Access and Closure Techniques with the Impella Left Ventricular Assist Device
 Over the last decade, significant developments have been made in the treatment of heart disease, including the introduction of percutaneous left ventricular assist technologies and percutaneous repair catheters for structural heart disease and endovascular aortic repair. Typically, these procedures require placement of large arterial sheaths (13-24 French [Fr]) for delivery. Meticulous vascular access is essential for successful large-vessel sheath insertion and to minimize vascular complications. The Impella 2.5 (Abiomed, Inc.) device is a 12 Fr intravascular microaxial blood pump that delivers up to 2.5L/min of cardiac output and is utilized in complex coronary interventions. The Impella 5.0 is a 21 Fr miniaturized rotary blood pump that is capable of delivering up to 5L/min, which most often requires surgical implantation. Herein is a description of various techniques for vascular access and closure with the Impella devices.
Over the last decade, significant developments have been made in the treatment of heart disease, including the introduction of percutaneous left ventricular assist technologies and percutaneous repair catheters for structural heart disease and endovascular aortic repair. Typically, these procedures require placement of large arterial sheaths (13-24 French [Fr]) for delivery. Meticulous vascular access is essential for successful large-vessel sheath insertion and to minimize vascular complications. The Impella 2.5 (Abiomed, Inc.) device is a 12 Fr intravascular microaxial blood pump that delivers up to 2.5L/min of cardiac output and is utilized in complex coronary interventions. The Impella 5.0 is a 21 Fr miniaturized rotary blood pump that is capable of delivering up to 5L/min, which most often requires surgical implantation. Herein is a description of various techniques for vascular access and closure with the Impella devices.
 It has been almost sixty years since Seldinger (1953)1 first described his technique for gaining access to a vessel. Yet it is only in recent years that certain steps to improve the technique and minimize complications have become more widely applied. In addition to acknowledging the need for an anterior wall puncture, techniques are now designed to avoid ‘high sticks’ above the inferior-most sweep of the epigastric artery which marks the inguinal ligament (predisposing patients to an eighteen-fold increase in retroperitoneal hemorrhage) and ‘low sticks’ below the femoral bifurcation into the superficial femoral artery and profunda femoral artery [predisposing patients to pseudoaneurysms and arterio-venous (AV) fistulas].
It has been almost sixty years since Seldinger (1953)1 first described his technique for gaining access to a vessel. Yet it is only in recent years that certain steps to improve the technique and minimize complications have become more widely applied. In addition to acknowledging the need for an anterior wall puncture, techniques are now designed to avoid ‘high sticks’ above the inferior-most sweep of the epigastric artery which marks the inguinal ligament (predisposing patients to an eighteen-fold increase in retroperitoneal hemorrhage) and ‘low sticks’ below the femoral bifurcation into the superficial femoral artery and profunda femoral artery [predisposing patients to pseudoaneurysms and arterio-venous (AV) fistulas].
Traditional anatomic landmark guidance for femoral access using the inguinal skin crease, maximal femoral pulse, and bony landmarks are being abandoned for fluoroscopy-guided access.2 Fluoroscopy-guided access of the common femoral artery at a ‘sweet spot’ or ‘cumulative target zone’ 5-14 mm below the middle of the femoral head3 is increasingly accepted as the standard practice to reduce complications.4 The use of adjunctive ultrasound guidance, though often practiced in radiology labs, is not as well adopted in cardiology labs. Further improvements, including use of a micropuncture needle and contralateral angiography techniques, can be used to further minimize complications while optimizing accuracy.5,6
Identifying individual patient risk factors is an important aspect of care during cardiac catheterization and vascular access.7 The following list summarizes patient characteristics that may increase the risk for development of vascular complications:
- Age >70
- Female gender
- Thin patients
- Morbid obesity
- Presence of peripheral vascular disease
- Hypertension
- Renal failure
- Low platelet count and low hematocrit at baseline
- Congestive heart failure
- Chronic obstructive pulmonary disease
- Coagulopathies
Once vascular access is obtained, it is important to perform an angiogram to evaluate iliofemoral dimensions, peripheral vascular disease burden, and vessel tortuosity, and to identify prior vascular surgeries. Alternatively, in settings in which the Impella is a supportive adjunct to an elective or semi-elective procedure, evaluation of the vascular anatomy using computed tomography (CT) angiography or another non-invasive modality can be used to assess the more accommodating iliofemoral route, taking into account vessel dimension, calcium burden, and tortuosity. The Impella 2.5 device has a maximal diameter of 12 Fr at the motor; thus, placement of a 13-14 Fr (4.3–4.7 mm) sheath is required. It is recommended that the femoral artery be at least 6 mm in diameter to help avoid lower extremity ischemia. For patients with tortuous iliac arteries, or in those patients who need percutaneous interventions with stent implantation to the iliac artery, a 30 cm, 14 Fr sheath can be placed in the terminal aorta to aid in passing the Impella device. The 13 Fr repositioning sheath incorporated with the Impella device is useful in those patients who may be maintained on prolonged support.
The Impella 2.5
Manual closure is entirely acceptable for the Impella 2.5 device. Manual compression has been the standard for sheath removal for decades. Typically, the activated clotting time should be <160-180 seconds for safe sheath removal. Direct pressure is applied, with the fingers positioned over and proximal to the arteriotomy site, while maintaining a faint distal pulse. There are a variety of mechanical compression devices such as the FemoStop (St. Jude Medical) and CompressAR system (Advanced Vascular Dynamics) to assist in those patients needing prolonged pressure owing to large sheath size. Most studies have shown lower vascular complications with mechanical compression devices, although one study found that better results were achieved with manual pressure. The FemoStop uses an inflatable bladder to apply pressure at the arteriotomy site. Near systolic pressure should be applied for 15-30 minutes, and the pressure then decreases by 10-15 mm Hg every 10-20 minutes. It is important to maintain vigilance when these external devices are utilized, as arterial and venous thrombosis can occur from too high a pressure or prolonged use.8-10
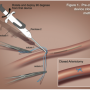 The increase in large-bore access procedures is resulting in an increasing interest and experience with vascular closure devices.11 In settings in which the Impella is a supportive adjunct to the procedure and its removal is intended at the end of the procedure, a modified pre-close approach can be adopted. Very little prospective data on how best to select patients for vascular closure has been published, but the consensus opinion is that fluoroscopic guidance can help assist in planning vascular closure.12,13 Here again, non-invasive imaging, such as computed tomography (CT) angiography, may also enhance selection of patients for vascular closure. Applied methodically and systematically, a vascular closure approach has been reported to improve aid in hemostasis in endovascular aortic repair (EVAR).14,15 The choice of device dictates the strategy in terms of sequence of placement and number of devices to be placed.16 Choice of device depends largely on experience and the literature reports success with several devices, including the Prostar XL, Starclose, and Perclose (all from Abbott Vascular).17-19 The “pre-close” technique (our preferred technique) involves deploying two Perclose devices at the arteriotomy site prior to placing large caliber sheaths (Figure 1). An 18-gauge needle is used to cannulate the common femoral artery under fluoroscopy along its anterior aspect at the mid-level of the femoral head. An 0.035-inch wire is introduced into the vessel, then two 6 Fr Perclose Proglide devices are placed. The first device is placed at a 30°–45° angle and then deployed in the standard manner. The Perclose suture strands are extracted from the device and tagged with hemostatic forceps. Before complete removal of the first carrier device, the 0.035-inch guide wire is reinserted into the femoral artery via a marked monorail wire tube in the first device. A second Perclose device is then introduced at a 90˚ angle to the first device and deployed. The sutures are again secured with a hemostat. The guide wire is reinserted into the device and then sheaths can then be advanced into the femoral artery. At the end of the case, the sutures are cinched down after catheter removal in a sequential manner to close the arteriotomy.
The increase in large-bore access procedures is resulting in an increasing interest and experience with vascular closure devices.11 In settings in which the Impella is a supportive adjunct to the procedure and its removal is intended at the end of the procedure, a modified pre-close approach can be adopted. Very little prospective data on how best to select patients for vascular closure has been published, but the consensus opinion is that fluoroscopic guidance can help assist in planning vascular closure.12,13 Here again, non-invasive imaging, such as computed tomography (CT) angiography, may also enhance selection of patients for vascular closure. Applied methodically and systematically, a vascular closure approach has been reported to improve aid in hemostasis in endovascular aortic repair (EVAR).14,15 The choice of device dictates the strategy in terms of sequence of placement and number of devices to be placed.16 Choice of device depends largely on experience and the literature reports success with several devices, including the Prostar XL, Starclose, and Perclose (all from Abbott Vascular).17-19 The “pre-close” technique (our preferred technique) involves deploying two Perclose devices at the arteriotomy site prior to placing large caliber sheaths (Figure 1). An 18-gauge needle is used to cannulate the common femoral artery under fluoroscopy along its anterior aspect at the mid-level of the femoral head. An 0.035-inch wire is introduced into the vessel, then two 6 Fr Perclose Proglide devices are placed. The first device is placed at a 30°–45° angle and then deployed in the standard manner. The Perclose suture strands are extracted from the device and tagged with hemostatic forceps. Before complete removal of the first carrier device, the 0.035-inch guide wire is reinserted into the femoral artery via a marked monorail wire tube in the first device. A second Perclose device is then introduced at a 90˚ angle to the first device and deployed. The sutures are again secured with a hemostat. The guide wire is reinserted into the device and then sheaths can then be advanced into the femoral artery. At the end of the case, the sutures are cinched down after catheter removal in a sequential manner to close the arteriotomy.
A further measure of control can be gained in this setting, if there is already contralateral access, by placing a crossover sheath to provide the option of contralateral balloon tamponade in the event of failure of the closure elements. This is accomplished by using a diagnostic internal mammary catheter to cannulate, from the contralateral access, the ipsilateral common iliac artery that holds the large caliber sheath. A 0.035-inch hydrophilic wire is then advanced into the ipsilateral superficial femoral artery, and the diagnostic catheter is advanced over the wire beyond the larger caliber sheath. An 0.035 stiff wire (e.g., Amplatz, Cook Medical) wire is advanced through the diagnostic catheter, and then the catheter is removed. Over the stiff wire, a 6-7 Fr crossover sheath is advanced into the ipsilateral external iliac artery. A peripheral balloon (typically 6-8 mm in diameter) is then advanced to the arteriotomy and inflated to tamponade the site. Also, to decrease bleeding during large-caliber sheath removal, a peripheral balloon can be placed in the proximal iliac artery. It is inflated at a low pressure while the catheter is removed and the arteriotomy is closed with the Perclose sutures.19
Contralateral balloon tamponade closure can also be contemplated in a non pre-close setting to assist manual or surgical closure, and perhaps in the future, alternative device closure, though currently there is no percutaneous device able to take advantage of this in a non pre-close setting.
The Impella 5.0
The Impella 5.0 was originally designed for surgical insertion in the right femoral artery through a 3-cm incision. After hemostatic control is achieved with a purse-string suture, a 6 Fr all-purpose diagnostic catheter such as an Amplatz or a pigtail is advanced across the aortic valve under fluoroscopy. With fluoroscopic and/or transesophageal echocardiographic guidance, the Impella 5.0 LP pump can then be positioned across the aortic valve, using an 0.21-inch or a stiff 0.14-inch guidewire. Alternatively, an 8 mm or 10 mm Dacron graft can be anastomosed to the femoral artery in an end-to-side fashion.20
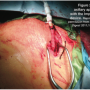 A novel approach involves using the axillary artery as the access vessel (Figure 2). The patient is taken to the operating room or hybrid suite, where the surgeon makes a small incision below the left clavicle and the axillary artery is exposed. The patient is heparinized for an activated clotting time (ACT) > 200 seconds. Vessel loops are placed proximally and distally around the artery. A 10 mm Dacron graft is spatulated and anastomosed with the artery, using a running 4-0 suture. A 9 Fr sheath can be tied to the end of the graft to prevent blood loss. Some advocate using a 24 Fr DrySeal Sheath (Gore Medical), which is trimmed and placed into the Dacron graft to allow for placement of the Impella device with minimal blood loss. Through the sheath, a diagnostic Amplatz catheter is placed into the left ventricle, and an 0.014-inch stiff guidewire is passed though the catheter. The 9 Fr sheath and Amplatz catheter are removed, and the Dacron graft and wire are clamped. The Impella 5.0 device is back-loaded onto the stiff 0.025-inch wire and placed under fluoroscopic guidance into the left ventricle.20
A novel approach involves using the axillary artery as the access vessel (Figure 2). The patient is taken to the operating room or hybrid suite, where the surgeon makes a small incision below the left clavicle and the axillary artery is exposed. The patient is heparinized for an activated clotting time (ACT) > 200 seconds. Vessel loops are placed proximally and distally around the artery. A 10 mm Dacron graft is spatulated and anastomosed with the artery, using a running 4-0 suture. A 9 Fr sheath can be tied to the end of the graft to prevent blood loss. Some advocate using a 24 Fr DrySeal Sheath (Gore Medical), which is trimmed and placed into the Dacron graft to allow for placement of the Impella device with minimal blood loss. Through the sheath, a diagnostic Amplatz catheter is placed into the left ventricle, and an 0.014-inch stiff guidewire is passed though the catheter. The 9 Fr sheath and Amplatz catheter are removed, and the Dacron graft and wire are clamped. The Impella 5.0 device is back-loaded onto the stiff 0.025-inch wire and placed under fluoroscopic guidance into the left ventricle.20
We specifically like the axillary approach. It allows for a more stable placement, avoids the problems of significant iliofemoral atherosclerosis and/or tortuosity, and obviates the need for the supine position. The device is then removed and the axillary artery is repaired by the surgeon.
Summary
Large-caliber vascular sheath interventions are expected to rise dramatically with percutaneous structural heart disease technologies entering the market and increased adoption of left ventricular assist devices like the Impella. Access site complications can be minimized with proper patient screening and meticulous vascular access techniques. Manual compression, the use of vascular closure devices in the pre-close technique, and surgical closure provide a safe array of options for access site management after explantation of Impella devices.
Dr. Grise may be contacted at markgrise@gmail.com.
References
- Seldinger S. Catheter placement of the needle in percutaneous arteriography. A new technique. Acta Radiol 1953;39:368-376.
- Grier D, Hartnell G. Percutaneous femoral artery puncture: practice and anatomy. Br J Radiol 1990;63(752):602-604.
- Turi ZG. An evidence-based approach to femoral arterial access and closure. Rev Cardiovasc Med 2008;9(1):7-18.
- Fitts J, Ver Lee P, Hofmaster P, Malenka D. Fluoroscopy-guided femoral artery puncture reduces the risk of PCI-related vascular complications. J Interv Cardiol 2008;21(3):273-278.
- Seto AH, Abu-Fadel MS, Sparling JM, Zacharias SJ, et al. Real-time ultrasound guidance facilitates femoral arterial access and reduces vascular complications: FAUST (Femoral Arterial Access With Ultrasound Trial). JACC Cardiovasc Interv 2010;3(7):751-758.
- Cilingiroglu M, Feldman T, Salinger MH, Levisay J, Turi ZG. Fluoroscopically-guided micropuncture femoral artery access for large-caliber sheath insertion. J Invasive Cardiol 2011;23(4):157-161.
- Dumont CJ, Keeling AW, Bourguignon C, et al. Predictors of vascular complications post diagnostic cardiac catheterization and percutaneous coronary interventions. Dimens Crit Care Nurs 2006 May-Jun;25(3):137-142.
- Pracyk JB, Wall TC, Longabaugh et al. A randomized trial of vascular hemostasis techniques to reduce femoral vascular complications after coronary intervention. Am J Cardiol 1998; 81: 970-976.
- Semler HJ. Transfemoral catheterization: mechanical versus manual control of bleeding. Radiology 1985; 154: 234-235.
- Bogart MA. Time to hemostasis: a comparison of manual versus mechanical compression of the femoral artery. Am J Crit Care 1995;4:149-156.
- Turi ZG. Closing in on large hole closure. Catheter Cardiovasc Interv 2011;78(1):125-126.
- Abu-Fadel MS, Sparling JM, Zacharias SJ, Aston CE, et al. Fluoroscopy vs. traditional guided femoral arterial access and the use of closure devices: a randomized controlled trial. Catheter Cardiovasc Interv 2009;74(4):533-539.
- Turi ZG. Fluoroscopy guided vascular access: asking the right question, but getting the wrong answer? Catheter Cardiovasc Interv 2009;74(4):540-542.
- Goodney PP, Chang RW, Cronenwett JL. A percutaneous arterial closure protocol can decrease complications after endovascular interventions in vascular surgery patients. J Vasc Surg 2008;48(6):1481-1488.
- Lee WA, Brown MP, Nelson PR, Huber TS, Seeger JM. Midterm outcomes of femoral arteries after percutaneous endovascular aortic repair using the Preclose technique. J Vasc Surg 2008;47(5):919-923.
- Hogg ME, Kibbe MR. Percutaneous thoracic and abdominal aortic aneurysm repair: techniques and outcomes. Vascular 2006;14(5):270-281.
- Sharp AS, Michev I, Maisano F, Taramasso M, et al. A new technique for vascular access management in transcatheter aortic valve implantation. Catheter Cardiovasc Interv 2010;75(5):784-793.
- Kahlert P, Eggebrecht H, Erbel R, Sack S. A modified “preclosure” technique after percutaneous aortic valve replacement. Catheter Cardiovasc Interv 2008;72(6):877-884.
- Badawi RA, Collins TJ, Ramee SR. A simple percutaneous technique for hemostasis and closure after transcatheter aortic valve implantation. Catheter Cardiovasc Interv 2012;79(1):152-155
- Grise, MA. Escalation of care for the decompensated heart failure patient: The Ochsner Heart and Vascular Institute experience with axillary placement of Impella 5.0. Cath Lab Digest Aug 2011;19(8):1-10.
Disclosure: Dr. Grise reports he is a paid speaker for Abiomed. Dr. Badawi reports no conflicts of interest regarding the content herein.










