hennebry TA. Transcatheter aortic valve replacement: a potential option for the nonsurgical patient. Clin Cardiol. 2009;32(6):296-301.
- Stähli BE, Bünzli R, Grünenfelder J, et al. Transcatheter aortic valve implantation (TAVI) outcome according to standardized endpoint definitions by the valve academic research consortium (VARC). J Invasive Cardiol. 2011;23(8):307-312.
- Franke A, Lante W, Fackeldey V, et al. Pro-inflammatory cytokines after different kinds of cardio-thoracic surgical procedures: is what we see what we know? Eur J Cardiothorac Surg. 2005;28(4):569-575.
- Butler J, Rocker GM, Westaby S. Inflammatory response to cardiopulmonary bypass. Ann Thorac Surg. 1993;55(2):552-559.
- Karu I, Taal G, Zilmer K, et al. Inflammatory/oxidative stress during the first week after different types of cardiac surgery. Scand Cardiovasc J. 2010;44(2):119-124.
_________________________________________________
From the 1Department of Cardiology and the 2Clinic for Cardiovascular Surgery, Cardiovascular Center, University Hospital Zürich, Switzerland, and 3FB Mathematics and Computer Science, University of Bremen, Germany.
Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. Dr Altwegg has partially been supported by grants of the Swiss National Research Foundation (Special Programme University Medicine: Grant #33CM30-1241112/1 and #3100-068118.02/1) and is a proctor for Edwards Lifesciences. Dr Falk has received lecture honoraria from Medtronic. The other authors report no disclosures.
Manuscript submitted February 13, 2012, provisional acceptance given March 12, 2012, final version accepted April 9, 2012.
Address for correspondence: Lukas A. Altwegg, MD, Department of Cardiology, Cardiovascular Center, University Hospital Zürich, Rämistrasse 100, 8091 Zürich, Switzerland. Email: lukas.altwegg@access.uzh.ch












