ADVERTISEMENT
Hemoptysis Caused by Saphenous Vein Graft Perforation During Percutaneous Coronary Intervention
Download a PDF of this article.
ABSTRACT. Percutaneous coronary intervention carries the risk of coronary artery perforation, which may lead to serious adverse effects such as cardiac tamponade, myocardial infarction, and even death. We describe a patient who developed hemoptysis immediately after saphenous vein graft rupture, which is a rare complication during coronary intervention. Stenting using a covered stent, along with simultaneous administration of protamine, helped seal the vein graft perforation and stop the hemoptysis.
J INVASIVE CARDIOL 2013;25(1):E8-E10
Key words: coronary artery disease, coronary angiography, catheter intervention complications
__________________________________________
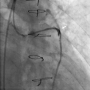
Figure 1. Contrast extravasation with jet stream effect was noted at the dilated site of the saphenous vein graft (SVG; arrowhead) (click thumbnail to view larger image).
Coronary artery perforation is a rarely reported complication of percutaneous coronary intervention (PCI), with an incidence rate ranging from 0.4% to 0.6%.1,2 This complication may result in serious adverse events such as myocardial infarction (MI), cardiac tamponade, and even death. Here, we report a case of saphenous vein graft (SVG) perforation presenting with hemoptysis during PCI. Subsequent implantation of a polytetrafluoroethylene-covered stent helped seal the perforation, and hemoptysis was stopped successfully.
Case report
A 66-year-old man was admitted to our hospital with acute onset of chest tightness for 2 hours. He had a history of hypertension and end-stage renal disease (ESRD) and had undergone hemodialysis. He also had a 3-vessel coronary artery disease and had undergone coronary artery bypass grafting 1 year prior, receiving a sequential SVG to the right coronary artery (RCA), and obtuse marginal (OM) and left internal mammary artery (LIMA) to the left anterior descending artery (LAD). On physical examination, the patient was found to have a blood pressure of 157/98 mm Hg, heart rate of 84/min, and respiratory rate of 20/min. The cardiovascular examination results were unremarkable. A 12-lead electrocardiogram showed ST elevation in leads II, III, and aVF. Laboratory data showed normocytic anemia and poor renal function, which was related to the ESRD. From the ST elevation, we identified the artery causing the inferior wall acute MI and performed emergent coronary angiography within 90 minutes after the admission. The coronary angiography revealed 61% stenosis at the left main artery, 86% stenosis at LAD, and 77% stenosis at the ostium of the RCA. The bypass graft of LIMA to LAD was patent; however, an 87% stenosis at the proximal SVG to OM2 and RCA was noted. A percutaneous transluminal coronary angioplasty for the SVG lesion was performed immediately, using a 6 Fr JR 4 guiding catheter and EXTRA S’PORT wire (Abbott Vascular Devices). The initial activated clotting time (ACT) was 187 s, and the patient was heparinized with a bolus of 10,000 units before the procedure. First, a Sprinter 2.0 × 20 mm balloon (Medtronic) and then a Firestar 3.0 × 30 mm balloon (Johnson & Johnson) were used to dilate the lesion. Because of unsatisfactory results in comparison with the distal SVG size, a Maverick 4.0 × 30 mm balloon (Boston Scientific) was used next. After a single inflation at 10 atm, contrast extravasation with jet stream effect was noted at the site of the dilation (Figure 1).
Although his vital signs were hemodynamically stable, the patient began to develop hemoptysis of fresh blood. Mild tachypnea was noted at the same time, and an oxygen mask (oxygen supply of 8 L/min) was applied without endotracheal intubation. His oxygen saturation was kept higher than 90% under the use of oxygen. The ACT was measured as 480 s. The Firestar 3.0 × 30 mm balloon was inflated at a low pressure (4 atm) to stop the extravasation of blood. The echocardiography performed immediately after did not show any evidence of pericardial effusion. Prolonged balloon inflation failed to stop the extravasation of blood. Therefore, the guiding catheter was changed to 7 Fr JR 3.5, and a GraftMaster 3.0 × 16 mm stent (Abbott Vascular) was deployed to the perforated site by using a maximum pressure of 14 atm. The contrast leakage from the lesion site decreased, but it did not stop completely. Concurrent administration of protamine was used to correct the anticoagulation of heparin, with a final ACT level of 189 s.
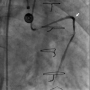
Figure 2. A stent graft was deployed to the perforation site, and the extravasation was successfully reduced (arrowhead) (click thumbnail to view larger image).
This aided in the successful sealing of the perforation (Figure 2) and stopping of the contrast leakage.
The patient’s hemoptysis and tachypnea improved after successful sealing of the perforation. No more hemoptysis was noted throughout the hospital course. Nevertheless, he developed an episode of abnormal liver function, which might be related to a lipid-lowering agent. The lipid-lowering agent was discontinued and the patient’s liver function was checked regularly during admission. He was discharged 16 days later, after the liver function had returned to normal. However, a 320-slice cardiac computed tomographic (CT) image did not show a direct contact between the perforated SVG and the lung parenchyma or bronchial trees (Figure 3).
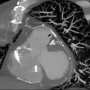
Figure 3. Computed tomographic reconstruction image showing a thin layer of tissue between the SVG and lung parenchyma (arrowhead, stent graft) (click thumbnail to view larger image).
Discussion
PCI is often associated with the risk of complications such as perforation. It usually results from guidewire trauma, balloon rupture, balloon oversizing, or puncture of the arterial wall by a stent edge and may lead to serious adverse effects such as cardiac tamponade, MI, and even death.1,2
Perforation of an SVG during coronary intervention is rarely reported in the medical literature.3-6 Previous reports suggest that the advanced age of vein grafts increases the risk of perforation.3,5,6 In this case, because of an initial unsatisfactory dilation result, we used a larger balloon, which unfortunately caused the perforation. The size of the balloon and reference vessel diameter were 3.89 and 3.16 mm, respectively, which led to the ratio of 1.23. We believe that the advanced age of the SVG and the oversized balloon played a major role in the perforation.
There have been only few reports on hemoptysis caused by an SVG aneurysm (SVGA) rupture previously.7-11 According to previous reports, the size of the ruptured SVGA ranged from approximately 2 to 5 cm. Three major different mechanisms are related to the hemoptysis caused by the rupture of an aneurysm. First, the aneurysm densely adherent to the lung had ruptured with bleeding into the pleura and lung parenchyma. Large hematoma eroding into the lung tissue could cause hemoptysis.9,10 Second, a fistula developed between a vein graft aneurysm and the bronchial trees.7,8 Hemoptysis is caused by bleeding from the aneurysm into a segmental bronchus. Third, the SVGA connected to lung parenchyma with no evidence of fistula.11 To our knowledge, this is the first report of such a case of hemoptysis after an SVG rupture due to coronary intervention. The cardiac CT image showed no evidence of fistula between the vein graft and a bronchus or the existence of direct connection between the proximal SVG and the lung parenchyma or bronchial trees. Only a thin layer of tissue between the SVG and lung parenchyma was noted from the CT reconstruction image; therefore, the SVG may be densely adherent to the lung. The coronary angiography revealed contrast extravasation with jet stream effect at the site of balloon dilation. Comparing the reconstructed CT image with the coronary angiographic image from a similar angle, we speculate that blood jet stream from the SVG graft perforation could have directly leaked into the lung tissue, causing the hemoptysis.
Prolonged balloon inflation is the initial management method for coronary or bypass graft perforation.2,12 An alternative approach is to deploy a covered stent at the site of extravasation. The polytetrafluoroethylene membrane of the covered stent (stent graft) has been validated for patients undergoing intervention for stenosis of a vein graft13 as well as for sealing a perforation.3,4 A 6 Fr guiding catheter is feasible for graft stent with the size of 3.0 to 4.0 mm and a 7 Fr guiding catheter for the size of 4.0 to 5.0 mm. Regarding the lesion with a high risk for perforation, for example, SVG, it would be better to use a larger guiding catheter (7 Fr) to make the graft stent deployment smoothly, and it is an important consideration in procedure preparation.
In this case, we deployed a stent graft to the perforation site and reduced the extravasation successfully. In addition, by reversing the anticoagulant effect of heparin with protamine, we were able to finally seal the perforation and stop the hemoptysis. Mild tachypnea was noted during hemoptysis. By means of the use of an oxygen mask, we were able to keep his oxygen saturation higher than 90% and finally resolve the problem after sealing the perforation. However, if the oxygen mask fails to maintain the optimal oxygen saturation, endotracheal intubation should be considered.
In conclusion, hemoptysis during coronary intervention could be the manifestation of an SVG perforation, and stenting using a covered stent, along with simultaneous administration of protamine is a viable option to seal the vein graft perforation and stop the hemoptysis.
References
- Gunning MG, Williams IL, Jewitt DE, et al. Coronary artery perforation during percutaneous intervention: incidence and outcome. Heart. 2002;88(5):495–498.
- Ellis SG, Ajluni S, Arnold AZ, et al. Increased coronary perforation in the new device era: incidence, classification, management, and outcome. Circulation. 1994;90(6):2725–2730.
- Latsios G, Tsioufis K, Tousoulis D, et al. Perforation of a saphenous vein graft during percutaneous angioplasty: demonstration by means of intravascular ultrasound and consequent treatment with a polytetrafluoroethylene-covered stent. Int J Cardiol. 2009;134(1):e15–e16.
- Hernandez-Antolin RA, Banuelos C, Alfonso F, et al. Successful sealing of an angioplasty-related saphenous vein graft rupture with a PTFE-covered stent. J Invasive Cardiol. 2000;12(11):589–593.
- Subraya RG, Tannenbaum AK. Successful sealing of perforation of saphenous vein graft by coronary stent. Catheter Cardiovasc Interv. 2000;50(4):460–462.
- Shammas NW, Thondapu VR, Winniford MD, Kalil DA. Perforation of saphenous vein graft during coronary stenting: a case report. Cathet Cardiovasc Diagn. 1996;38(3):274–276.
- Nielsen JF, Stentoft J, Aunsholt NA. Haemoptysis caused by aneurysm of saphenous bypass graft to a coronary artery. Scand J Thorac Cardiovasc Surg. 1988;22(2):189–191.
- Dieter RS, Patel AK, Yandow D, et al. Conservative vs. invasive treatment of aortocoronary saphenous vein graft aneurysms: treatment algorithm based upon a large series. Cardiovasc Surg. 2003;11(6):507–513.
- Nishimura Y, Okamura Y, Hiramatsu T, et al. Hemoptysis caused by saphenous vein graft aneurysm late after coronary artery bypass grafting. J Thoracic Cardiovasc Surg. 2005;129(6):1432–1433.
- Ivert T, Orre L. Right thoracotomy for saphenous vein graft aneurysm causing hemoptysis. Ann Thorac Surg. 2006;81(5):1885–1887.
- Jimenez-Jaimez J, Molina E, Melgares R. Saphenous vein graft aneurysm connected to lung parenchyma: a very unusual cause of haemoptysis. Eur Heart J. 2011;32(11):1330.
- Ajluni SC, Glazier S, Blankenship L, et al. Perforations after percutaneous coronary interventions: clinical, angiographic, and therapeutic observations. Cathet Cardiovasc Diagn. 1994;32(3):206–212.
- Schachinger V, Hamm CW, Munzel T, et al. A randomized trial of polytetrafluoroethylene-membrane-covered stents compared with conventional stents in aortocoronary saphenous vein grafts. J Am Coll Cardiol. 2003;42(8):1360–1369.
__________________________________________
From the Second Section of Cardiology, Department of Medicine, Chang Gung Memorial Hospital, Chang Gung University College of Medicine, Taoyuan, Taiwan.
Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. The authors report no conflicts of interest regarding the content herein.
Manuscript submitted May 15, 2012, provisional acceptance given June 26, 2012, final version accepted July 26, 2012.
Address for correspondence: I-Chang Hsieh, MD, No.5, Fu-Hsing Street, Kwei-Shan, Taoyuan, Taiwan. Email: hsiehic@ms28.hinet.net











